Healthcare Providers’ Perceptions of the Utility of Psychosocial Screening Tools in Childhood Cancer: A Pilot Study
Purpose/Objectives: To examine the perceptions of healthcare providers (HCPs) regarding the utility of two psychosocial screening tools designed for pediatric oncology, the Psychosocial Assessment Tool–Revised (PATrev)and the Psychosocial Care Checklist (PCCL).
Design: Repeated measures comparative study.
Setting: Four pediatric health centers in Ontario, Canada.
Sample: 15 oncologists, 14 nurses, and 8 social workers.
Methods: Using a visual analog scale (VAS), participants were asked to rate how useful they found (a) the psychosocial summary derived from the parent-completed PATrev, used to assess family psychosocial risk, and (b) the HCP-completed PCCL, used to identify family psychosocial needs. Measures were completed soon after diagnosis and six months later. Mann-Whitney U tests were used for analyses.
Main Research Variable: VAS scores.
Findings: Pediatric oncology HCPs differ in their acceptance of the psychosocial screening tools tested. The highest utility ratings for both instruments were from nurses, and the lowest utility ratings were from social workers; moderate ratings were obtained from oncologists.
Conclusions: Psychosocial screening tools can identify the psychosocial needs of children with cancer and their families throughout the cancer trajectory. Consequently, these tools could foster communication among colleagues (medical and nonmedical) who are caring for children with cancer about the psychosocial needs of this population and the allocation of resources to address those needs.
Implications for Nursing: Nurses seem to value these tools more than other HCPs, which may have positive implications for their clinical practice.
Jump to a section
Pediatric cancer care in most developed countries is based on the premise that the child’s medical treatment needs to be provided in the context of the family, and many programs endorse a child- and family-centered care approach (Kazak, Simms, & Rourke, 2002; Wiener & Pao, 2012). In spite of substantial evidence in the field regarding the psychosocial effects of childhood cancer on the affected child and family (Alderfer & Hodges, 2010; Barrera, Atenafu, Doyle, Berlin-Romalis, & Hancock, 2012; Bearden, Feinstein, & Cohen, 2012; Boman, Lindahl, & Björk, 2003; Dolgin et al., 2007; Kazak et al., 2004; Kazak, Boeving, Alderfer, Hwang, & Reilly, 2005; Kazak et al., 2006; Rodriguez et al., 2012), research on psychosocial screening in pediatric oncology is in its infancy, and tools to screen for distress or psychosocial risk in this population are rare (Kazak et al., 2001, 2007; Pai et al., 2007, 2008).
In addition, evidence exists regarding healthcare providers’ (HCPs’) limited use of these tools to document (a) their knowledge of psychosocial difficulties in the patient and his or her family and (b) how useful they find these tools (Mitchell, Clarke, & Sloper, 2005; Selove, Kroll, Coppes, & Cheng, 2012). Early psychosocial screening can guide interventions to reduce or prevent adverse psychosocial outcomes and to foster better use of resources in clinical practice (Kazak et al., 2007).
Two psychosocial screening tools have been used in pediatric oncology (Patenaude & Kupst, 2005): the Distress Thermometer (DT) (National Comprehensive Cancer Network [NCCN], 2003) and the Psychosocial Assessment Tool (PAT) (Kazak et al., 2001, 2007, 2012; Pai et al., 2008). The DT is widely used for screening in adult cancer (Bultz et al., 2011; Carlson, Waller, Groff, Zhong, & Bultz, 2012; Holland & Bultz, 2007; NCCN, 2003) and was adapted for children (Patel et al., 2011). The current authors chose to work with the PAT because (a) the tool is intended to be completed by the family members (e.g., parents, siblings) of children newly diagnosed with cancer (Kazak et al., 2001, 2007, 2012; Pai et al., 2008) and (b) evidence suggests that families who complete this tool receive more psychosocial care (Kazak et al., 2011).
