Message Framing and Physical Activity Promotion in Colorectal Cancer Survivors
Purpose/Objectives: To test effects of gain-framed versus loss-framed mailed brochures on increasing physical activity (PA) among colorectal cancer (CRC) survivors.
Design: Randomized trial with repeated measures at baseline, 1 month, and 12 months postintervention.
Setting: Mail recruitment from tumor registries.
Sample: 148 inactive CRC survivors who had completed primary therapy.
Methods: PA and constructs from the Theory of Planned Behavior (TPB) were assessed at baseline, 1 month, and 12 months. Participants were randomized to receive pamphlets describing PA benefits (gain framed) or disadvantages of not being physically active (loss framed). Baseline characteristics were compared using descriptive statistics. Repeated measures linear models were used to test PA changes.
Main Research Variables: Minutes of PA and TPB constructs.
Findings: Significant PA increases were observed in both study arms. Results did not differ by message frame. At one month, about 25% of previously inactive participants increased activity to national recommendations. Those who increased PA compared to those who did not had higher baseline scores on subjective norms, perceived behavioral control, and PA intentions.
Conclusions: Independent of message framing, mailed brochures are highly effective in producing within-subject short- and long-term increases in PA.
Implications for Nursing: CRC survivors may increase short- and long-term levels of PA by receiving inexpensive print brochures.
Jump to a section
Among colorectal cancer (CRC) survivors, higher levels of physical activity (PA) postdiagnosis are related to lower risk of cancer recurrence and cancer-specific and all-cause mortality (Meyerhardt et al., 2009; Meyerhardt, Giovannucci, et al., 2006; Meyerhardt, Heseltine, et al., 2006). For example, among 1,825 stage I–III CRC survivors who were followed longitudinally for five years postdiagnosis, those who engaged in some level of PA after diagnosis had 25%–28% lower all-cause mortality risk compared to sedentary survivors (Baade et al., 2011). In addition, a meta-analysis of seven studies indicated that the risk of overall mortality decreases by 28% with an increase to roughly 150 minutes of moderate-intensity activity per week (Schmid & Leitzmann, 2014). Because of these and other PA benefits (e.g., improved quality of life), the American Cancer Society advises 150 minutes of moderate-intensity or 75 minutes of vigorous-intensity aerobic activity weekly for cancer survivors (Doyle et al., 2006). Unfortunately, as many as 65% of CRC survivors fail to meet this recommendation (Blanchard, Courneya, & Stein, 2008).
Of the few interventions that have aimed to promote PA among CRC survivors, some have resulted in significant increases relative to baseline (Hawkes et al., 2013; Lee et al., 2013; Pinto, Papandonatos, Goldstein, Marcus, & Farrell, 2013), whereas others have not (Courneya et al., 2003). Most effective interventions have been resource intensive, consisting of in-person sessions conducted individually or in a group setting (Anderson, Caswell, Wells, Steele, & MacAskill, 2009; Bourke et al., 2011; Lee et al., 2013; Spence, Heesch, Eakin, & Brown, 2007). Home-based interventions have also proven effective but are still fairly resource intensive because they have relied on telephone counseling and tailoring of feedback during multiple time points (Demark-Wahnefried et al., 2012; Hawkes et al., 2013; Hawkes, Gollschewski, Lynch, & Chambers, 2009; Ho et al., 2013; Ligibel et al., 2011; Morey et al., 2009).
Less intensive interventions that require fewer financial and staffing resources may be most easily disseminated to increase PA among survivors. Examples of such interventions include a single telephone call, brief counseling sessions, or educational brochures. Health education print brochures have increased PA among breast and prostate cancer survivors (Demark-Wahnefried et al., 2007; Short, James, Girgis, D’Souza, & Plotnikoff, 2014; Vallance, Courneya, Plotnikoff, Yasui, & Mackey, 2007) and, therefore, also may be effective among CRC survivors. These more simple interventions may be effective for cancer survivors compared to other segments of the population who require intensive interventions, such as health coaching, for several reasons. First, cancer survivors are generally motivated to pursue healthful lifestyle change (Demark-Wahnefried, Aziz, Rowland, & Pinto, 2005). Second, survivors seek basic PA information as they report wanting PA to be included in survivorship care plans (Smith, Singh-Carlson, Downie, Payeur, & Wai, 2011). Finally, cancer survivors indicate print materials are a preferred PA intervention delivery method (Stull, Snyder, & Demark-Wahnefried, 2007).
The efficacy of print messages in promoting behavior change may depend on how information is framed. Consistent with prospect theory (Kahneman & Tversky, 1979), messages stressing the benefits of engaging in PA (gain-framed messages) have been more effective than messages focused on the disadvantages of not engaging in PA (loss-framed messages) (Gallagher & Updegraff, 2011; Latimer, Brawley, & Bassett, 2010).
