A Phase I Study Examining the Feasibility and Safety of an Aerobic Exercise Intervention in Patients With Rectal Cancer During and After Neoadjuvant Chemoradiotherapy
Purpose/Objectives: To assess the feasibility and safety of an aerobic exercise intervention in patients with rectal cancer during and after neoadjuvant chemoradiotherapy (NACRT).
Design: A prospective, single-group design with assessments at pre-NACRT, post-NACRT, and presurgery.
Setting: The Cross Cancer Institute and University of Alberta in Edmonton, Canada.
Sample: 18 patients with rectal cancer scheduled to receive long-course NACRT followed by definitive surgery.
Methods: Participants received a supervised moderate-intensity aerobic exercise program three days per week during six weeks of NACRT followed by an unsupervised aerobic exercise program for 150 minutes or more per week for 6–8 weeks prior to surgery.
Main Research Variables: Eligibility rate, recruitment rate, follow-up rate, exercise adherence, serious adverse events, health-related fitness outcomes, and patient-reported outcomes.
Findings: Follow-up rates post-NACRT were 83% for health-related fitness outcomes and 94% for patient-reported outcomes. Patients attended a median of 83% of their supervised exercise sessions and completed a mean of 222 minutes per week (SD = 155) of their unsupervised exercise. No serious adverse events were observed or reported. Most health-related fitness outcomes and patient-reported outcomes declined during NACRT and recovered after NACRT.
Conclusions: Aerobic exercise is feasible and safe for patients with rectal cancer during and after NACRT.
Implications for Nursing: Patients with rectal cancer are able to engage in moderate-intensity aerobic exercise during NACRT.
Jump to a section
Standard treatment for locally advanced rectal cancer usually includes long-course neoadjuvant chemoradiotherapy (NACRT), consisting of 5–6 weeks of radiotherapy (45–54 Gy) with concurrent chemotherapy (capecitabine [Xeloda®] or 5-fluorouracil [Adrucil®]) (Cravo et al., 2014). About 6–8 weeks after NACRT, patients are usually offered a total mesorectal excision (usually with an abdominal perineal resection or low anterior resection) followed by adjuvant chemotherapy (Cravo et al., 2014). NACRT leads to improved local control compared to surgery alone, preoperative radiation, or postoperative chemoradiation; however, the improvements in overall survival are less clear (Cravo et al., 2014; Dewdney, Cunningham, & Chau, 2013; Sauer et al., 2012). Unfortunately, NACRT is associated with acute side effects, including fatigue, diarrhea, radiation dermatitis, hematologic toxicity, hand-foot syndrome, and cardiotoxicity (Gavaruzzi, Giandomenico, & Pucciarelli, 2013; Swellengrebel et al., 2011). Interventions to manage these side effects and improve treatment outcomes are highly desirable.
Substantial evidence has shown that exercise during cancer treatment is safe and can help mitigate some of the side effects of treatments, such as physical deconditioning, fatigue, pain, depression, and poor quality of life (Mishra et al., 2012). Most of this research, however, has focused on patients with breast, prostate, and hematologic cancers (Mishra et al., 2012), with few studies examining patients with colorectal cancer and no studies examining patients with rectal cancer during NACRT (Cramer, Lauche, Klose, Dobos, & Langhorst, 2014). In addition, limited research has suggested that exercise during chemotherapy may improve chemotherapy completion rates (Courneya et al., 2007; Van Waart et al., 2015), treatment response (Courneya et al., 2009), and even long-term disease-free survival (Courneya et al., 2014); however, this evidence is limited to patients with breast cancer and lymphoma. Finally, presurgical exercise interventions in patients with cancer may improve cardiorespiratory fitness, quality of life, and postsurgical complications; however, none of these studies have included patients receiving neoadjuvant treatment (Singh, Newton, Galvao, Spry, & Baker, 2013).
West et al. (2014) demonstrated that NACRT in patients with rectal cancer has a negative impact on cardiovascular fitness, which may predict postsurgical complications. In addition, in the first exercise intervention study in this clinical setting, West et al. (2015) showed that an exercise program initiated immediately after NACRT was feasible and improved cardiovascular fitness. The promising findings raise the question of whether initiating an exercise program during NACRT may have even greater benefits for patients with rectal cancer, such as preventing declines in cardiovascular fitness, managing side effects, and improving treatment outcomes. Given the substantial toxicity and side effects of NACRT, however, whether or not patients with rectal cancer would be willing and able to complete an exercise program during NACRT is unclear.
The primary purpose of this phase I study was to test the feasibility and safety of an aerobic exercise intervention in patients with rectal cancer during and immediately after long-course NACRT. A secondary purpose was to track changes in objective health-related fitness outcomes and patient-reported outcomes. The authors hypothesized that patients with rectal cancer initiating NACRT would be willing and able to complete an aerobic exercise program. To demonstrate feasibility, the authors’ prespecified criteria included a recruitment rate of 20% or greater, an adherence rate to the supervised exercise program during NACRT of 70% or greater, and a follow-up assessment rate for the health-related fitness outcomes and patient-reported outcomes at post-NACRT of 80% or greater.
Methods
The study was conducted at the Cross Cancer Institute and the University of Alberta in Edmonton, Canada. The study was approved by the Health Research Ethics Board of Alberta–Cancer Committee. Written informed consent was obtained from participants. Eligibility criteria were patients with rectal cancer (a) scheduled to receive long-course NACRT followed by definitive surgery, (b) aged 18–80 years, (c) with no uncontrolled medical or psychiatric conditions, (d) cleared to participate in exercise as determined by the Physical Activity Readiness Questionnaire for Everyone (PAR-Q+), and (e) able to understand English.