The PAT is well suited for implementation of family-centered care. A child- and family-centered care approach considers the importance of the family in the child’s development and care. Such an approach also recognizes the reciprocal influences within the family and, therefore, the impact of the disease and its treatment on family members; in addition, it acknowledges the benefits of developing a partnership of care with the patient and family members.
The current authors validated the PAT for use in a Canadian sample, with the revised version referred to as the PATrev (Barrera, Hancock, Rokeach, Cataudella, et al., 2014). The original and revised versions of the PAT were found to have strong psychometric properties. In addition, provision of a psychosocial risk summary (derived from the PATrev and completed by parents) to the HCPs treating the child resulted in improved quality of life in relation to pain for the child (n2 = 0.08) and reduced anxiety for the parent (n2 = 0.08) (Barrera, Hancock, Rokeach, Atenafu, et al., 2014). To assess HCPs’ knowledge of child and family psychosocial needs, the authors developed the Psychosocial Care Checklist (PCCL) (Barrera et al., 2011). Preliminary evaluation of the PCCL’s psychometric properties has been conducted (Barrera et al., 2011); these properties are described in some detail in the methods section. Using the PCCL, the authors found that social workers reported more knowledge of psychosocial problems than did nurses or oncologists (Barrera et al., 2011).
The purpose of the current pilot study was to assess HCPs’ perceptions of the utility of psychosocial screening tools that could be integrated into clinical practice (i.e., a summary of the parent-completed PATrev and the PCCL). Acceptance was assessed by measuring how useful these instruments were perceived to be. Systematic assessment of acceptance and barriers to using clinical tools is an important and often ignored step in the knowledge translation of clinical tools.
Methods
Participants
Study participants were HCPs (i.e., oncologists, nurses, and social workers) of (a) a child newly diagnosed with cancer who was expected to survive at least six months postdiagnosis and (b) parents who had completed the PATrev within two to four weeks after the child’s cancer diagnosis as part of a larger project. Eighty-six HCPs treating a total of 49 families from four pediatric health centers in Ontario, Canada (Hospital for Sick Children in Toronto, London Health Sciences Centre in London, Children’s Hospital of Eastern Ontario in Ottawa, and Kingston General Hospital in Kingston), were approached to participate.
Procedure
This study was conducted as part of a larger study. Ethics approval was obtained at each of the participating health centers prior to study initiation. Individuals identified as the primary care provider to a child who had been diagnosed with cancer and was two to four weeks postdiagnosis were approached to participate. Participating HCPs completed the PCCL four to eight weeks after the diagnosis of the child, received a summary of the findings from the PATrev (Barrera, Hancock, Rokeach, Cataudella, et al., 2014), and were asked to rate how useful they found each tool using a visual analog scale (VAS).
Measures
The PATrev is a standardized, parent-completed psychosocial screening tool, validated with a Canadian sample and used to assess psychosocial risk and problems in children with cancer, as well as their parents and siblings (Barrera, Hancock, Rokeach, Cataudella, et al., 2014). The inter-rater and test-retest reliability of this tool were found to be r = 0.77 and r = 0.75, respectively, whereas the internal consistency was r = 0.85. The PATrev has 15 sections that ask for information about seven psychosocial domains: family structure and resources, social support, child problems, sibling problems, caregiver stress reactions, family problems, and family beliefs. The total score is derived by summing the total of endorsed problematic items in each subscale and mapping that total onto one of the three levels of risk: universal (typical risk), targeted (medium risk), and clinical (severe risk) (Pai et al., 2008). The psychosocial summary profile reports the family risk level and whether the family identified “no or few stressors” (typical risk), “some stressors” (medium risk), or “many stressors” (high risk); the profile also states which specific items were endorsed by the family. For example, if family members indicated that they were having financial difficulties, this was specifically stated on the form. HCPs receiving this summary profile are then encouraged to share the information with other team members involved in the child’s care.
