Vaginal Testosterone for Management of Aromatase Inhibitor–Related Sexual Dysfunction: An Integrative Review
Problem Identification: Women taking aromatase inhibitors (AIs) as part of the management of hormone receptor–positive breast cancer experience more symptoms of sexual dysfunction, including vaginal atrophy, as opposed to postmenopausal women and women treated with tamoxifen (Nolvadex®). Vaginal testosterone could be an alternative to estrogen, which is contraindicated in this population.
Literature Search: A systematic review was completed by searching PubMed and Scopus databases.
Data Evaluation: 64 search results were reduced to a final sample of 3 articles after applying inclusion and exclusion criteria.
Synthesis: Published results suggest that vaginally applied testosterone doses of 150 mcg and 300 mcg improve symptoms of sexual dysfunction in women taking AIs. Minimal side effects are observed, and estradiol levels are not affected by vaginally applied testosterone. Additional research is needed to evaluate vaginal testosterone in women taking AIs.
Conclusions: Vaginal testosterone shows preliminary promise as an option to manage sexual side effects of AI therapy in postmenopausal cancer survivors; however, available data are too limited to draw practice-changing conclusions.
Implications for Research: Large-scale randomized, controlled trials need to be completed to evaluate the efficacy and safety of vaginal testosterone in women taking AIs.
Jump to a section
Breast cancer is the most prevalent cancer among women in the United States; American women have a 12% lifetime risk of being diagnosed with breast cancer (American Cancer Society [ACS], 2015). ACS (2015) estimated that, in 2014, about 3.1 million women in the United States with a history of breast cancer were either cancer-free, had current evidence of disease, or were actively undergoing treatment. Of these 3.1 million cases, about 84% of breast cancers express the estrogen receptor and/or progesterone receptor, for which long-term systemic endocrine therapy may be indicated (ACS, 2015; Lester, Pahouja, Andersen, & Lustberg, 2015). Endocrine therapy usually consists of either a selective estrogen receptor modulator (SERM) in premenopausal women or an aromatase inhibitor (AI), used most frequently in postmenopausal women. Both classes of drugs suppress circulating estrogen and, therefore, have the potential to initiate or exacerbate symptoms of sexual dysfunction (Lester et al., 2015).
Background
The benefit of adjuvant endocrine therapy in hormone receptor–positive breast cancers is well established and continues to be the standard of care for this patient population (National Comprehensive Cancer Network [NCCN], 2016). In studies comparing tamoxifen (Nolvadex®) to letrozole (Femara®), AIs were superior to SERMs (Regan, Price, Giobbie-Hurder, Thurlimann, & Gelber, 2011). Specifically, letrozole improved disease-free survival, time to distant recurrence, and overall survival when compared to tamoxifen (Regan et al., 2011). Given the established benefit of AIs, their use is frequently part of the comprehensive disease management strategy for patients with breast cancer with hormone receptor–positive disease. In addition, AIs have demonstrated efficacy in primary prevention of breast cancer in postmenopausal women and, therefore, are increasingly prescribed for postmenopausal women identified as having an elevated risk of breast cancer (Cuzick et al., 2014).
Problem
The ability of AIs to inhibit endogenous estrogen synthesis results in low estrogen levels, which, in turn, affects sexual functioning (Kallak et al., 2015). Women treated with AIs often exhibit more severe symptoms of vaginal atrophy as compared to postmenopausal women and women treated with tamoxifen (Kallak et al., 2014). Kallak et al. (2014) found that the vaginal pH of women treated with AIs was higher than that of women treated with tamoxifen and that of postmenopausal controls on vaginal estrogen therapy. A cross-sectional survey found that 93% of women taking AIs for 18–24 months experienced sexual dysfunction, and 75% of these women reported distress as a result (Schover, Baum, Fuson, Brewster, & Melhem-Bertrandt, 2014). Although estrogen is standard therapy for vaginal atrophy and its related effects on sexual functioning, in AI-treated women, estrogen replacement is contraindicated (Kallak et al., 2015). As the number of women taking AIs continues to rise, practitioners should be knowledgeable of appropriate strategies and therapies to manage the side effects of these medications, specifically sexual dysfunction.