The study was a prospective, single-group design with assessments at pre-NACRT, post-NACRT, and presurgery. From April to October 2014, potentially eligible patients were screened by their radiation oncologist and the study coordinator at the time of their first radiation consultation. The study coordinator followed-up with eligible patients by phone and scheduled interested patients for pre-NACRT testing.
Exercise Intervention
The exercise intervention was divided into two phases: during NACRT and post-NACRT. During NACRT, all exercise sessions were supervised by an exercise specialist at the Behavioral Medicine Fitness Center, which is within walking distance of the cancer center. During NACRT, the primary goal of the exercise intervention was to complete 18 supervised aerobic exercise sessions (three sessions per week for six weeks). A secondary goal was to determine if patients could reach 150 minutes of moderate-intensity aerobic exercise in three training sessions per week (50 minutes per session). The exercise prescription was individually tailored to each patient based on the results of the pre-NACRT treadmill test (40%–60% of estimated volume of oxygen consumption [VO2] reserve). Each training session consisted of a five minute aerobic warm up and cool down at low intensity. The intensity of the exercise sessions was monitored via heart rate monitors and the Borg rating of perceived exertion scale. Exercise modalities included treadmill, upright bike, recumbent bike, elliptical, and rower. For each supervised session, the exercise trainer recorded attendance, duration, intensity, and any symptoms or serious adverse events experienced during or immediately after the exercise.
After NACRT, patients were provided the option of continuing with the supervised exercise program, completing an unsupervised exercise program, or a combination. During this phase, the goal of the exercise program was to maintain (or achieve) 150 minutes of moderate-intensity aerobic exercise per week. The exercise prescription was individually tailored to each patient based on the results of the post-NACRT treadmill test (40%–60% of estimated VO2 reserve). Patients completing unsupervised exercise were provided with a heart rate monitor and were asked to record exercise frequency, intensity, duration, and modality. The study coordinator completed weekly telephone calls or email follow-ups to support patients in meeting the exercise prescription.
Demographic and behavioral variables were assessed by self-report, and medical data was extracted from medical records. Baseline exercise was assessed by the Godin Leisure Time Exercise Questionnaire (GLTEQ) (Godin & Shephard, 1985).
The feasibility outcomes included eligibility rate, recruitment rate, follow-up rate, and exercise adherence rate. Exercise adherence during NACRT was assessed as the number of sessions attended out of 18. Exercise adherence after NACRT was self-reported using the GLTEQ (Godin & Shephard, 1985). Safety was assessed by monitoring and recording any serious adverse events that occurred during exercise testing or the supervised exercise sessions.
The authors’ goal was to complete the pre-NACRT assessment on the same day as the radiation planning session. The post-NACRT assessment was planned for within one week of completing NACRT or the same day as the last radiation session for out-of-town patients. The presurgery assessment was planned for 1–2 weeks prior to surgery. For out-of-town patients, the goal was to do the presurgery test when they were in town for other presurgical appointments.
Health-Related Fitness Outcomes
Cardiorespiratory fitness was assessed using a multistage submaximal aerobic exercise test with direct measures of gas exchange and ventilation on a calibrated metabolic measurement system (Parvo Medics TrueOne® 2400). The test was terminated at the end of the stage during which the patient reached 85% of his or her age-predicted maximal heart rate. Functional aerobic capacity (VO2 max) was estimated using the heart rate and VO2 from two submaximal stages of the treadmill test during which patients achieved steady-state heart rate between 115 and 150 beats per minute. The slope was then determined by calculating the ratio of the difference between the two submaximal VO2 measures and the corresponding heart rates (Heyward, 2006; Thompson, Gordon, & Pescatello, 2010). The Senior’s Fitness Test (SFT) was used to assess physical functioning (Jones & Rikli, 2002; Rikli & Jones, 1999). Body composition was assessed by height, weight, waist, and hip circumference (National Institutes of Health, 2000; World Health Organization, 2008).
Patient-Reported Outcomes
Health-related quality of life was measured using the validated SF-36®, which has a reliability of 0.85 (Ware et al., 2007). The SF-36 contains 36 items that yield eight health domains: physical functioning, role-physical, bodily pain, general health, vitality, social functioning, role-emotional, and mental health. Scores for each subscale were transformed into norm-based scores with higher scores indicating higher functioning. The physical health component summary (physical functioning, role-physical, bodily pain, and general health) and mental health component summary (vitality, social functioning, role-emotional, and mental health) also were calculated.
Cancer-specific quality of life was measured by the Functional Assessment of Cancer Therapy–Colorectal (FACT-C) scales, which have reliablities ranging from 0.85–0.91 (Ward et al., 1999). The FACT-C includes the 27 items from the FACT–General (FACT-G) scale and the 11-item colorectal subscale. The FACT-G is divided into four quality-of-life domains: physical well-being (PWB), social/family well-being (SWB), emotional well-being (EWB), and functional well-being (FWB). In addition, the 11-item FACT-D subscale was used to assess diarrhea and the 13-item FACT-F subscale was used to assess fatigue (Cella & Nowinski, 2002; Yellen, Cella, Webster, Blendowski, & Kaplan, 1997). The PWB, FWB, and colorectal subscales were combined to form the Trial Outcome Index–Colorectal. On all scales, a higher score indicates better quality of life.