The PCCL (Barrera et al., 2011) is a tool designed to be completed by HCPs to assess the psychosocial needs of children with cancer and their families. The PCCL consists of 21 items (seven for the child, seven for the child’s sibling, if applicable, and seven for the child’s family as a whole). HCPs indicate “yes” or “no” for each item, yielding a total score for the family and subdomain scores for the child, family, and siblings. Examples of items include “child withdrawn,” “moody,” “child acting out,” and “child social problems.” The PCCL also documents any actions taken to address observed concerns. Preliminary psychometrics indicate that alpha coefficients for the total domain and family and sibling subdomains of the PCCL were strong (alpha = 0.9, 0.74, and 0.96, respectively) (Barrera et al., 2011). Test-retest reliability interclass correlation coefficients ranged from 0.65–0.88. However, inter-rater reliability was mixed, ranging from 0.04–0.65, suggesting differences in knowledge of psychosocial concerns by HCPs. Additional analyses confirmed the differential knowledge of the family psychosocial needs among HCPs (Barrera, Yogalingham, et al., 2014). Construct validity proportion of agreement between mothers and HCPs ranged from 0.7–0.84, whereas agreement between fathers and HCPs ranged from 0.7–0.92.
A VAS is an instrument that measures a characteristic or attitude that is believed to range across a continuum of values. In the current study, the VAS was a 10 cm horizontal line drawn on a page, with 0 representing the minimum score (anchor of “not useful at all”) and 10 the maximum score (anchor of “extremely useful”), endorsed for the utility of the measure. For each of the two instruments examined in this study, HCPs were instructed to mark a vertical line through the section of the VAS that corresponded best to their opinion. Specifically, they were asked (a) “How useful was the information provided by the research team regarding the PATrev results?” and (b) “How useful did you find the PCCL?”
Data Analysis
The VAS utility scores were measured and then summed; mean and standard deviation scores were calculated for the PATrev and the PCCL, stratified by HCP discipline. VAS scores for the PATrev and PCCL descriptive data are reported for Time 1 (T1) and Time 2 (T2). Because of small sample size and unequal variances, the Mann-Whitney U test was used to compare VAS scores by HCP discipline: oncologists versus social workers, oncologists versus nurses, and social workers versus nurses. Effect sizes (r) are reported, where 0.3 is the criterion for medium effect size and 0.5 for large effect size (Field, 2009). Change within HCP group over time could not be tested because of insufficient T2 sample sizes. In addition, comparisons of VAS scores by HCP discipline at T2 could be conducted only for the PCCL because of the small sample for the PATrev VAS scores.
Results
Thirty-seven HCPs (15 oncologists, 14 nurses, 8 social workers) consented to participate in the current study. Because a HCP had to complete the PCCL for more than one family under his or her care, this resulted in 72 PCCLs (34 by oncologists, 26 by nurses, 12 by social workers). Given that each HCP’s PCCL was specific to a family, each response was considered to be an independent observation. Although no HCPs actively declined to participate, a number of them (8 oncologists and 6 nurses) simply did not complete the PCCL.
The age range of HCPs varied greatly; the majority of oncologists were aged 40–59 years. The age range for nurses and social workers was wider: 30–60 years of age. All nurses and social workers were women; eight oncologists were women, and seven were men.