Individuals are generally more motivated to engage in health behaviors when they believe little uncertainty and risk are involved (Wakker, 2010). Therefore, the authors believe that gain-framed messages are more effective for PA because they pose little risk and uncertainty. In contrast, loss-framed messages are hypothesized to be more effective for health behaviors associated with uncertainty and risk. Among non-cancer populations, gain-framed messages have generally been more effective than loss-framed messages in PA interventions (Latimer et al., 2008; McCall & Martin Ginis, 2004); however, some interventions have resulted in no PA differences between gain- and loss-framed messages (Jones, Sinclair, Rhodes, & Courneya, 2004). No research, to date, has focused on understanding the effects of message framing on PA among cancer survivors in general, and CRC survivors specifically.
Theoretical Background
In this randomized trial, the effects of gain- versus loss-framed educational brochures that provided strategies to increase PA among inactive CRC survivors were explored. Both gain- and loss-framed messages were crafted to target the main constructs of the Theory of Planned Behavior (TPB) (Ajzen, 1991). The TPB has been used to promote PA among CRC survivors (Ho et al., 2013; Packel, Prehn, Anderson, & Fisher, 2014; Speed-Andrews et al., 2013) and contains constructs that predict adherence to exercise among cancer survivors (Husebø, Dyrstad, Søreide, & Bru, 2013).
According to the TPB, behavioral intentions are most proximal to behavior. Intentions are influenced by attitudes (i.e., one’s overall evaluation of a behavior), subjective norms (i.e., perceived social pressure to engage or not to engage in a behavior), and perceived behavioral control (i.e., individual’s evaluation of personal control over a behavior). Therefore, the content of the brochures contained tips intended to increase positive attitudes, subjective norms, and perceived behavioral control related to PA. Following the TPB framework, successful increases in each of these constructs increases intentions, which will, in turn, increase PA.
The overarching purpose of this study is to compare the effectiveness of gain- versus loss-framed messages targeted at TPB constructs to increase PA among CRC survivors. Two hypotheses were tested: (a) levels of PA relative to baseline will increase in both intervention arms and (b) levels of PA will increase more in the gain-framed intervention arm compared to the loss-framed intervention arm. In addition, an exploratory analysis was conducted to follow the theoretical framework by testing intervention effects on all TPB constructs. This was done to provide insights into how the messages produced any changes.
Methods
Sample
Study inclusion criteria were (a) patients aged 18 years and older with diagnoses of early-stage (I–II) CRC who had adequately recovered from surgical excision of cancer and completed adjuvant therapy (if appropriate) within the previous six months to five years; (b) no evidence of recurrence; (c) no preexisting medical condition(s) that precluded adherence to an unsupervised PA program (e.g., severe orthopedic conditions, scheduled for a hip or knee replacement within six months, paralysis and/or dementia, unstable angina, or those who had experienced a heart attack, congestive heart failure, pulmonary conditions that required oxygen or hospitalization within six months); (d) approved for contact by their oncologic care physician; (e) community dwelling (i.e., not residing in a skilled nursing facility); (f) English-speaking and writing and who had completed the fifth grade or higher; and (g) inactive (i.e., participating in fewer than 150 minutes of moderate- or strenuous-intensity PA per week).
Potential study participants were recruited from the Duke University Medical Center’s Tumor Registry and the North Carolina Central Cancer Registry. Upon receipt of names and contact information, potential study participants received an invitation packet that included a consent form, a screener questionnaire that assessed eligibility, and a self-addressed stamped envelope to return the completed questionnaire and consent. Upon receipt of the questionnaire, those found eligible were telephoned for a baseline survey. Those who did not return the questionnaire within two weeks were contacted to complete the questionnaire via telephone. Those deemed eligible provided verbal consent for participation and completed the baseline survey. The study proscribed to state and national ethical standards and was approved by the Duke University School of Nursing Center and Medical Center institutional review board. Informed written consent was obtained from the CRC survivors.
Intervention
All participants received a single-page tri-folded educational brochure with an insert that had four main sections: (a) tips on how to become more physically active (e.g., get friends and family to help, blocking off time on your daily calendar), with examples of activities of moderate-intensity PA; (b) description of other diseases for which CRC survivors are at increased risk (e.g., heart disease, diabetes, second cancers) and the protective influence of PA on these comorbid conditions; (c) description and results of two epidemiologic studies that showed a significant inverse relationship between self-reported PA and risk of cancer-specific mortality and all-cause mortality in CRC survivors; and (d) a summary of benefits of being or disadvantages of not being PA. The brochure content aimed to increase the TPB constructs. For example, benefits of PA were included to increase attitudes, messages about what other people think were included to increase subjective norms, and messages about being able to do something positive for oneself were included to increase perceived behavioral control. Table 1 includes examples of gain- and loss-framed messages targeting TPB constructs from the study brochures. Two gender-specific focus groups of 4–5 CRC survivors per group were used to solicit input to develop the materials. The revised brochures were then reevaluated by focus group participants for clarity and comprehensiveness before their use in the trial.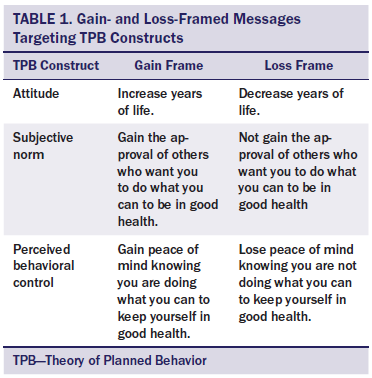
Measures
PA and PA intentions were assessed through surveys at baseline, 1 month, and 12 months after the intervention. Attitudes, subjective norms, and perceived behavioral control also were assessed at baseline and at 1 month. At all time points, questions were asked in reference to engaging in regular exercise during the next month, defined for participants as any exercise sessions that last more than 30 minutes, during their free time, at least three times per week. TPB constructs of attitudes, subjective norms, and perceived behavioral control were validated previously with patients with cancer (Cronbach alpha > 0.72) (Lowe, Watanabe, Baracos, & Courneya, 2012). Participants received $10 for completing each survey ($30 total).