Depression was measured using the 10-item Center for Epidemiologic Studies Depression Scale (CES-D), which has demonstrated validity and a reliability of 0.71 (Kohout, Berkman, Evans, & Cornoni-Huntley, 1993). The CES-D measures the frequency of depressive symptoms during the past week on a scale of 0–3, with total scores ranging from 0–30. A higher score indicates more depressive symptoms. Anxiety was measured using the validated 10-item Spielberger State Anxiety Scale (SSAS) (Spielberger, Gorsuch, Lushene, Vagg, & Jacobs, 1983), which measures symptoms of anxiety during the past week on a scale of 1–4. The total score for the SSAS ranges from 10–40, and a higher score indicates more anxiety. Stress was measured using the 14-item Perceived Stress Scale (PSS), which has shown reliabilities ranging from 0.84 to 0.86 (Cohen, Kamarck, & Mermelstein, 1983). The PSS measures symptoms of stress during the past month on a scale of 0–4, with the total score ranging from 0–56. A higher score indicates more perceived stress. Self-esteem was measured using the validated 10-item Rosenberg Self-Esteem Scale (RSES) (Rosenberg, 1965). The RSES measures current self-esteem on a scale of 1–4, with total scores ranging from 10–40. Higher scores indicate higher self-esteem. Finally, sleep quality was assessed using the 19-item Pittsburgh Sleep Quality Index (PSQI), which has been validated in patients with cancer and has a reliability of 0.81 (Beck, Schwartz, Towsley, Dudley, & Barsevick, 2004). Seven sleep components are assessed during the past month (subjective sleep quality, sleep latency, sleep duration, habitual sleep efficiency, sleep disturbances, use of sleeping medications, and daytime dysfunction) and are rated on a scale of 0–3 with a lower score indicating better sleep quality (Buysse, Reynolds, Monk, Berman, & Kupfer, 1989). The seven components can be summed to form a global sleep quality score ranging from 0–21, and a score of 5 or greater is indicative of poor sleep quality (Buysse et al., 1989).
Statistical Analysis
For this phase I feasibility study, the authors’ goal was to recruit 10–20 patients. The primary statistical analysis was to calculate the frequencies and percentages for various feasibility outcomes. For the health-related fitness outcomes and patient-reported outcomes, the authors report only descriptive statistics of the mean and standard deviation at each time point and the mean change and 95% confidence interval (CI) between each pair of time points using all available data (pairwise) and for complete cases only (listwise). Data were analysed using SPSS®, version 22.0.
Results
Flow of patients through the trial is summarized in Figure 1. From April 17 to October 23, 2014, the authors assessed 45 patients with rectal cancer for eligibility; of these, 32 (71%) were eligible, and 18 (56%) were recruited. All patients completed health-related fitness testing and patient-reported outcomes pre-NACRT. For logistical reasons, eight of the recruited patients completed their pre-NACRT fitness test immediately after their first radiation treatment.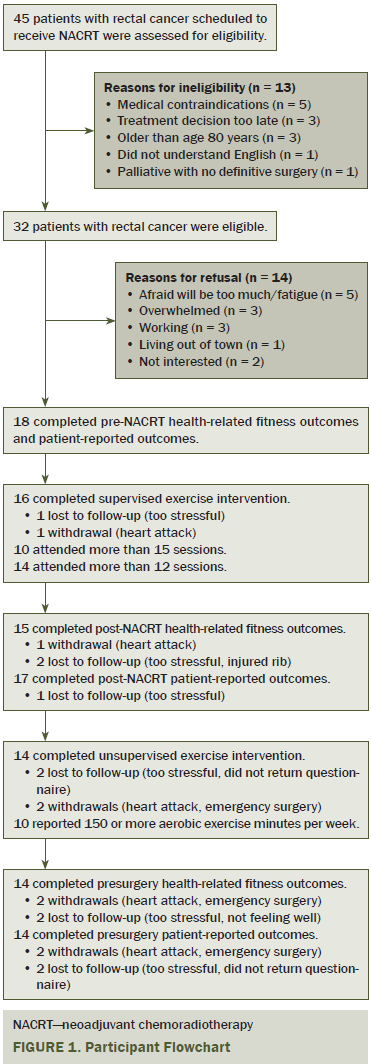
The baseline characteristics of the patients are reported in Table 1. NACRT included 50.4–54 Gy in 28–30 fractions on weekdays. Two patients had a delay of five days or greater in their radiation treatment because of enteritis. The mean cumulative dose of chemotherapy was 88% (range = 57%–100%) of the planned dose. Four patients needed a dose reduction for a mean of seven days (range = 5–8 days) because of hand-foot syndrome (n = 3) or enteritis (n = 1). Six patients, two of whom had a previous dose reduction, stopped chemotherapy for a mean of 9.5 days (range = 5–13 days) because of hand-foot syndrome (n = 3), diarrhea (n = 1), enteritis (n = 1), or cardiotoxicity (n = 1). The median duration of NACRT was 6.5 weeks (range = 6–8 weeks), and the median time between the completion of NACRT and definitive surgery was 8 weeks (range = 5–11 weeks).
Follow-Up Completion Rate
At post-NACRT, 15 patients (83%) completed health-related fitness outcomes and 17 (94%) completed the patient-reported outcomes. At presurgery, 14 patients (78%) completed health-related fitness testing and patient-reported outcomes. In total, 13 patients (72%) completed the health-related fitness outcomes and 14 patients (78%) completed the patient-reported outcomes at all three time points.