Table 1 provides the VAS mean utility ratings, stratified by assessment time point, HCP discipline, and measure. At T1, a total of 35 VAS utility ratings for the PATrev (16 by oncologists, 12 by nurses, 7 by social workers) and 68 VAS utility ratings for the PCCL (33 by oncologists, 25 by nurses, 10 by social workers) were obtained. At T2, a total of 10 VAS utility ratings for the PATrev (1 by an oncologist, 7 by nurses, 2 by social workers) and 27 utility ratings for the PCCL (8 by oncologists, 16 by nurses, 3 by social workers) were obtained. [[[TABLE1]]]
Utility Ratings for the Psychosocial Assessment Tool–Revised
At T1, utility ratings for the PATrev were significantly higher for nurses (Median [Mdn] = 13.33) than for social workers (Mdn = 4.29) (U = 2, p = 0.001, r = 0.83). Similarly, oncologists rated the PATrev as significantly more useful (Mdn = 14.28) than did social workers (Mdn = 6.79; U = 19.5, p = 0.015, r = 0.62). No significant differences were found between oncologists’ (Mdn = 12.41) and nurses’ (Mdn = 17.29) utility ratings at T1 (U = 62.5, p = 0.12) (see Figure 1). 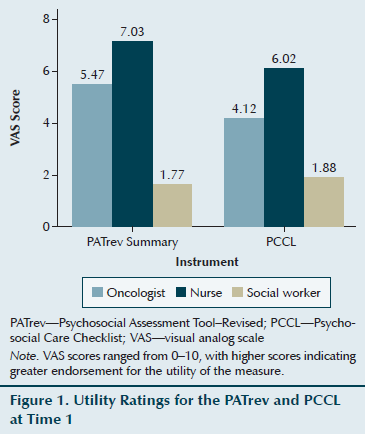
Utility Ratings for the Psychosocial Care Checklist
At T1, utility ratings for the PCCL were significantly greater for nurses (Mdn = 22.06) than for social workers (Mdn = 7.85) (U = 23.5, p < 0.001, r = 0.83). Oncologists rated the PCCL as significantly more useful (Mdn = 24.61) than did social workers (Mdn = 13.4) (U = 79, p = 0.01, r = 0.62). Nurses rated the PCCL as significantly more useful (Mdn = 38.36) than did oncologists (Mdn = 22.79) (U = 191, p = 0.001, r = 0.36). At T2, nurses rated the utility of the PCCL significantly greater (Mdn = 11.19) than did social workers (Mdn = 3.67) (U = 5, p = 0.03, r = 0.65). However, at T2, no significant differences in utility ratings of the PCCL were found between oncologists (Mdn = 6.94) and social workers (Mdn = 3.5) (U = 4.5, p = 0.125) or oncologists (Mdn = 8.81) and nurses (Mdn = 14.34) (U = 34.5, p = 0.071).
Discussion
This pilot study identified differences among HCPs’ perception of the utility of two psychosocial screening tools designed for use in pediatric oncology: the PATrev and the PCCL. A consistent hierarchical pattern emerged from the data, where the highest utility ratings for both instruments were from nurses and the lowest from social workers; moderate ratings were obtained from oncologists. Although these differences seem to make sense given the primary focus of each discipline (for example, as part of their professional role, social workers are expected to seek information about the psychosocial needs of their patients), they represent a previously undocumented and potentially significant barrier to implementation and uptake of psychosocial screening tools in pediatric oncology. A review by Carlson, Waller, and Mitchell (2012) strongly recommended the involvement of frontline clinicians and stakeholders, as well as the provision of training for frontline staff in development and delivery of psychosocial screening. The information from the current study adds to this knowledge base, suggesting that special care should be directed toward identifying the discipline-specific needs of frontline providers to foster effective and clinically meaningful psychosocial knowledge translation regarding these clinical tools.
The current pilot data also suggest that psychosocial screening tools may be most useful for informing nurses within the treating team. The usefulness of the tools may have been rated highest for nurses because they tend to have the most contact with the target child and family and, consequently, a greater need for information about psychosocial risk. However, social workers may have rated the psychosocial screening tools least useful because the nature of their role suggests their having a strong sense of the target child’s and family’s psychosocial needs. One factor that was not controlled for in this study was whether the social worker had met and assessed the family before receiving information about psychosocial risk and before completing the PCCL and assessing the tool’s utility.
Although the sample size was insufficient to adequately explore utility ratings in the postacute period (T2) for the PATrev, the hierarchical pattern was maintained for PCCL ratings at T2: Nurses continued to endorse the greatest utility, social workers the least, and oncologists in the middle. What changed at T2 was the strength of this hierarchical relationship; at T2, the differences between HCP ratings lost their statistical significance, except for comparisons between nurses and social workers. This suggests that the HCPs may have endorsed utility with similar strength. In addition, the data show a slight reduction in scores across all HCPs at T2. The familiarity with the instrument may have reduced the overall impact and, consequently, the utility of the ratings. The PCCL may be most effective when used during the acute period as a method to enable the initial discussion and provision of these psychosocial services.