Physical activity: Self-reported PA was evaluated using the Godin Leisure-Time Exercise Questionnaire (GLTEQ) to assess weekly minutes of aerobic PA (Godin & Shephard, 1985). The GLTEQ lists three questions that assess the average frequency of mild- (minimal effort, no perspiration), moderate- (not exhausting, light perspiration), and strenuous-intensity (heart beats rapidly, sweating) PA during free time in a typical week. Participants were asked to indicate the average duration (in minutes) within each PA intensity level during the past week. Separate scores were calculated for total PA minutes, as well as subcategories of strenuous-, moderate-, and mild-intensity PA. In line with the American Cancer Society’s and the American College of Sports Medicine’s recommendations that cancer survivors should engage in at least 150 minutes per week of moderate-intensity PA (Rock et al., 2012; Schmitz et al., 2010), the proportion of survivors who met this minimum standard by message frame was assessed. The GLTEQ demonstrates 75% sensitivity and 59% specificity to measure PA among breast cancer survivors (Amireault, Godin, Lacombe, & Sabiston, 2015).
Attitudes: Six bipolar scales were used to assess PA attitudes: unenjoyable versus enjoyable, harmful versus beneficial, boring versus interesting, foolish versus wise, unpleasant versus pleasant, and bad versus good. Items were rated on a Likert-type scale from 1 (strongly disagree) to 7 (strongly agree), summed, and then averaged.
Subjective norms: Three items assessed subjective norms. Most people who are important to me . . . (a) think I should exercise regularly over the next month, (b) would encourage me to exercise regularly over the next month, or (c) would approve of me exercising regularly over the next month. Items were rated on a Likert-type scale from 1 (strongly disagree) to 7 (strongly agree).
Perceived behavioral control: Three questions assessed perceived behavioral control. “If you were really motivated, exercising regularly over the next month would be (1 [extremely hard] to 7 [extremely easy])”, “If you were really motivated, how confident will you be at exercising regularly over the next month?” (1 [not at all confident] to 7 [extremely confident]), and “If you were really motivated, how much control do you feel you have over exercising regularly over the next month?” (1 [very little control] to 7 [complete control]).
Intention: Three questions assessed PA intention. “How motivated are you to exercise regularly over the next month?” (1 [extremely unmotivated] to 7 [extremely motivated]), “I intend to do everything I can to exercise regularly over the next month.” (1 [strongly disagree] to 7 [strongly agree]), and “How committed are you to exercise regularly over the next month?” (1 [extremely uncommitted] to 7 [extremely committed]).
Evaluation and use of the brochures: At the one-month follow-up, participants rated their brochure on usefulness and accuracy (from 1 [not at all] to 7 [extremely]), how much they read (from 1 [not at all] to 7 [read it all]), and whether they reviewed the brochure with anyone (“yes” or “no”).
Manipulation check: At the one-month follow-up, participants were asked, “Overall, to what extent did the brochure focus on the benefits you gain from exercising regularly or the disadvantages of not exercising regularly” (from 1 [emphasized benefits] to 7 [emphasized disadvantages]).
Data Analyses
As with many studies that focus on PA (Demark-Wahnefried et al., 2014), several study measures were non-normally distributed, so both parametric (t tests) and non-parametric (Wilcoxon rank sum tests) methods were explored to evaluate frame effects. In general, both methods supplied similar inferences; therefore, for simplicity, results from the parametric analyses are presented. In contrast, all change scores were normally distributed, so no adjustments were necessary. Hypothesis tests were evaluated using t tests and Ordinary Least Squares analysis. The latter was associated with repeated measures (i.e., pre-/post-) linear models to test changes in mean minutes of PA. Unadjusted associations (e.g., changes in minutes of PA with changes in TPB constructs collapsing across framing condition) were evaluated using Pearson correlations. To identify clusters of individuals following similar progressions of minutes of PA, group base trajectory modeling was conducted using the SAS® Proc TRAJ method (Jones, Nagin, & Roeder, 2001). Whether frame type (gain or loss) was associated with gender, type of cancer (colon versus rectum), time elapsed since diagnosis, and sharing of the brochures to affect total (moderate plus strenuous), strenuous, and moderate levels of PA, controlling for baseline values was explored. All the aforementioned analyses were conducted using SAS, version 9.2.