Exercise Program Adherence
Including all 18 patients who initiated the study, the mean attendance was 13.3 (74%) and the median attendance was 15 (83%) of the 18 supervised exercise sessions during NACRT. One patient withdrew from the study because it was too difficult to keep up adherence to the medical appointments and exercise sessions. The most common reasons for missed supervised exercise sessions were hand-foot syndrome, not feeling well, poor sleep, diarrhea, nausea, fatigue, and logistical reasons. The mean duration of the supervised exercise sessions was 40 minutes (SD = 9), and the intensity goal (moderate) was met 100% of the time. The most frequently used modality was the treadmill (67% of sessions).
After NACRT, 14 of 16 patients still on trial chose to complete their exercise in an unsupervised format. Self-report exercise was collected from 14 of 18 patients at the presurgery time point. The mean total aerobic exercise minutes was 222 minutes (SD = 155), with 10 of 14 patients still on trial meeting the aerobic exercise goal of 150 minutes per week or greater.
No serious adverse events were observed or reported during or immediately after the 47 health-related fitness tests or the 239 supervised exercise sessions. The two serious adverse events that occurred while patients were on trial (heart attack and emergency surgery) were deemed by the attending oncologists to be unrelated to exercise.
Health-Related Fitness Outcomes
Descriptive statistics for changes in health-related fitness are reported in Table 2. Estimated VO2 max declined from pre- to post-NACRT (mean change = –1.3 ml/kg per minute; 95% CI [–3.6, 1.7]) and increased from post-NACRT to presurgery (mean change = 2.4 ml/kg per minute; 95% CI [–0.9, 5.7]), resulting in a slight improvement from pre-NACRT to presurgery (mean change = 1.1 ml/kg per minute; 95% CI [–1.7, 3.9]). The six-minute walk distance improved across all three time points, achieving a mean change of 47 meters (95% CI [14, 80]) from pre-NACRT to presurgery.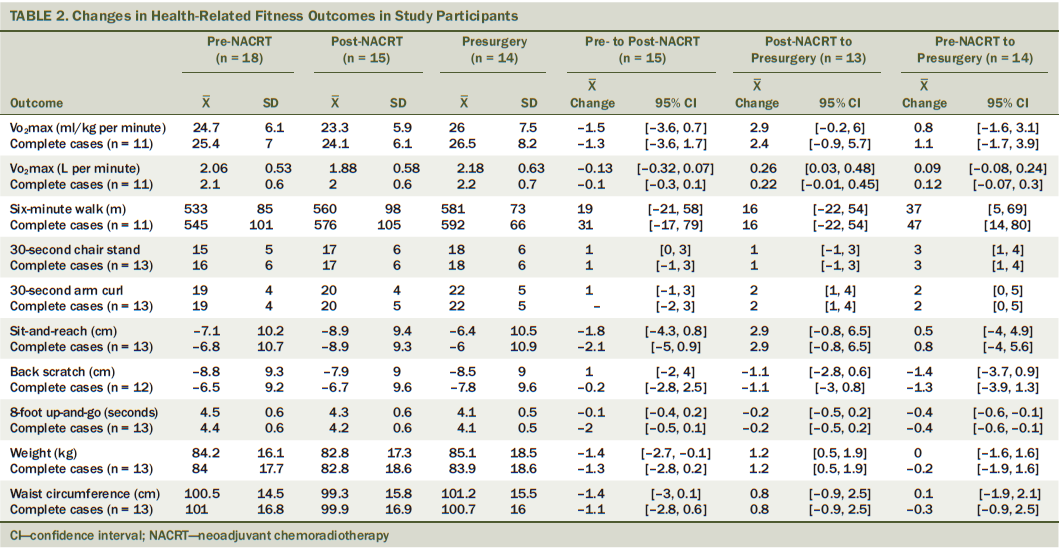
Patient-Reported Outcomes
Descriptive statistics for changes in health-related quality of life are presented in Table 3. The observed pattern showed a decline from pre- to post-NACRT and an improvement from post-NACRT to presurgery for almost all outcomes. Changes in cancer-specific quality of life are reported in Table 4. The pattern showed a decline for almost all outcomes from pre- to post-NACRT, and an improvement from post-NACRT to presurgery. Changes in psychosocial functioning are reported in Table 5. Perceived stress and sleep quality worsened from pre- to post-NACRT, and returned to near pre-NACRT levels from post-NACRT to presurgery.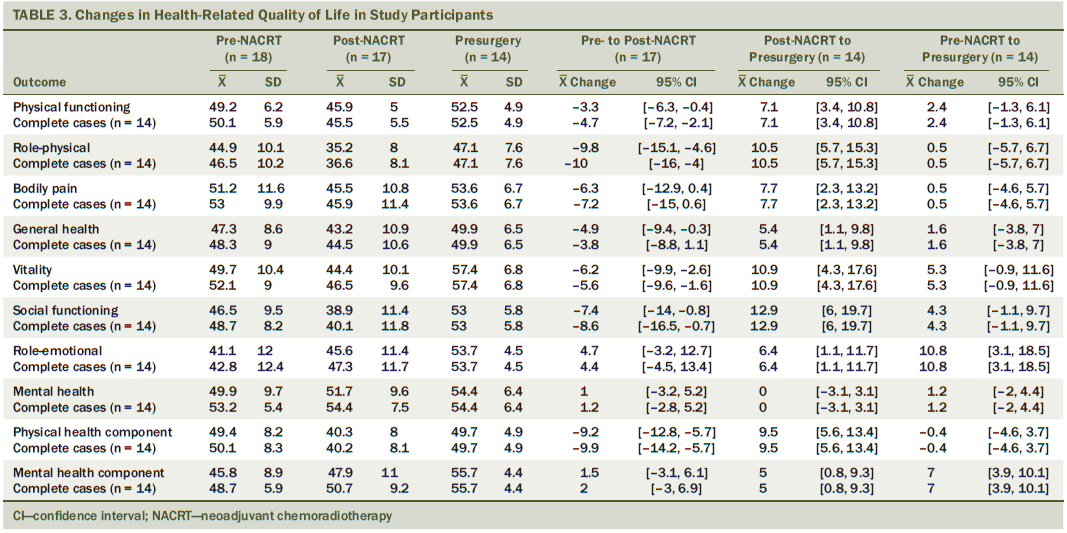
Discussion