Efforts directed at how best to integrate psychosocial screening tools into pediatric oncology are required to bridge the gap between standards of psychosocial screening in adult versus pediatric cancer. In adult cancer care, screening for distress during cancer treatment is becoming an essential component of care (Carlson et al., 2012). Distress screening has been accepted as the “sixth vital sign” and as a driver to achieve person-centered care (Bultz, Groff, & Fitch, 2009). Efforts to enhance knowledge translation and uptake of psychosocial screening tools in pediatric oncology are emerging (Kazak et al., 2011), with electronic communication becoming the best vehicle in these efforts to improve patient outcomes (Haverman et al., 2013). Specifically, the acceptance and use of psychosocial screening tools in pediatric oncology would inform HCPs of the child’s and family’s psychosocial needs. This would guide implementation of interventions to reduce or prevent adverse psychosocial outcomes and of triage services to families in need; it also would foster better uses of resources in clinical practice throughout the course of cancer treatment. Pediatric patients and their families deserve appropriate identification of psychosocial distress and access to the provision of psychosocial services, both of which should be within reach of all families of children with cancer.
Results of this pilot study document differences among HCPs in their views regarding two psychosocial screening tools: the psychosocial summary profile of the PATrev and the PCCL. The findings suggest that barriers to uptake of psychosocial screening tools exist. Further inquiry into the efficacy and utility of psychosocial screening tools in pediatric oncology is warranted before integrating these tools into standard pediatric clinical care.
Limitations
The results of this pilot study need to be considered in light of several limitations. The current pilot study was underpowered, particularly at T2, to fully assess the differences in utility ratings among HCPs and across time. The sample for the utility assessment of the PATrev summary was particularly small because only those HCPs who received the PATrev psychosocial summary were able to assess the utility of it. As a result, the effects of time and the HCP’s discipline could not be well examined at T2. HCPs were asked to assess only the psychosocial summary profile derived from the PATrev, not the entire instrument. Assessment of the utility of the PATrev summary may not constitute utility assessment of the actual tool. A larger multisite study could ensure larger sample size to assess utility ratings provided by HCPs. The utility ratings themselves were generally modest (they were highest in nurses [7/10]); a ceiling effect may exist regarding the utility of psychosocial screening tools. In addition, the VAS assessments may have been better addressed using a Likert-type scale. Because HCPs received a request to rate the psychosocial screening tool more than once, based on the number of consenting families who were within their care, familiarity with the tool and rater fatigue may have affected utility ratings. Despite these limitations, the findings of the pilot study are unique; they emphasize differences in perceived utility of psychosocial screening tools across HCPs, which may affect the tools’ implementation in pediatric cancer centers.
Implications for Research
Future research should investigate how psychosocial screening tools can be incorporated in a clinically meaningful way into pediatric oncology comprehensive clinical care to document psychosocial needs. For example, the timing of the introduction of these tools is critical. Utility ratings in the current study may reflect the utility of the information at the time in which it was received; the HCPs may have received the information too soon or too late after diagnosis for it to be of use. Consequently, future studies may wish to assess whether the time of provision of psychosocial risk information to the practicing team affects HCPs’ utility ratings, as well as whether HCPs found the information provided by the psychosocial risk tools to be redundant and, if so, at what time points and if timing and redundancy vary across HCP disciplines.
Research on this subject may be enhanced with the inclusion of qualitative interviews to investigate HCPs’ opinions and beliefs in psychosocial screening tools and to identify potential barriers to implementation. Investigation into what may benefit social workers and the rest of the clinical team, as far as psychosocial screening tools are concerned, will be an important source of information regarding how to best target the psychosocial information needs of HCPs in a team-based care system. Ultimately, the aim is to improve psychosocial outcomes of children with cancer and their families. 