Results
Sample
A total of 1,777 invitation packets were mailed to survivors in 17 states. Accrual is shown in Figure 1. Characteristics of the study sample are presented in Table 2. No significant differences were noted in medical and demographic characteristics between participants who completed and did not complete the 1- or 12-month follow-up phone survey.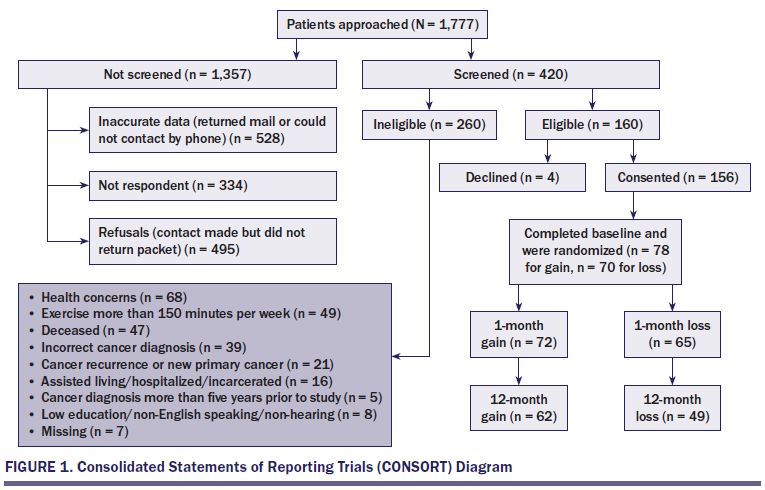
Manipulation Check
With respect to use and evaluation of brochures, participants in both framing conditions reported reading most of the brochure (mean = 6.4 versus mean = 6.6, out of a max score of 7, respectively; t = 0.94; p = 0.35). As intended, the gain-frame brochure was perceived as emphasizing the benefits of regular PA, whereas the loss-frame brochure was perceived as emphasizing the disadvantages of not exercising regularly (mean = 1.8 versus mean = 2.7, respectively; t = 2.57; p < 0.02). Both the gain- and loss-framed brochures were viewed as highly useful (mean = 5.8 versus mean = 6.1, respectively; t = 1.73; p = 0.09) and accurate (mean = 6.2 versus mean = 6.3, respectively; t = 0.6; p = 0.55).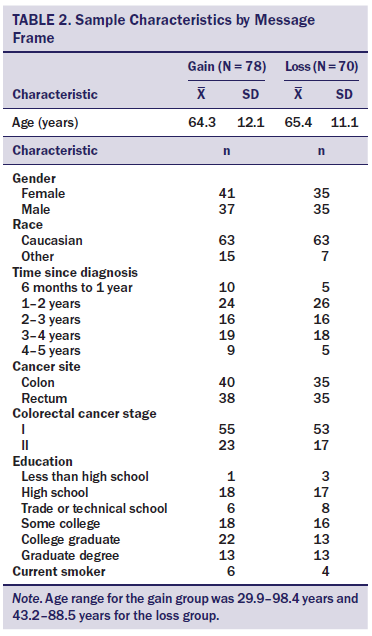
Changes in Physical Activity
PA increased in both intervention arms; therefore, the authors’ first hypothesis was supported. Specifically, across all intensity levels, mean minutes of PA increased at both follow-up time points relative to baseline (p < 0.0001). Overall, based on the trajectory analysis, 83% (n = 92) exhibited an increase in total minutes of moderate to strenuous PA relative to baseline, with mean change relative to baseline of 70 and 66 minutes per week at 1 month and 12 months, respectively (p < 0.001). At 1 month and 12 months, 25% (n = 34) and 10% (n = 11) of survivors, respectively, achieved the PA goal. Among those who achieved at least 150 weekly minutes of moderate-intensity PA at 1 month (n = 34) and completed the 12-month follow-up, 42% (n = 11) maintained the goal.