The current study is the first to examine the feasibility and safety of aerobic exercise in patients with rectal cancer receiving NACRT. The 71% eligibility rate in the study suggests that the majority of patients with rectal cancer receiving NACRT are eligible for an aerobic exercise intervention. Although no studies exist to directly compare results with, the eligibility rate is similar to the 67% reported for a supervised exercise intervention in colorectal cancer survivors receiving adjuvant chemotherapy (Lin, Shun, Lai, Liang, & Tsauo, 2014). In addition, the current study’s rate is higher than the eligibility rate for supervised exercise trials in patients with breast cancer or lymphoma receiving chemotherapy (33%–41%) (Courneya et al., 2007, 2009, 2013) and patients with prostate cancer receiving radiotherapy (37%) (Segal et al., 2009). In the only other exercise intervention study targeting presurgical patients with rectal cancer, West et al. (2015) did not report the eligibility rate. The main reasons for ineligibility in the current study were medical conditions that are significant contraindications to exercise. The eligibility criteria are unlikely to be addressed in any future trials. However, the 71% eligibility rate suggests a substantial subpopulation of patients with rectal cancer initiating NACRT who are eligible for an exercise intervention.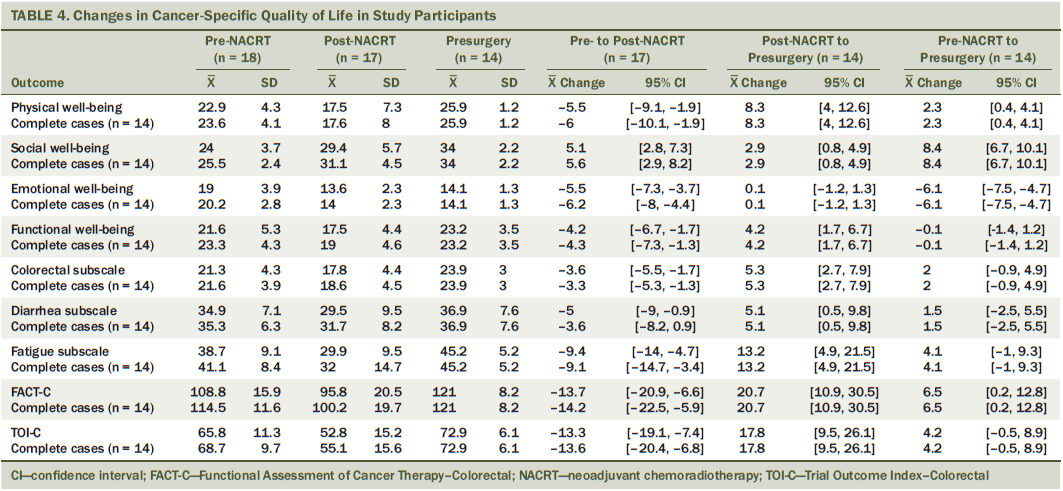
The recruitment rate of 56% is higher than the 26%–49% rates reported for other studies recruiting patients with cancer to exercise interventions during adjuvant treatment (Courneya et al., 2007, 2009, 2013; Lin et al., 2014; Segal et al., 2009). Again, the West et al. (2015) study did not report the recruitment rate or reasons for refusal. In the current study, patients mainly refused because they were afraid it would be “too much” during NACRT. This reason may be addressed in future studies by informing patients of the success of patients in the current study. The greater than 50% recruitment rate suggests strong interest of patients with rectal cancer in an exercise intervention during NACRT.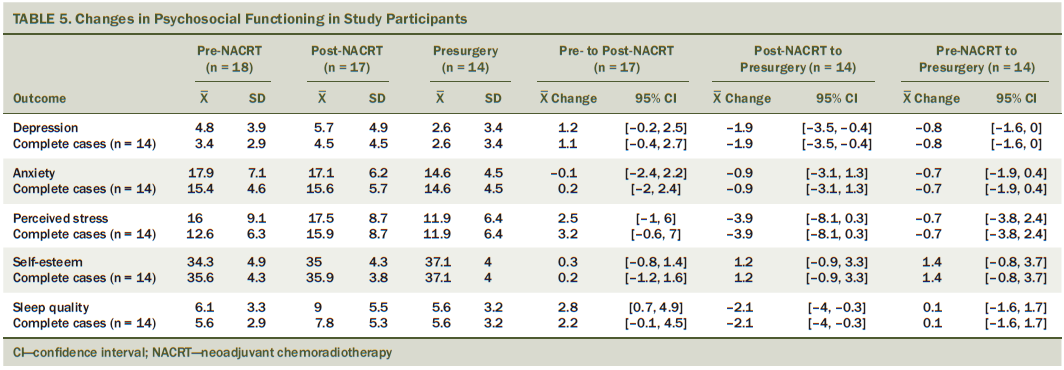
Follow-up assessment rates for the health-related fitness testing and questionnaires were excellent at the post-NACRT time point (83% and 94%, respectively) and even the presurgical time point (78% for all assessments). Other exercise trials in patients with cancer receiving adjuvant treatments have reported slightly higher rates (Courneya et al., 2007, 2009, 2013). In addition, West et al. (2015) reported a perfect follow-up rate for fitness assessments in the exercise group; however, only 59% of the control group provided follow-up fitness assessments, perhaps because the control group consisted of patients who declined exercise and/or were from out of town. In the current study, getting the out-of-town patients back for the presurgery fitness testing was feasible. The main reason for missed fitness testing was acute medical contraindications, which were deemed unrelated to exercise.