Intervention
An association between subcutaneous, intramuscular, transdermal, gel, and oral androgens and improved sexual functioning in women has been demonstrated through results from several large cross-sectional and longitudinal studies (Davis et al., 2008; Davis & Wahlin-Jacobsen, 2015; Somboonporn, Davis, Seif, & Bell, 2005). Studies have suggested that androgens may have a direct effect on vaginal function, as androgen receptors have been identified in the vaginal epithelium (Krapf, Belkin, Dreher, & Goldstein, 2015). However, research exploring the efficacy and safety of vaginal testosterone to treat sexual dysfunction, including vaginal atrophy, in women with breast cancer treated with AIs is limited. A literature review yielded few studies examining the efficacy and safety of vaginal testosterone for women taking AIs. The aim of this article is to present an integrative examination of research on vaginal testosterone’s role in managing sexual dysfunction in women taking AIs with a history of breast cancer. Only two pilot studies explored the role of vaginal testosterone in managing various domains of sexual functioning in women with a history of breast cancer taking AIs, both of which demonstrated improvement in sexual functioning with vaginal testosterone application (Dahir & Travers-Gustafson, 2014; Witherby et al., 2011). Because of the paucity of data available regarding vaginal testosterone in women taking AIs, to comprehensively review the current data, this article also examines current evidence of vaginal testosterone use for managing sexual dysfunction in patients with breast cancer on AIs, as well as those not on AIs.
Methods
The electronic databases of PubMed and Scopus were searched using the following keywords: aromatase inhibitor, breast cancer, cancer, vaginal testosterone, vaginal atrophy, sexual dysfunction, and female sexual functioning. Sixty-four studies with human participants, published from January 2007 to February 2016, were identified and reviewed for vaginal testosterone as an intervention for sexual dysfunction (see Figure 1). The identified studies were further examined for those relevant to women treated with AIs. Only two studies specifically addressed outcomes for women on AIs, so to comprehensively review the relevant data on vaginal testosterone, articles that focused on the use of testosterone in women with breast cancer not on AIs were also included. Three articles were identified for this integrative review: one randomized, controlled trial (RCT) and two prospective cohort studies (see Table 1).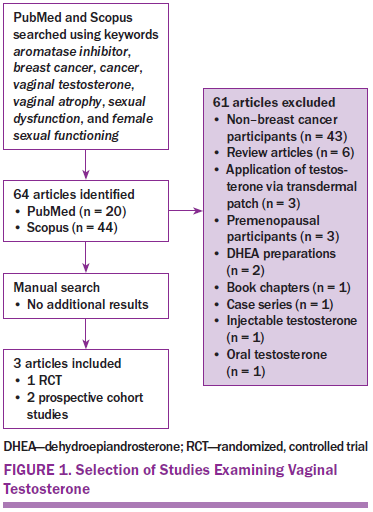
Results
Efficacy
Women taking aromatase inhibitors: Witherby et al. (2011) and Dahir and Travers-Gustafson (2014) have explored the role of vaginal testosterone in patients concurrently taking AIs, yielding similarly significant results. Investigators completed four-week prospective cohort pilot studies evaluating the efficacy of vaginal testosterone (Dahir & Travers-Gustafson, 2014; Witherby et al., 2011). Each study investigated different domains, with Witherby et al. (2011) observing symptom scores related to vaginal atrophy, pH, maturation values, and estradiol levels, and Dahir and Travers-Gustafson (2014) using the Female Sexual Function Index (FSFI) to measure sexual dysfunction. The FSFI is a 19-item scale that assesses six domains of sexual functioning; it has been shown to be an appropriate tool to screen for sexual dysfunction in women with breast cancer (Bartula & Sherman, 2015). After completing four weeks of vaginal testosterone (150 mcg versus 300 mcg dosing), participants (N = 20) in both arms of Witherby et al.’s (2011) pilot study reported a significant decrease in symptom scores (p < 0.001), which lasted for one month post-treatment. Dahir and Travers-Gustafson’s (2014) study of 12 participants garnered similar results; compared to baseline, a statistically significant increase was noted in total domain scores (p = 0.00) after four weeks of treatment with 300 mcg of testosterone. Specifically, domains of the FSFI reflected improved desire (p = 0.00), arousal (p = 0.002), lubrication (p = 0.018), orgasm (p = 0.005), satisfaction (p = 0.001), and pain (p = 0.00). Witherby et al. (2011) and Dahir and Travers-Gustafson (2014) postulate that aromatization to oestradiol is not necessary to provide symptomatic relief.