No significant between-arm differences were noted in PA; therefore, the authors’ second hypothesis was not supported. No main effects of frame or time by frame interactions were found. Effects of message framing on self-reported PA are presented in Table 3.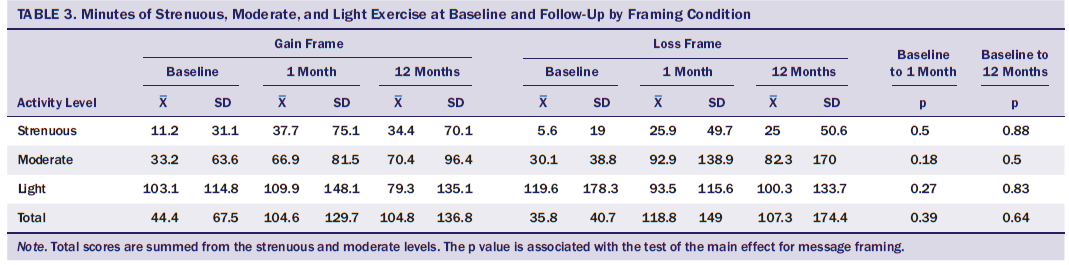
Theory of Planned Behavior Constructs
In this study, baseline and follow-up measures of the TPB constructs had internal consistency scores of Cronbach alpha > 0.7. Neither the gain- nor loss-framed brochures produced significant changes in the TPB constructs from baseline to the 1-month follow-up, nor in PA intentions at the 12-month follow-up (see Table 4).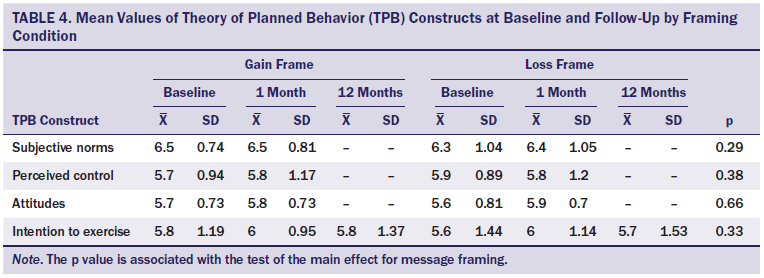
Baseline Predictors of Changes in Physical Activity
Baseline TBP constructs were tested as predictors of participants increasing or not increasing PA. Overall, the group that increased PA had higher baseline scores on subjective norms (mean = 6.5 versus mean = 5.9, p < 0.03) and perceived control (mean = 5.9 versus mean = 5.4, p < 0.05), and stronger intentions to exercise (mean = 5.8 versus mean = 5). The two groups did not differ on attitudes. No significant main effects or interactions with regard to gender, age, type of cancer (colon versus rectal), and time elapsed since diagnosis were observed.
Discussion
This study is the first to test effects of message-framing brochures as a strategy to increase PA among inactive CRC survivors and to determine if this strategy merits testing in a subsequent larger randomized, controlled trial. The main findings of this study, supporting the first hypothesis, are that both conditions produced significant increases in PA. At the one-month follow-up, about 25% (n = 34) of previously inactive participants were meeting the national recommendations for PA for cancer survivors. Overall, changes of this magnitude are noteworthy and could have major clinical and public health significance if sustained over the long-term.
The second hypothesis was not supported, as there were no differences by framing condition. These findings are divergent with previous research that indicates gain-framed messages are more effective than loss-framed messages to increase PA (Gallagher & Updegraff, 2011; Latimer et al., 2010). In the current study, it is possible that there were not between-framing condition differences because the loss-framed intervention materials were not effective. In other words, based on the manipulation check, the loss-frame brochure was not perceived as strongly emphasizing the disadvantages of inactive behavior. Therefore, it may have been perceived as a milder form of the gain-frame brochure, rather than a loss-framed message as intended. In addition, both brochures provided tips and ideas on improving health via PA, and both provided information on studies that found benefit with PA. Therefore, this information may have attenuated differences between gain- and loss-framed messages as the loss-framed group received some information about the benefits of being PA.
No changes in the TPB constructs were noted, despite an increase in PA. It is possible that the study participants were motivated prior to receipt of any materials to engage in PA. The mean scores of attitudes and intentions were both above scale midpoint scores of 4 at baseline. Therefore, participants eager to join an exercise study may have enrolled in this study. As such, the gain- and loss-framed materials may have served equally to prime participants into engaging in PA (i.e., ceiling effect). This is consistent with the trajectory analyses, of which 83% evidenced some form of increase in PA. Overall, higher baseline mean scores on social norms, perceived control, and intentions all predicted increased PA over time.
Although the current study is one of the largest to date to assess the impact of a PA intervention on CRC survivors, some important limitations need to be considered. As with any behavior change intervention, the list of possible confounding factors is endless and cannot all be controlled for in a single study. Factors, such as time elapsed from diagnosis, could be controlled, but many other factors could not be controlled. Other limitations include the reliance on self-reported data (rather than objective measures), the self-select nature of the study sample, a largely Caucasian and educated sample, and a trajectory analysis based on a relatively small number of cases. Finally, an important caveat to the findings is that the study did not include a usual care group (e.g., a group that received no information). As such, it remains unclear whether the message framing along with the accompanying materials did better than no information at all. It is possible that the relatively motivated and self-selected sample may have increased their level of PA just by being primed by the authors’ contact. In addition, noted limitations are minimized by use of a well-validated questionnaire and the broad sampling approach that reached 17 states.
Implications for Nursing
Nurses are increasingly leading survivorship care and are well positioned to promote exercise. Minimally intensive interventions that can be implemented readily for a range of patients in clinic settings can help with this goal (Pinto & Floyd, 2008). These study findings indicate that minimal interventions, such as the use of print brochures, can be an effective means of promoting PA among CRC survivors. PA brochures can be easily disseminated by nurses through the mail, incorporated into existing patient teaching materials, or made available in patient waiting areas. These strategies are promising because they do not require a lot of additional time or money, which is important in the setting of increased survivorship care demands.