The intention-to-treat mean adherence rate of 74% and median adherence rate of 83% for the supervised exercise sessions is excellent and similar to that reported in other trials during chemotherapy (Courneya et al., 2007, 2009, 2013). The two most frequently reported barriers to exercise during NACRT were hand-foot syndrome and logistical issues. Hand-foot syndrome has not been previously reported as a barrier to exercise, but it should be addressed in future trials with this patient population because exercise has the potential to exacerbate this symptom. Logistical issues included the necessity of scheduling out-of-town patients for their health-related fitness test on the same day as their first/last radiation session, which reduced the number of supervised exercise sessions available.
In the post-NACRT phase, only 2 of 16 patients were interested in continuing with supervised exercise. This finding contrasts with West et al. (2015), who reported a 96% adherence rate to a hospital-based supervised exercise program for patients with rectal cancer after NACRT. One difference between the studies is that West et al. (2015) only included local patients in the exercise intervention, whereas out-of-town patients were assigned to the control group. However, in the current study, even local patients did not express interest in continuing with the supervised exercise after NACRT. Fortunately, adherence to the unsupervised exercise in the post-NACRT phase was excellent, with a mean of greater than 200 minutes of self-reported aerobic exercise and 10 of 14 patients meeting the goal of 150 minutes or more per week. The data suggest that an unsupervised exercise program after NACRT and prior to surgery may be feasible.
Most importantly, the current study suggests that aerobic exercise during and after NACRT is safe. No adverse events were observed or reported during or after exercise testing or training. Two serious adverse events occurred while patients were on trial; however, these events were deemed to be treatment related by the oncologists. In any case, continued monitoring of safety outcomes is necessary in this clinical setting to further evaluate the safety of exercise.
The current study suggested a small decline in cardiovascular fitness from pre- to post-NACRT of 1.3 ml/kg per minute despite the aerobic exercise intervention. Without an exercise intervention, West et al. (2015) observed a decline in cardiovascular fitness during NACRT of 2.5 ml/kg per minute, suggesting that the aerobic exercise intervention may have prevented some of the decline in fitness during NACRT. In addition, the current study’s findings suggested an improvement in cardiovascular fitness from post-NACRT to presurgery of 2.4 ml/kg per minute. In the study by West et al. (2015), the post-NACRT supervised exercise intervention resulted in an improvement of 2.7 ml/kg per minute (compared to a decline of 1.3 ml/kg per minute in a nonrandomized control group), suggesting that the unsupervised exercise intervention in the current study may be as effective as supervised exercise for improving cardiorespiratory fitness in the post-NACRT setting. In addition, in another observational study, West et al. (2014) observed a decline in cardiovascular fitness from pre-NACRT to presurgery of 1.4 ml/kg per minute compared to the suggested improvement in the current study of 1.1 ml/kg per minute. Of course, as a phase I study without a proper comparison group, the authors were unable to determine the role of aerobic exercise in preventing declines in aerobic fitness during NACRT or facilitating recovery of aerobic fitness after NACRT. Randomized, controlled trials are needed to answer that question.
The pattern of decline for many of the generic and cancer-specific quality-of-life outcomes exceeded the two- to three-point clinically meaningful difference during NACRT, despite the aerobic exercise intervention. In addition, most quality-of-life outcomes recovered after NACRT, and some even exceeded pre-NACRT levels at the presurgery time point. Once again, however, without a proper comparison group, the authors were unable to comment on whether exercise modified the declines in quality of life during NACRT or the recovery in quality of life after NACRT. Randomized, controlled trials will be critical to determine the potential benefits and harms of aerobic exercise for symptom management and quality of life in this clinical setting.
The study has important strengths and limitations. The authors closely tracked eligibility and recruitment rates to determine the potential reach of the intervention. The exercise intervention during NACRT was supervised, and all serious adverse events and reasons for missed exercise sessions were documented. In addition, the authors used validated measures to assess health-related fitness and patient reported-outcomes. One limitation is the lack of a comparison group. Given that the study was the first to test an exercise intervention in patients with rectal cancer during NACRT, the authors felt that it was prudent to conduct a phase I study to determine the feasibility and safety of exercise before embarking on randomized, controlled trials to determine efficacy.
Conclusion and Implications for Nursing
This phase I study demonstrated acceptable eligibility, recruitment, adherence, and follow-up assessment rates for an aerobic exercise intervention during and after NACRT. Despite the substantial side effects and toxicity associated with NACRT, a supervised aerobic exercise intervention in patients with rectal cancer during NACRT followed by an unsupervised exercise program after NACRT appears to be feasible and safe for patients. Phase II trials in patients with rectal cancer during and after NACRT with proper comparison groups are now needed to establish the benefits (and potential harms) of aerobic exercise on various outcomes, including cardiorespiratory fitness, physical functioning, symptom management, quality of life, treatment toxicity, treatment completion, treatment response, and postsurgical recovery. Nurses will play a key role in disseminating the knowledge generated from these trials to their patients, including the potential benefits and risks of exercising during NACRT.