[[{"type":"media","view_mode":"media_original","fid":"32066","attributes":{"alt":"","class":"media-image","height":"518","typeof":"foaf:Image","width":"757"}}]]
Women with breast cancer: Barton et al. (2007) explored the use of testosterone in patients with cancer, though not specifically those on AIs. Barton et al. (2007) examined libido in postmenopausal cancer survivors (N = 131) who used transdermal testosterone for a duration of four weeks. This phase III, randomized, placebo-controlled crossover clinical trial compared a daily dose of transdermal testosterone (10 mg) to placebo. Testosterone cream was applied to the abdomen or thigh area rather than vaginally (Barton et al., 2007). Women on AIs and SERMs were included in this study, however stratification of women on SERMs was reported; women on AIs were not a reported subset within treatment groups (Barton et al., 2007). Libido was measured using the Changes in Sexual Functioning Questionnaire, which includes the following domains: desire, interest, and frequency (Barton et al., 2007). No statistically significant differences were observed between groups (p = 0.71) (Barton et al., 2007).
Side Effects
In studies that used testosterone without estrogen, which includes Witherby et al. (2011), Dahir and Travers-Gustafson (2014), and Barton et al. (2007), minimal side effects were identified. Witherby et al. (2011) reported a toxicity profile that included abnormal hair growth, acne, vaginal rash, pruritus, and moderate symptoms of vaginal discharge. In Witherby et al.’s (2011) cohort, symptoms that improved while taking vaginal testosterone included acne, depression, and vaginal and thigh burning. No side effects led to discontinuation in any study, and 91% of participants in Dahir and Travers-Gustafson’s (2014) study continued to use the vaginal testosterone after study completion, indicating a well-tolerated toxicity profile (Dahir & Travers-Gustafson, 2014; Witherby et al., 2011). Barton et al. (2007) reported no untoward side effects associated with the use of testosterone. Although these studies show promise regarding the safety profile, they lack the adequate number of participants to support definitive statements about safety.
Ideal Dose
Witherby et al. (2011) and Dahir and Travers-Gustafson (2014) used a dose of 300 mcg testosterone applied to the vagina daily, which led to a significant improvement in symptoms. Witherby et al. (2011) also tested a dose of 150 mcg in half of the cohort (n = 10), which also yielded a significant improvement in symptoms (p < 0.001). Witherby et al. (2011) detected no significant difference in clinical symptom scores between the two cohorts, although the 300 mcg dose caused pathologic changes in the post-treatment maturation. Barton et al. (2007) found no significant effect (p = 0.42) on sexual functioning using a compound cream containing a daily dose of testosterone (10 mg dose applied transdermally).
Systemic Hormone Levels
A concern with the use of testosterone to manage sexual dysfunction in patients with breast cancer is the potential risk of increasing serum estradiol levels. Two studies included in this integrative review assessed and reported estradiol levels; neither showed an increase in serum estradiol after using testosterone alone (Barton et al., 2007; Witherby et al., 2011). Witherby et al. (2011) assessed pre- and post-treatment serum estradiol levels illustrating that serum estradiol levels continued to be low post-treatment; 18 were undetectable, one was 7 picograms per ml, and one was 7.6 picograms per ml (Witherby et al., 2011). Similarly, Barton et al. (2007) reported no statistical differences (p = 0.82) in serum estradiol between placebo and treatment groups; although serum estradiol levels decreased with testosterone use, no statistically significant difference was seen (Barton et al., 2007).
Discussion
The prevalence of sexual dysfunction among women taking AIs is significant; this side effect has been cited as a major reason for nonadherence (Kyvernitakis et al., 2014). Given the well-established efficacy of AIs regarding decreasing breast cancer risk, clinicians and patients should be well equipped with strategies to minimize side effects that may contribute to nonadherence. Witherby et al. (2011) and Dahir and Travers-Gustafson (2014) completed appropriately designed pilot studies that yielded promising results, suggesting the effectiveness of vaginal testosterone. Vaginal testosterone has emerged as a potential strategy to manage AI-associated sexual dysfunction. When examining the evidence, except for Barton et al.’s (2007) RCT, which used transdermal and not vaginal testosterone, current literature suggests testosterone may have a role in the management of sexual dysfunction that results from systemic endocrine therapy.