Implications for Research
Although this study produced significant increases in PA, room for improvement still exists. For example, about 18% of the sample did not show increases in PA in response to the interventions. For these individuals, a subsequent and more intensive intervention may be needed to boost their motivation for change, as well as their ability to adhere to the intervention. Such an approach would be consistent with a stepped or adaptive design strategy in which individuals are initially given a minimally intensive intervention (e.g., print brochure) and then assessed for meeting a criterion (e.g., reaching the PA goal); those not meeting the goal would then be assigned a more intensive intervention (e.g., health coaching). To date, a stepped care trial has not been conducted to increase PA in cancer survivors.
Conclusion
The data strongly suggest that brief educational brochures emphasizing strategies to increase PA are well received, regardless of message frame, and produced significant increases in PA among inactive CRC survivors. Given that brochures are used often to motivate PA behavior as part of usual care, testing the short- and longer-term effects of brochures among CRC survivors may offer a more cost-effective approach than intense and resource-demanding interventions that currently characterize the field.
The authors gratefully acknowledge the Duke University Medical Center Tumor Registry and the North Carolina Central Cancer Registry for their support of this study. The authors also thank Peter Salovey, PhD, for his constructive feedback on the message framing manipulation.
References
Ajzen, I. (1991). The Theory of Planned Behavior. Organizational Behavior and Human Decision Processes, 50, 179–211.
Amireault, S., Godin, G., Lacombe, J., & Sabiston, C.M. (2015). Validation of the Godin-Shephard Leisure-Time Physical Activity Questionnaire classification coding system using accelerometer assessment among breast cancer survivors. Journal of Cancer Survivorship: Research and Practice, 9, 532–540. doi:10.1007/s11764-015-0430-6
Anderson, A.S., Caswell, S., Wells, M., Steele, R.J., & MacAskill, S. (2009). “It makes you feel so full of life” LiveWell, a feasibility study of a personalised lifestyle programme for colorectal cancer survivors. Supportive Care in Cancer, 18, 409–415. doi:10.1007/s00520-009-0677-4
Baade, P.D., Meng, X., Youl, P.H., Aitken, J.F., Dunn, J., & Chambers, S.K. (2011). The impact of body mass index and physical activity on mortality among patients with colorectal cancer in Queensland, Australia. Cancer Epidemiology, Biomarkers and Prevention, 20, 1410–1420. doi:10.1158/1055-9965.EPI-11-0079
Blanchard, C.M., Courneya, K.S., & Stein, K. (2008). Cancer survivors’ adherence to lifestyle behavior recommendations and associations with health-related quality of life: Results from the American Cancer Society’s SCS-II. Journal of Clinical Oncology, 26, 2198–2204. doi:10.1200/JCO.2007.14.6217
Bourke, L., Thompson, G., Gibson, D.J., Daley, A., Crank, H., Adam, I., . . . Saxton, J. (2011). Pragmatic lifestyle intervention in patients recovering from colon cancer: A randomized controlled pilot study. Archives of Physical Medicine and Rehabilitation, 92, 749–755. doi:10.1016/j.apmr.2010.12.020
Courneya, K.S., Friedenreich, C.M., Quinney, H.A., Fields, A.L., Jones, L.W., & Fairey, A.S. (2003). A randomized trial of exercise and quality of life in colorectal cancer survivors. European Journal of Cancer Care, 12, 347–357.
Demark-Wahnefried, W., Aziz, N.M., Rowland, J.H., & Pinto, B.M. (2005). Riding the crest of the teachable moment: Promoting long-term health after the diagnosis of cancer. Journal of Clinical Oncology, 23, 5814–5830. doi:10.1200/JCO.2005.01.230
Demark-Wahnefried, W., Clipp, E.C., Lipkus, I.M., Lobach, D., Snyder, D.C., Sloane, R., . . . Kraus, W.E. (2007). Main outcomes of the FRESH START trial: A sequentially tailored, diet and exercise mailed print intervention among breast and prostate cancer survivors. Journal of Clinical Oncology, 25, 2709–2718. doi:10.1200/JCO.2007.10.7094
Demark-Wahnefried, W., Jones, L.W., Snyder, D.C., Sloane, R.J., Kimmick, G.G., Hughes, D.C., . . . Lipkus, I.M. (2014). Daughters and Mothers Against Breast Cancer (DAMES): Main outcomes of a randomized controlled trial of weight loss in overweight mothers with breast cancer and their overweight daughters. Cancer, 120, 2522–2534. doi:10.1002/cncr.28761
Demark-Wahnefried, W., Morey, M.C., Sloane, R., Snyder, D.C., Miller, P.E., Hartman, T.J., & Cohen, H.J. (2012). Reach out to enhance wellness home-based diet-exercise intervention promotes reproducible and sustainable long-term improvements in health behaviors, body weight, and physical functioning in older, overweight/obese cancer survivors. Journal of Clinical Oncology, 30, 2354–2361. doi:10.1200/JCO.2011.40.0895
Doyle, C., Kushi, L.H., Byers, T., Courneya, K.S., Demark-Wahnefried, W., Grant, B., . . . Andrews, K.S. (2006). Nutrition and physical activity during and after cancer treatment: An American Cancer Society guide for informed choices. CA: A Cancer Journal for Clinicians, 56, 323–353.