References
Beck, S.L., Schwartz, A.L., Towsley, G., Dudley, W., & Barsevick, A. (2004). Psychometric evaluation of the Pittsburgh Sleep Quality Index in cancer patients. Journal of Pain and Symptom Management, 27, 140–148. doi:10.1016/j.jpainsymman.2003.12.002
Buysse, D.J., Reynolds, C.F., 3rd, Monk, T.H., Berman, S.R., & Kupfer, D.J. (1989). The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research. Psychiatry Research, 28, 193–213. doi:10.1016/0165-1781(89)90047-4
Cella, D., & Nowinski, C.J. (2002). Measuring quality of life in chronic illness: The Functional Assessment of Chronic Illness Therapy measurement system. Archives of Physical Medicine and Rehabilitation, 83(Suppl. 2), S10–S17. doi:10.1053/apmr.2002.36959
Cohen, S., Kamarck, T., & Mermelstein, R. (1983). A global measure of perceived stress. Journal of Health and Social Behavior, 24, 385–396.
Courneya, K.S., McKenzie, D.C., Mackey, J.R., Gelmon, K., Friedenreich, C.M., Yasui, Y., . . . Segal, R.J. (2013). Effects of exercise dose and type during breast cancer chemotherapy: Multicenter randomized trial. Journal of the National Cancer Institute, 105, 1821–1832. doi:10.1093/jnci/djt297
Courneya, K.S., Segal, R.J., Mackey, J.R., Gelmon, K., Reid, R.D., Friedenreich, C.M., . . . McKenzie, D.C. (2007). Effects of aerobic and resistance exercise in breast cancer patients receiving adjuvant chemotherapy: A multicenter randomized controlled trial. Journal of Clinical Oncology, 25, 4396–4404. doi:10.1200/JCO.2006.08.2024
Courneya, K.S., Segal, R.J., McKenzie, D.C., Dong, H., Gelmon, K., Friedenreich, C.M., . . . Mackey, J.R. (2014). Effects of exercise during adjuvant chemotherapy on breast cancer outcomes. Medicine and Science in Sports and Exercise, 46, 1744–1751. doi:10.1249/MSS.0000000000000297
Courneya, K.S., Sellar, C.M., Stevinson, C., McNeely, M.L., Peddle, C.J., Friedenreich, C.M., . . . Reiman, T. (2009). Randomized controlled trial of the effects of aerobic exercise on physical functioning and quality of life in lymphoma patients. Journal of Clinical Oncology, 27, 4605–4612. doi:10.1200/JCO.2008.20.0634
Cramer, H., Lauche, R., Klose, P., Dobos, G., & Langhorst, J. (2014). A systematic review and meta-analysis of exercise interventions for colorectal cancer patients. European Journal of Cancer Care, 23, 3–14. doi:10.1111/ecc.12093
Cravo, M., Rodrigues, T., Ouro, S., Ferreira, A., Féria, L., & Maio, R. (2014). Management of rectal cancer: Times they are changing. GE Portuguese Journal of Gastroenterology, 21, 192–200. doi:10.1016/j.jpg.2014.06.003
Dewdney, A., Cunningham, D., & Chau, I. (2013). Selecting patients with locally advanced rectal cancer for neoadjuvant treatment strategies. Oncologist, 18, 833–842. doi:10.1634/theoncologist .2013-0022
Gavaruzzi, T., Giandomenico, F., & Pucciarelli, S. (2013). Quality of life and functions after chemoradiation for rectal cancer: A review of recent publications. Current Colorectal Cancer Reports, 9, 157–167. doi:10.1007/s11888-013-0161-x
Godin, G., & Shephard, R.J. (1985). A simple method to assess exercise behavior in the community. Canadian Journal of Applied Sport Sciences, 10, 141–146.
Heyward, V.H. (2006). Advanced fitness assessment and exercise prescription (5th ed.). Champaign, IL: Human Kinetics.
Jones, C.J., & Rikli, R.E. (2002). Measuring functional fitness of older adults. Journal on Active Aging, 1, 24–30.
Kohout, F.J., Berkman, L.F., Evans, D.A., & Cornoni-Huntley, J. (1993). Two shorter forms of the CES-D (Center for Epidemiological Studies Depression) depression symptoms index. Journal of Aging and Health, 5, 179–193. doi:10.1177/089826439300500202
Lin, K.Y., Shun, S.C., Lai, Y.H., Liang, J.T., & Tsauo, J.Y. (2014). Comparison of the effects of a supervised exercise program and usual care in patients with colorectal cancer undergoing chemotherapy. Cancer Nursing, 37, E21–E29. doi:10.1097/NCC.0b013e3182791097
Mishra, S.I., Scherer, R.W., Snyder, C., Geigle, P.M., Berlanstein, D.R., & Topaloglu, O. (2012). Exercise interventions on health-related quality of life for people with cancer during active treatment. Cochrane Database of Systematic Reviews, 8, 1–459. doi:10.1002/14651858.CD008465.pub2
National Institutes of Health. (2000). The practical guide identification, evaluation, and treatment of overweight and obesity in adults. Retrieved from http://www.nhlbi.nih.gov/files/docs/guidelines/prctgd_c.pdf
Rikli, R.E., & Jones, C.J. (1999). Development and validation of a functional fitness test for community-residing older adults. Journal of Aging and Physical Activity, 7, 129–161.
Rosenberg, M. (1965). Society and the adolescent self-image. Princeton, NJ: Princeton University Press.