The application of testosterone to manage sexual dysfunction is not novel; subcutaneous, intramuscular, and transdermal preparations with and without estrogen have been therapies for hypoactive sexual disorder in menopausal women. Fernandes, Costa-Paiva, and Pinto-Neto (2014) compared the effect of vaginal estrogen, testosterone, polyacrylic acid, or oil lubricant alone on postmenopausal sexual dysfunction in an RCT in which 20 patients received 300 mcg of testosterone, vaginally, for 12 weeks. Women using 12 weeks of vaginal testosterone showed an improvement in all domains of the FSFI (Fernandes et al., 2014). Practice guidelines for the use of testosterone in women were published by the Endocrine Society (Wierman et al., 2014). However, application of these guidelines in women with a history of hormone receptor–positive breast cancer and, even more specifically, those taking AIs, has yet to be established. Witherby et al. (2011) and Dahir and Travers-Gustafson (2014) present compelling findings, but for a consensus to be reached, additional studies need to be conducted.
Although the risk–benefit analysis should encourage further research of the use of vaginal testosterone in the management of sexual dysfunction, clinicians and researchers may remain apprehensive about the potential of rising estradiol levels. Two reviewed studies measured serum estradiol after testosterone use. Witherby et al. (2011) and Barton at al. (2007), who used testosterone alone, found no concerning elevation in estradiol levels post-testosterone use. These findings support the safety profile of vaginal testosterone for patients currently taking AIs, given that the goal of this treatment is to suppress estrogen. However, further research is necessary regarding the pharmacokinetics of vaginal testosterone and to determine ideal levels of serum testosterone in females.
Although a consensus on the best management options for sexual dysfunction related to AI use continues to be debated, what has been established is the importance of assessing, at regular intervals, for symptoms of sexual dysfunction (NCCN, 2015). One study included in this integrative review used the FSFI to measure pre- and postintervention measures of sexual function. The FSFI has been validated in patients with breast cancer and all female cancer survivors as a reliable instrument for measuring sexual dysfunction (Bartula & Sherman, 2015; Baser, Li, & Carter, 2012). The 19-item tool assesses six different domains of sexual functioning: desire, arousal, lubrication, orgasm, satisfaction, and pain; it does not require any additional training to administer or score, making it user friendly for a variety of healthcare professionals (Bartula & Sherman, 2015). The total FSFI score is the sum of scores given to each of the six domains; the maximum score is 36, and high scores indicate better functioning (Baser et al., 2012). The FSFI has drawbacks; it can take 15–20 minutes to complete and fails to incorporate factors unique to cancer populations that may be affecting sexual desire, such as physical and mental impact of disease (Baser et al., 2012). Despite these drawbacks, the FSFI is a validated instrument for use in this unique population, to provide useful information as an objective measurement of a domain which is anything but objective. In addition, the use of this instrument may facilitate further research for baseline and measurement of subsequent interventions designed to improve sexual function. Assessment of sexual functioning is a necessary and important first step in developing strategies to manage related symptoms.
Limitations to this review include the limited number of articles and the limited number of participants in the reviewed articles; both preclude practice-changing conclusions being drawn. Of note, Barton et al. (2007) used a transdermal route of administration as opposed to vaginal; however, the study did include patients with breast cancer and, therefore, adds to this body of knowledge. Because the studies reviewed used compounded creams of testosterone, which are not currently approved by the U.S. Food and Drug Administration (FDA) for use in women, pharmacokinetic data are not available, precluding evaluation of the differences in absorption between the vaginal and transdermal route. Despite limitations, the evidence is promising and should prompt additional investigative efforts by nurse scientists.
As the field of survivorship continues to expand, more institutions are beginning to include sexuality and intimacy as key constructs of survivorship care (Krychman & Katz, 2012). This integrative review seeks to explore an established survivorship problem that should have clear and effective guidelines for symptom management. Evidence presented suggests that vaginal testosterone may be a potential option for improving symptoms related to sexual dysfunction, although inadequate data regarding safety and pharmacokinetics impede clinical application of vaginal testosterone.
Implications for Nursing
RNs and advanced practice RNs (APRNs) are skilled at identifying and managing treatment side effects in the oncology population. Sexual dysfunction can be perceived as a sensitive topic by patients and clinicians. RNs and APRNs, whose training is grounded in holistic care, can often assess and treat a multitude of different symptoms, including sexual dysfunction. Options are currently limited for the treatment of sexual dysfunction in women taking AIs; therefore, further research efforts are needed to identify and develop strategies to manage this side effect.