Conclusion
Oncology nurses play a critical role in bridging the medical and psychosocial needs of the child who is being treated for cancer and his or her family, given their frequent contact during nursing care. Their use of psychosocial screening tools could enhance their knowledge of the child and family, which, in turn, would allow them to communicate this knowledge to the rest of the oncology team and to mobilize additional supportive services for the family before psychosocial problems become psychosocial crises. Many pediatric cancer centers have limited psychosocial resources (e.g., social workers, psychologists). The use of psychosocial screening tools by nurses would facilitate raising awareness of the importance of addressing the child’s psychosocial needs within the center or the community.
This pilot study has identified a previously unknown barrier to uptake of psychosocial screening tools in pediatric oncology services: variable belief in the utility and endorsement of psychosocial screening tools across practitioners in pediatric oncology. Nurses found the tools most useful, whereas social workers found them least useful. Future research should examine specific barriers to uptake and implementation of these tools to provide meaningful and integrated clinical care. For example, the timing of the completion of the tools may determine how useful they are found in clinical practice. Investigation of discipline-specific barriers to the acceptance and use of psychosocial screening tools will continue to be important in the quest to enhance the knowledge translation into clinically meaningful change for clinicians and families of children with cancer.
References
Alderfer, M.A., & Hodges, J.A. (2010). Supporting siblings of children with cancer: A need for family–school partnerships. School Mental Health, 2, 72–81. doi:10.1007/s12310-010-9027-4
Barrera, M., Atenafu, E., Doyle, J., Berlin-Romalis, D., & Hancock, K. (2012). Differences in mothers’ and fathers’ psychological distress after pediatric SCT: A longitudinal study. Bone Marrow Transplantation, 47, 934–939. doi:10.1038/bmt.2011.206
Barrera, M., Hancock, K., Johnston, D., Cataudella, D., Punnett, A., Bartels, U., . . . Greenberg, C. (2011, November). Reliability and validity of the Psychosocial Checklist (PCCL): An instrument to be used by childhood cancer health care providers. Poster presented at the Pediatric Oncology Group of Ontario Symposium, Toronto, ON, Canada.
Barrera, M., Hancock, K., Rokeach, A., Atenafu, E., Cataudella, D., Punnett, A., . . . Greenberg, C. (2014). Does the use of the revised psychosocial assessment tool (PATrev) result in improved quality of life and reduced psychosocial risk in Canadian families with a child newly diagnosed with cancer? Psycho-Oncology, 23, 165–172. doi:10.1002/pon.3386
Barrera, M., Hancock, K., Rokeach, A., Cataudella, D., Atenafu, E., Johnston, D., . . . Greenberg, C. (2014). External validity and reliability of the Psychosocial Assessment Tool (PAT) among Canadian parents of children newly diagnosed with cancer. Pediatric Blood and Cancer, 61, 165–170. doi:10.1002/pbc.24774
Barrera, M., Yogalingam, P., Rokeach, A., Hancock, K., Cataudella, D., Johnston, D., . . . Shama, W. (2014, February). Health care professionals treating a child with cancer: Their knowledge of family psychosocial problems. Poster presented at the meeting of the American Psychosocial Oncology Society, Tampa, FL.
Bearden, D.J., Feinstein, A., & Cohen, L.L. (2012). The influence of parent preprocedural anxiety on child procedural pain: Mediation by child procedural anxiety. Journal of Pediatric Psychology, 37, 680–686. doi:10.1093/jpepsy/jss041
Boman, K., Lindahl, A., & Björk, O. (2003). Disease-related distress in parents of children with cancer at various stages after the time of diagnosis. Acta Oncologica, 42, 137–146.
Bultz, B.D., Groff, S.L., Fitch, M., Blais, M.C., Howes, J., Levy, K., & Mayer, C. (2011). Implementing screening for distress, the 6th vital sign: A Canadian strategy for changing practice. Psycho-Oncology, 20, 463–469. doi:10.1002/pon.1932
Bultz, B.D., Groff, S.L., & Fitch, M.I. (2009). Guide to implementing screening for distress, the 6th vital sign, part A: Background, recommendations, and implementation. Toronto, ON, Canada: Canadian Partnership Against Cancer.