Gallagher, K.M., & Updegraff, J.A. (2011). Health message framing effects on attitudes, intentions, and behavior: A meta-analytic review. Annals of Behavioral Medicine, 43, 101–116.
Godin, G., & Shephard, R.J. (1985). A simple method to assess exercise behavior in the community. Canadian Journal of Applied Sport Sciences, 10(3), 141–146.
Hawkes, A.L., Chambers, S.K., Pakenham, K.I., Patrao, T.A., Baade, P.D., Lynch, B.M., . . . Courneya, K.S. (2013). Effects of a telephone-delivered multiple health behavior change intervention (CanChange) on health and behavioral outcomes in survivors of colorectal cancer: A randomized controlled trial. Journal of Clinical Oncology, 31, 2313–2321. doi:10.1200/JCO.2012.45.5873
Hawkes, A.L., Gollschewski, S., Lynch, B.M., & Chambers, S. (2009). A telephone-delivered lifestyle intervention for colorectal cancer survivors “CanChange”: A pilot study. Psycho-Oncology, 18, 449–455. doi:10.1002/pon.1527
Ho, J.W.C., Lee, A.M., Macfarlane, D.J., Fong, D.Y.T., Leung, S., Cerin, E., . . . Cheng, K. (2013). Study protocol for “Moving Bright, Eating Smart”—A phase 2 clinical trial on the acceptability and feasibility of a diet and physical activity intervention to prevent recurrence in colorectal cancer survivors. BMC Public Health, 13, 487. doi:10.1186/1471-2458-13-487
Husebø, A.M., Dyrstad, S.M., Søreide, J.A., & Bru, E. (2013). Predicting exercise adherence in cancer patients and survivors: A systematic review and meta-analysis of motivational and behavioural factors. Journal of Clinical Nursing, 22(1–2), 4–21. doi:10.1111/j.1365-2702.2012.04322.x
Jones, B.L., Nagin, D.S., & Roeder, K. (2001). A SAS procedure based on mixture models for estimating developmental trajectories. Sociological Methods and Research, 29, 374–393.
Jones, B.L., Sinclair, R.C., Rhodes, R.E., & Courneya, K.S. (2004). Promoting exercise behaviour: An integration of persuasion theories and the Theory of Planned Behaviour. British Journal of Health Psychology, 9(Pt. 4), 505–521. doi:10.1348/1359107042304605
Kahneman, D., & Tversky, A. (1979). Prospect theory: An analysis of decision under risk. Econometrica, 47, 263–292.
Latimer, A.E., Brawley, L.R., & Bassett, R.L. (2010). A systematic review of three approaches for constructing physical activity messages: What messages work and what improvements are needed? International Journal of Behavioral Nutrition and Physical Activity, 7, 36. doi:10.1186/1479-5868-7-36
Latimer, A.E., Rench, T.A., Rivers, S.E., Katulak, N.A., Materese, S.A., Cadmus, L., . . . Salovey, P. (2008). Promoting participation in physical activity using framed messages: An application of prospect theory. British Journal of Health Psychology, 13(Pt. 4), 659–681. doi:10.1348/135910707X246186
Lee, D.H., Kim, J.Y., Lee, M.K., Lee, C., Min, J.H., Jeong, D.H., . . . Jeon, J.Y. (2013). Effects of a 12-week home-based exercise program on the level of physical activity, insulin, and cytokines in colorectal cancer survivors. Supportive Care in Cancer, 21, 2537–2545.
Ligibel, J.A., Meyerhardt, J., Pierce, J.P., Najita, J., Shockro, L., Campbell, N., . . . Shapiro, C. (2011). Impact of a telephone-based physical activity intervention upon exercise behaviors and fitness in cancer survivors enrolled in a cooperative group setting. Breast Cancer Research and Treatment, 132, 205–213.
Lowe, S.S., Watanabe, S.M., Baracos, V.E., & Courneya, K.S. (2012). Determinants of physical activity in palliative cancer patients: An application of the Theory of Planned Behavior. Journal of Supportive Oncology, 10, 30–36. doi:10.1016/j.suponc.2011.07.005
McCall, L.A., & Martin Ginis, K.A. (2004). The effects of message framing on exercise adherence and health beliefs among patients in a cardiac rehabilitation program. Journal of Applied Biobehavioral Research, 9, 122–135.
Meyerhardt, J.A., Giovannucci, E.L., Holmes, M.D., Chan, A.T., Colditz, G.A., & Fuchs, C.S. (2006). Physical activity and survival after colorectal cancer diagnosis. Journal of Clinical Oncology, 24, 3527–3534.