Vaginal testosterone shows early promise as an alternative to other hormonal agents, such as estrogen, which has historically been used to manage sexual dysfunction outside of the oncology population. Further research of this intervention is needed in patients with breast cancer taking systemic endocrine therapy, such as AIs. Based on findings from Witherby et al. (2011) and Dahir and Travers-Gustafson (2014), either the 150 mcg or 300 mcg vaginal daily dose would be an acceptable dose to examine comparatively in future research investigations.
Conclusion
Sexual dysfunction is a commonly reported side effect of AIs. The number of breast cancer survivors continues to increase, and the role of chemoprevention using AIs continues to expand. Practitioners need to be equipped with strategies to manage sexual dysfunction to minimize the number of women stopping therapy as a result of untoward side effects and improve women’s quality of life. Vaginal testosterone for the management of sexual dysfunction in women taking AIs is a non–FDA-approved therapy that has demonstrated efficacy and safety in small studies. These small studies are limited and lack adequate data to support any practice change. Large-scale studies are needed to validate these early findings.
[[{"type":"media","view_mode":"media_original","fid":"32071","attributes":{"alt":"","class":"media-image","height":"478","typeof":"foaf:Image","width":"365"}}]]
About the Author(s)
Lemke is an advanced practice RN fellow in the Department of Nursing at the University of Texas MD Anderson Cancer Center; Madsen is an assistant professor in the School of Nursing at the University of Texas Health Science Center; and Dains is an associate professor in the Department of Nursing at the University of Texas MD Anderson Cancer Center, all in Houston. No financial relationships to disclose. Mention of specific products and opinions related to those products do not indicate or imply endorsement by the Oncology Nursing Society. Lemke completed the data collection. All authors contributed to the conceptualization and design, analysis, and manuscript preparation. Lemke can be reached at ealemke@mdanderson.org, with copy to editor at ONFEditor@ons.org. Submitted June 2016. Accepted for publication August 15, 2016.
References
American Cancer Society. (2015). Breast cancer facts and figures, 2015–2016. Retrieved from https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and…
Barton, D.L., Wender, D.B., Sloan, J.A., Dalton, R.J., Balcueva, E.P., Atherton, P.J., . . . Loprinzi, C.L. (2007). Randomized controlled trial to evaluate transdermal testosterone in female cancer survivors with decreased libido; North Central Cancer Treatment Group protocol N02C3. Journal of the National Cancer Institute, 99, 672–679. doi:10.1093/jnci/djk149
Bartula, I., & Sherman, K.A. (2015). The Female Sexual Functioning Index (FSFI): Evaluation of acceptability, reliability, and validity in women with breast cancer. Supportive Care in Cancer, 23, 2633–2641. doi:10.1007/s00520-015-2623-y
Baser, R.E., Li, Y., & Carter, J. (2012). Psychometric validation of the Female Sexual Function Index (FSFI) in cancer survivors. Cancer, 118, 4606–4618. doi:10.1002/cncr.26739
Cuzick, J., Sestak, I., Forbes, J.F., Dowsett, M., Knox, J., Cawthorn, S., . . . Howell, A. (2014). Anastrozole for prevention of breast cancer in high-risk postmenopausal women (IBIS-II): An international, double-blind, randomised placebo-controlled trial. Lancet, 383, 1041–1048. doi:10.1016/s0140-6736(13)62292-8
Dahir, M., & Travers-Gustafson, D. (2014). Breast cancer, aromatase inhibitor therapy, and sexual functioning: A pilot study of the effects of vaginal testosterone therapy. Sexual Medicine, 2, 8–15. doi:10.1002/sm2.22