Carlson, L.E., Waller, A., Groff, S.L., Zhong, L., & Bultz, B.D. (2012). Online screening for distress, the 6th vital sign, in newly diagnosed oncology outpatients: Randomised controlled trial of computerised vs personalised triage. British Journal of Cancer, 107, 617–625. doi:10.1038/bjc.2012.309
Carlson, L.E., Waller, A., & Mitchell, A.J. (2012). Screening for distress and unmet needs in patients with cancer: Review and recommendations. Journal of Clinical Oncology, 30, 1160–1177. doi:10.1200/JCO.2011.39.5509
Dolgin, M.J., Phipps, S., Fairclough, D.L., Sahler, O.J., Askins, M., Noll, R.B., . . . Katz, E.R. (2007). Trajectories of adjustment in mothers of children with newly diagnosed cancer: A natural history investigation. Journal of Pediatric Psychology, 32, 771–782.
Field, A. (2009). Discovering statistics using SPSS (3rd ed.). London, UK: Sage.
Haverman, L., van Rossum, M.A., van Veenendaal, M., van den Berg, J.M., Dolman, K.M., Swart, J., . . . Grootenhuis, M.A. (2013). Effectiveness of a web-based application to monitor health-related quality of life. Pediatrics, 131, 533–543. doi:10.1542/peds.2012-0958
Holland, J.C., & Bultz, B.D. (2007). The NCCN guideline for distress management: A case for making distress the sixth vital sign. Journal of the National Comprehensive Cancer Network, 5, 3–7.
Kazak, A.E., Alderfer, M., Rourke, M.T., Simms, S., Streisand, R., & Grossman, J.R. (2004). Posttraumatic stress disorder (PTSD) and posttraumatic stress symptoms (PTSS) in families of adolescent childhood cancer survivors. Journal of Pediatric Psychology, 29, 211–219. doi:10.1093/jpepsy/jsh022
Kazak, A.E., Barakat, L.P., Ditaranto, S., Biros, D., Hwang, W.T., Beele, D., . . . Reilly, A. (2011). Screening for psychosocial risk at pediatric cancer diagnosis: The Psychosocial Assessment Tool. Journal of Pediatric Hematology/Oncology, 33, 289–294. doi:10.1097/MPH.0b013e31820c3b52
Kazak, A.E., Boeving, C.A., Alderfer, M.A., Hwang, W.T., & Reilly, A. (2005). Posttraumatic stress symptoms during treatment in parents of children with cancer. Journal of Clinical Oncology, 23, 7405–7410. doi:10.1200/jco.2005.09.110
Kazak, A.E., Brier, M., Alderfer, M.A., Reilly, A., Fooks Parker, M., Rogerwick, S., . . . Barakat, L.P. (2012). Screening for psychosocial risk in pediatric cancer. Pediatric Blood and Cancer, 59, 822–827. doi:10.1002/pbc.24166
Kazak, A.E., Kassam-Adams, N., Schneider, S., Zelikovsky, N., Alderfer, M.A., & Rourke, M. (2006). An integrative model of pediatric medical traumatic stress. Journal of Pediatric Psychology, 31, 343–355. doi:10.1093/jpepsy/jsj054
Kazak, A.E., Prusak, A., McSherry, M., Simms, S., Beele, D., Rourke, M., . . . Lange, B. (2001). The Psychosocial Assessment Tool (PAT): Development of a brief screening instrument for identifying high risk families in pediatric oncology. Families, Systems, and Health, 19, 303–317.