Sauer, R., Liersch, T., Merkel, S., Fietkau, R., Hohenberger, W., Hess, C., . . . Rodel, C. (2012). Preoperative versus postoperative chemoradiotherapy for locally advanced rectal cancer: Results of the German CAO/ARO/AIO-94 randomized phase III trial after a median follow-up of 11 years. Journal of Clinical Oncology, 30, 1926–1933. doi:10.1200/jco.2011.40.1836
Segal, R.J., Reid, R.D., Courneya, K.S., Sigal, R.J., Kenny, G.P., Prud’Homme, D.G., . . . Slovinec D’Angelo, M.E. (2009). Randomized controlled trial of resistance or aerobic exercise in men receiving radiation therapy for prostate cancer. Journal of Clinical Oncology, 27, 344–351. doi:10.1200/JCO.2007.15.4963
Singh, F., Newton, R.U., Galvao, D.A., Spry, N., & Baker, M.K. (2013). A systematic review of pre-surgical exercise intervention studies with cancer patients. Surgical Oncology, 22, 92–104. doi:10.1016/j.suronc.2013.01.004
Spielberger, C., Gorsuch, R., Lushene, R., Vagg, P.R., & Jacobs, G.A. (1983). Manual for the State-Trait Anxiety Inventory. Palo Alto, CA: Consulting Psychologists Press.
Swellengrebel, H.A., Marijnen, C.A., Verwaal, V.J., Vincent, A., Heuff, G., Gerhards, M.F., . . . Cats, A. (2011). Toxicity and complications of preoperative chemoradiotherapy for locally advanced rectal cancer. British Journal of Surgery, 98, 418–426. doi:10.1002/bjs.7315
Thompson, W.R., Gordon, N.F., & Pescatello, L.S. (2010). ACSM’s guidelines for exercise testing and prescription (8th ed.). Philadelphia, PA: Lippincott Williams and Wilkins.
Van Waart, H., Stuiver, M.M., van Harten, W.H., Geleijn, E., Kieffer, J.M., Buffart, L.M., . . . Aaronson, N.K. (2015). Effect of low-intensity physical activity and moderate- to high-intensity physical exercise during adjuvant chemotherapy on physical fitness, fatigue, and chemotherapy completion rates: Results of the PACES randomized clinical trial. Journal of Clinical Oncology, 33, 1918–1927. doi:10.1200/JCO.2014.59.1081
Ward, W.L., Hahn, E.A., Mo, F., Hernandez, L., Tulsky, D.S., & Cella, D. (1999). Reliability and validity of the Functional Assessment of Cancer Therapy-Colorectal (FACT-C) quality of life instrument. Quality of Life Research, 8, 181–195. doi:10.1023/A:1008821826499
Ware, J.E., Kosinski, M., Bjorner, J.B., Turner-Bowker, D.M., Gandek, B., & Maruish, M.E. (2007). User’s manual for the SF-36v2 health survey (2nd ed.). Lincoln, NE: Quality Metric Incorporated.
West, M.A., Loughney, L., Barben, C.P., Sripadam, R., Kemp, G.J., Grocott, M.P.Q., & Jack, S. (2014). The effects of neoadjuvant chemoradiotherapy on physical fitness and morbidity in rectal cancer surgery patients. European Journal of Surgical Oncology, 40, 1421–1428. doi:10.1016/j.ejso.2014.03.021
West, M.A., Loughney, L., Lythgoe, D., Barbe, C.P., Sripadam, R., Kemp, G.J., Grocott, M.P.W., & Jack, S. (2015). Effect of prehabilitation on objectively measured physical fitness after neoadjuvant treatment in preoperative rectal cancer patients: a blinded interventional pilot study. British Journal of Anaesthesia, 114, 244–251. doi:10.1093/bja/aeu318
World Health Organization. (2008). Waist circumference and waist-hip ratio: Report of a WHO expert consultation. Geneva, Switzerland: Author.
Yellen, S.B., Cella, D.F., Webster, K., Blendowski, C., & Kaplan, E. (1997). Measuring fatigue and other anemia-related symptoms with the Functional Assessment of Cancer Therapy (FACT) measurement system. Journal of Pain and Symptom Management, 13, 63–74. doi:10.1016/S0885-3924(96)00274-6
About the Author(s)
Morielli is a doctoral student in the Faculty of Physical Education and Recreation at the University of Alberta; Usmani is an associate professor in the Department of Oncology at the University of Alberta and a radiation oncologist at Cross Cancer Institute; Boulé is an associate professor in the Faculty of Physical Education and Recreation at the University of Alberta; Tankel, Severin, and Nijjar are all clinical professors in the Department of Oncology at the University of Alberta and radiation oncologists at Cross Cancer Institute; Joseph is an associate professor in the Department of Oncology at the University of Alberta and a radiation oncologist at Cross Cancer Institute; and Courneya is a professor in the Faculty of Physical Education and Recreation at the University of Alberta, all in Edmonton, Canada. Courneya was supported by the Canada Research Chairs Program. Morielli, Usmani, Boulé, and Courneya contributed to the conceptualization and design. Morielli, Usmani, Tankel, Severin, Nijjar, and Joseph completed the data collection. Morielli, Boulé, and Courneya provided the statistical support. Morielli and Courneya contributed to the analysis. Morielli, Usmani, Boulé, Tankel, Severin, Nijjar, Joseph, and Courneya contributed to the manuscript preparation. Mention of specific products and opinions related to those products do not indicate or imply endorsement by the Oncology Nursing Forum or the Oncology Nursing Society. Morielli can be reached at morielli@ualberta.ca, with copy to editor at ONFEditor@ons.org. Submitted September 2015. Accepted for publication November 10, 2015.