Davis, S.R., Moreau, M., Kroll, R., Bouchard, C., Panay, N., Gass, M., . . . Studd, J. (2008). Testosterone for low libido in postmenopausal women not taking estrogen. New England Journal of Medicine, 359, 2005–2017. doi:10.1056/NEJMoa0707302
Davis, S.R., & Wahlin-Jacobsen, S. (2015). Testosterone in women—The clinical significance. Lancet: Diabetes and Endocrinology, 3, 980–992. doi:10.1016/s2213-8587(15)00284-3
Fernandes, T., Costa-Paiva, L.H., & Pinto-Neto, A.M. (2014). Efficacy of vaginally applied estrogen, testosterone, or polyacrylic acid on sexual function in postmenopausal women: A randomized controlled trial. Journal of Sexual Medicine, 11, 1262–1270. doi:10.1111/jsm.12473
Kallak, T.K., Baumgart, J., Göransson, E., Nilsson, K., Poromaa, I.S., & Stavreus-Evers, A. (2014). Aromatase inhibitors affect vaginal proliferation and steroid hormone receptors. Menopause, 21, 383–390. doi:10.1097/GME.0b013e31829e41df
Kallak, T.K., Baumgart, J., Nilsson, K., Akerud, H., Poromaa, I.S., & Stavreus-Evers, A. (2015). Vaginal gene expression during treatment with aromatase inhibitors. Clinical Breast Cancer, 15, 527–535. doi:10.1016/j.clbc.2015.06.012
Krapf, J.M., Belkin, Z., Dreher, F., & Goldstein, A.T. (2015). Current and emerging treatment options for vulvovaginal atrophy. In A.M. Farage, W.K. Miller, N. Fugate Woods, & I.H. Maibach (Eds.), Skin, mucosa and menopause: Management of clinical issues (pp. 229–235). Berlin, Germany: Springer Berlin Heidelberg.
Krychman, M.L., & Katz, A. (2012). Breast cancer and sexuality: Multi-modal treatment options. Journal of Sexual Medicine, 9, 5–13. doi:10.1111/j.1743-6109.2011.02566.x
Kyvernitakis, I., Ziller, V., Hars, O., Bauer, M., Kalder, M., & Hadji, P. (2014). Prevalence of menopausal symptoms and their influence on adherence in women with breast cancer. Climacteric, 17, 252–259. doi:10.3109/13697137.2013.819327
Lester, J., Pahouja, G., Andersen, B., & Lustberg, M. (2015). Atrophic vaginitis in breast cancer survivors: A difficult survivorship issue. Journal of Personalized Medicine, 5, 50–66. doi:10.3390/jpm5020050
National Comprehensive Cancer Network. (2015). NCCN Clinical Guidelines in Oncology: Survivorship [v.2.2015]. Retrieved from http://www.nccn.org/professionals/physician_gls/pdf/survivorship.pdf
National Comprehensive Cancer Network. (2016). NCCN Clinical Practice Guidelines in Oncology: Breast cancer [v.1.2016]. Retrieved from https://www.nccn.org/professionals/physician_gls/pdf/breast.pdf
Regan, M.M., Price, K.N., Giobbie-Hurder, A., Thurlimann, B., & Gelber, R.D. (2011). Interpreting Breast International Group (BIG) 1–98: A randomized, double-blind, phase III trial comparing letrozole and tamoxifen as adjuvant endocrine therapy for postmenopausal women with hormone receptor-positive, early breast cancer. Breast Cancer Research, 13, 209. doi:10.1186/bcr2837
Schover, L.R., Baum, G.P., Fuson, L.A., Brewster, A., & Melhem-Bertrandt, A. (2014). Sexual problems during the first 2 years of adjuvant treatment with aromatase inhibitors. Journal of Sexual Medicine, 11, 3102–3111. doi:10.1111/jsm.12684
Somboonporn, W., Davis, S., Seif, M.W., & Bell, R. (2005). Testosterone for peri- and postmenopausal women. Cochrane Database of Systematic Reviews, 4, CD004509. doi:10.1002/14651858.CD004509.pub2
Wierman, M.E., Arlt, W., Basson, R., Davis, S.R., Miller, K.K., Murad, M.H., . . . Santoro, N. (2014). Androgen therapy in women: A reappraisal: An Endocrine Society clinical practice guideline. Journal of Clinical Endocrinology and Metabolism, 99, 3489–3510. doi:10.1210/jc.2014-2260
Witherby, S., Johnson, J., Demers, L., Mount, S., Littenberg, B., Maclean, C.D., . . . Muss, H. (2011). Topical testosterone for breast cancer patients with vaginal atrophy related to aromatase inhibitors: A phase I/II study. Oncologist, 16, 424–431. doi:10.1634/theoncologist.2010-0435