Kazak, A.E., Rourke, M.T., Alderfer, M.A., Pai, A., Reilly, A.F., & Meadows, A.T. (2007). Evidence-based assessment, intervention and psychosocial care in pediatric oncology: A blueprint for comprehensive services across treatment. Journal of Pediatric Psychology, 32, 1099–1110. doi:10.1093/jpepsy/jsm031
Kazak, A.E., Simms, S., & Rourke, M.T. (2002). Family systems practice in pediatric psychology. Journal of Pediatric Psychology, 27, 133–143. doi:10.1093/jpepsy/27.2.133
Mitchell, W., Clarke, S., & Sloper, P. (2005). Survey of psychosocial support provided by UK paediatric oncology centres. Archives of Disease in Childhood, 90, 796–800. doi:10.1136/adc.2004.065177
National Comprehensive Cancer Network. (2003). Distress management. Clinical practice guidelines. Journal of the National Comprehensive Cancer Network, 1, 344–374.
Pai, A.L., Greenley, R.N., Lewandowski, A., Drotar, D., Youngstrom, E., & Peterson, C.C. (2007). A meta-analytic review of the influence of pediatric cancer on parent and family functioning. Journal of Family Psychology, 21, 407–415. doi:10.1037/0893-3200.21.3.407
Pai, A.L.H., Patiño-Fernández, A.M., McSherry, M., Beele, D., Alderfer, M.A., Reilly, A.T., . . . Kazak, A.E. (2008). The Psychosocial Assessment Tool (PAT2.0): Psychometric properties of a screener for psychosocial distress in families of children newly diagnosed with cancer. Journal of Pediatric Psychology, 33, 50–62. doi:10.1093/jpepsy/jsm053
Patel, S.K., Mullins, W., Turk, A., Dekel, N., Kinjo, C., & Sato, J.K. (2011). Distress screening, rater agreement, and services in pediatric oncology. Psycho-Oncology, 20, 1324–1333. doi:10.1002/pon.1859
Patenaude, A.F., & Kupst, M.J. (2005). Psychosocial functioning in pediatric cancer. Journal of Pediatric Psychology, 30, 9–27. doi:10.1093/jpepsy/jsi012
Rodriguez, E.M., Dunn, M.J., Zuckerman, T., Vannatta, K., Gerhardt, C.A., & Compas, B.E. (2012). Cancer-related sources of stress for children with cancer and their parents. Journal of Pediatric Psychology, 37, 185–197. doi:10.1093/jpepsy/jsr054
Selove, R., Kroll, T., Coppes, M., & Cheng, Y. (2012). Psychosocial services in the first 30 days after diagnosis: Results of a web-based survey of Children’s Oncology Group (COG) member institutions. Pediatric Blood and Cancer, 58, 435–440. doi:10.1002/pbc.23235
Wiener, L., & Pao, M. (2012). Comprehensive and family-centered psychosocial care in pediatric oncology: Integration of clinical practice and research. In S. Kreitler, M.W. Ben-Arush, & A. Martin (Eds.), Pediatric psycho-oncology: Psychosocial aspects and clinical interventions (2nd ed., pp. 7–16). West Sussex, UK: John Wiley and Sons.
About the Author(s)
Ashley Di Battista, PhD, is a clinical research fellow in the Department of Critical Care Medicine and in the Neurosciences and Mental Health Program and Kelly Hancock, MA, is a clinical research coordinator in the Department of Psychology, both at the Hospital for Sick Children in Toronto; Danielle Cataudella, PhD, is a psychologist in the London Health Sciences Centre at Children’s Hospital in London; Donna Johnston, MD, is a pediatric hematologist/oncologist and Marilyn Cassidy, RN, is a pediatric interlink community nurse, both at Children’s Hospital of Eastern Ontario in Ottawa; and Angela Punnett, MD, is a staff physician, Wendy Shama, MSW, is a social worker, and Maru Barrera, PhD, is a pediatric health psychologist, all at the Hospital for Sick Children, all in Ontario, Canada. This study was supported, in part, by the Pediatric Oncology Group of Ontario Research Unit. Barrera can be reached at maru.barrera@sickkids.ca, with copy to editor at ONFEditor@ons.org. (Submitted September 2014. Accepted for publication February 9, 2015.)