Meyerhardt, J.A., Giovannucci, E.L., Ogino, S., Kirkner, G.J., Chan, A.T., Willett, W., & Fuchs, C.S. (2009). Physical activity and male colorectal cancer survival. Archives of Internal Medicine, 169, 2102–2108. doi:10.1001/archinternmed.2009.412
Meyerhardt, J.A., Heseltine, D., Niedzwiecki, D., Hollis, D., Saltz, L.B., Mayer, R.J., . . . Fuchs, C.S. (2006). Impact of physical activity on cancer recurrence and survival in patients with stage III colon cancer: Findings from CALGB 89803. Journal of Clinical Oncology, 24, 3535–3541. doi:10.1200/JCO.2006.06.0863
Morey, M.C., Snyder, D.C., Sloane, R., Cohen, H.J., Peterson, B., Hartman, T.J., . . . Demark-Wahnefried, W. (2009). Effects of home-based diet and exercise on functional outcomes among older, overweight long-term cancer survivors: RENEW: A randomized controlled trial. JAMA, 301, 1883–1891. doi:10.1001/jama.2009.643
Packel, L.B., Prehn, A.W., Anderson, C.L., & Fisher, P.L. (2014). Factors influencing physical activity in colorectal cancer survivors. American Journal of Health Promotion, 30, 85–92.
Pinto, B.M., & Floyd, A. (2008). Theories underlying health promotion interventions among cancer survivors. Seminars in Oncology Nursing, 24, 153–163. doi:10.1016/j.soncn.2008.05.003
Pinto, B.M., Papandonatos, G.D., Goldstein, M.G., Marcus, B.H., & Farrell, N. (2013). Home-based physical activity intervention for colorectal cancer survivors. Psycho-Oncology, 22, 54–64.
Rock, C.L., Doyle, C., Demark-Wahnefried, W., Meyerhardt, J., Courneya, K.S., Schwartz, A.L., . . . Gansler, T. (2012). Nutrition and physical activity guidelines for cancer survivors. CA: A Cancer Journal for Clinicians, 62, 242–274.
Schmid, D., & Leitzmann, M.F. (2014). Association between physical activity and mortality among breast cancer and colorectal cancer survivors: A systematic review and meta-analysis. Annals of Oncology, 25, 1293–1311. doi:10.1093/annonc/mdu012
Schmitz, K.H., Courneya, K.S., Matthews, C., Demark-Wahnefried, W., Galvao, D.A., Pinto, B.M., . . . Schwartz, A.L. (2010). American College of Sports Medicine roundtable on exercise guidelines for cancer survivors. Medicine and Science in Sports and Exercise, 42, 1409–1426. doi:10.1249/MSS.0b013e3181e0c112
Short, C.E., James, E.L., Girgis, A., D’Souza, M.I., & Plotnikoff, R.C. (2014). Main outcomes of the Move More for Life Trial: A randomised controlled trial examining the effects of tailored-print and targeted-print materials for promoting physical activity among post-treatment breast cancer survivors. Psycho-Oncology, 24, 771–778. doi:10.1002/pon.3639
Smith, S.L., Singh-Carlson, S., Downie, L., Payeur, N., & Wai, E.S. (2011). Survivors of breast cancer: Patient perspectives on survivorship care planning. Journal of Cancer Survivorship: Research and Practice, 5, 337–344. doi:10.1007/s11764-011-0185-7
Speed-Andrews, A.E., McGowan, E.L., Rhodes, R.E., Blanchard, C.M., Culos-Reed, S.N., Friedenreich, C.M., & Courneya, K.S. (2013). Correlates of strength exercise in colorectal cancer survivors. American Journal of Health Behavior, 37, 162–170.
Spence, R.R., Heesch, K.C., Eakin, E.G., & Brown, W.J. (2007). Randomised controlled trial of a supervised exercise rehabilitation program for colorectal cancer survivors immediately after chemotherapy: Study protocol. BMC Cancer, 7, 154.
Stull, V.B., Snyder, D.C., & Demark-Wahnefried, W. (2007). Lifestyle interventions in cancer survivors: Designing programs that meet the needs of this vulnerable and growing population. Journal of Nutrition, 137(1, Suppl.), 243S–248S.
Vallance, J.K., Courneya, K.S., Plotnikoff, R.C., Yasui, Y., & Mackey, J.R. (2007). Randomized controlled trial of the effects of print materials and step pedometers on physical activity and quality of life in breast cancer survivors. Journal of Clinical Oncology, 25, 2352–2359. doi:10.1200/JCO.2006.07.9988
Wakker, P.P. (2010). Prospect theory. Cambridge, MA: Cambridge University Press.
About the Author(s)
Hirschey is a PhD candidate and Lipkus is a professor, both in the School of Nursing at Duke University in Durham, NC; Jones is an exercise scientist at Memorial Sloan Kettering Cancer Center in New York, NY; Mantyh is chief of Colorectal Surgery and Sloane is a senior biostatistician, both at Duke University Medical Center in Durham; and Demark-Wahnefried is a professor in the School of Health Professions at the University of Alabama in Birmingham. This project was supported by a grant (CA125458) from the National Institutes of Health/National Cancer Institute. Hirschey, Lipkus, Jones, Mantyh, and Demark-Wahnefried contributed to the conceptualization and design. Lipkus, Mantyh, and Demark-Wahnefried completed the data collection. Sloane provided the statistical support. Sloane and Hirschey provided the analysis. Hirschey contributed to the manuscript preparation. Hirschey can be reached at rachel.hirschey@duke.edu, with copy to editor at ONFEditor@ons.org. Submitted September 2015. Accepted for publication February 26, 2016.

