Impact of Survivorship Care on Young Adult Survivors of Childhood Cancer With Post-Traumatic Stress Symptoms
Objectives: To assess post-traumatic stress symptoms (PTSSs) in young adult survivors of childhood cancer not receiving survivorship care and to determine whether attending a survivorship-focused healthcare visit was associated with changes in PTSSs.
Sample & Setting: 44 young adult survivors from the Yale Cancer Center in Connecticut without prior survivorship clinic attendance.
Methods & Variables: As part of a larger trial, participants were randomized to a model of survivorship-focused health care. The University of California at Los Angeles Post-Traumatic Stress Disorder (PTSD) Reaction Index assessed PTSS severity and frequency before and after the visit.
Results: At baseline, almost half of the participants were classified as partial PTSD likely or PTSD likely. Many met criteria for elevated levels of individual symptoms, particularly avoidance or numbing. At follow-up, PTSSs did not differ significantly from baseline.
Implications for Nursing: Survivorship care should be encouraged by nurses in healthcare settings that do not specialize in caring for long-term survivors. Nurses should facilitate screening for PTSSs and promote interventions among survivors completing cancer therapy to help them transition to survivorship care.
Jump to a section
Survivors of childhood cancer may be psychologically affected by the cancer experience throughout life (Brinkman, Recklitis, Michel, Grootenhuis, & Klosky, 2018). Potential outcomes they are at risk for include post-traumatic stress symptoms (PTSSs), such as avoidance or numbing (e.g., avoiding event-related stimuli), hyperarousal (e.g., hypervigilance), and re-experiencing (e.g., intrusive memories) (American Psychiatric Association [APA], 2013). Even a patient who does not meet diagnostic criteria for full-blown post-traumatic stress disorder (PTSD) can exhibit PTSSs, which can impair daily functioning. Although PTSSs have been identified in childhood cancer survivors (Koutná, Jelínek, Blatný, & Kepák, 2017; Stuber et al., 2010), PTSSs have not been examined specifically in young adult survivors who have not received survivorship care since completing treatment. PTSSs may stem from the diagnosis of a life-threatening illness, which is recognized as a potentially traumatic event (APA, 2013), and ongoing exposures to other potential traumas (e.g., surgeries, hospitalizations, deaths of other children) inherent in the cancer experience (Allen et al., 2018; Stuber, Kazak, Meeske, & Barakat, 1998). The emergence of such symptoms aligns with a pediatric medical traumatic stress model, which proposes that the experience of medical illness can cause physiologic or psychological effects (Price, Kassam-Adams, Alderfer, Christofferson, & Kazak, 2016).
Background
PTSSs are associated with other psychological outcomes (e.g., depression), impaired social functioning, poor quality of life, and long-term physical health decline (Ahmadi et al., 2018; Boscarino, 2004; Kroenke, Spitzer, Williams, Monahan, & Löwe, 2007; Meeske, Ruccione, Globe, & Stuber, 2001). Among individuals with medical-related trauma (e.g., intensive care stay), PTSSs have also been associated with inadequate medical care because of avoidance of experiences related to the trauma (French-Rosas, Moye, & Naik, 2011; Jones, 2010; Osterman, Hopper, Heran, Keane, & van der Kolk, 2001; Shemesh et al., 2001).
Compared to younger survivors, young adults (aged 18–40 years) can be more aware of the long-term consequences of cancer (Arnett, 2000; Kazak et al., 2004; Oeffinger et al., 2006). They are at a life stage involving new responsibilities and challenges, including greater independence from parents (Nathan et al., 2009) and higher mobility (Oeffinger & Wallace, 2006), creating increased socioemotional vulnerability (Arnett, 2000). Young adults are reaching new developmental milestones, such as starting a family (Docherty, Kayle, Maslow, & Santacroce, 2015), that may be directly affected by their cancer and late complications (e.g., infertility), forcing them to reflect on the cancer experience (Vannatta & Gerhardt, 2003). PTSSs, along with other deterrents including lack of time, interest, or knowledge (Berg, Stratton, Esiashvili, Mertens, & Vanderpool, 2016; Frederick, Bober, Berwick, Tower, & Kenney, 2017), can prevent young adults from receiving appropriate health care.
Post-Traumatic Stress Symptoms in Survivors
PTSSs have been identified in youth with other chronic diseases, including 30% of pediatric solid organ transplantation recipients (Supelana et al., 2016) and 32% of children and adolescents with chronic pain (Noel et al., 2016). Estimates of PTSSs in survivors of childhood cancer range greatly from 5% (Kazak et al., 2004) to 75% (Price et al., 2016). However, many studies have been conducted in the survivorship clinic setting or among patients who may have attended a survivorship clinic (Bruce, 2006; Lee & Santacroce, 2007). Consequently, inherent in past studies is a selection bias toward patients already engaged in survivorship care, which is only about 20% of childhood cancer survivors (Nathan et al., 2008). Research is needed that explicitly focuses on the remaining 80% of childhood cancer survivors who are not receiving survivorship care. These survivors are less likely to have received recommended psychological screening (Kazak et al., 2015) and may be particularly vulnerable to unrecognized and untreated PTSSs. Studies are needed to characterize this large segment of the survivor population. Patients who do not receive survivorship care may experience fewer PTSSs and, consequently, have less motivation to seek survivorship care, or PTSSs may remind patients of the cancer experience, including the healthcare setting, leading them to avoid such care (Santacroce & Lee, 2006; Vuotto, Procidano, & Annunziato, 2015). Inadequate psychological screening among childhood cancer survivors who have not received survivorship care may lead to an underestimation of the prevalence of PTSSs in this population. It is also unknown whether attending a survivorship visit is associated with an increase or decrease in PTSSs.
The burden of PTSSs among young adult survivors of childhood cancer not receiving recommended survivorship care is largely unknown. As part of a larger randomized trial comparing two models of survivorship-focused health care, the current study sought to (a) describe the severity and frequency of PTSSs in a sample of 44 young adult survivors who had not previously attended a survivorship clinic, (b) examine the association between treatment intensity and PTSS severity, and (c) determine whether attending a survivorship-focused healthcare visit was associated with changes in PTSSs. An additional exploratory aim was to observe whether changes in PTSSs varied by randomization group.
Methods
Design
This study was part of a larger randomized trial with a sample of 96 childhood cancer survivors. The primary objective of the larger trial was to compare the impact of two models of survivorship-focused health care on quality of care. A full description of the larger trial’s design and results related to the primary objective are reported elsewhere (Kadan-Lottick et al., 2018). A secondary aim of the larger randomized trial was to assess the psychological impact of receiving survivorship care; the current study addresses a research question under the scope of the secondary aim. The analysis of PTSSs was restricted to 44 young adults aged 18 years or older who completed all study assessments by self-report.
The trial was conducted at Yale University in New Haven, Connecticut, from May 2011 to November 2015 (ClinicalTrials.gov identifier: NCT02816866). Approval from the Yale Human Investigation Committee was obtained prior to initiation of survivor contact and data collection. Written informed consent was obtained from each participant before enrollment.
Participants
Participants were recruited from the Yale-New Haven Hospital Tumor Registry. Eligibility criteria were as follows:
• Being diagnosed with childhood cancer from January 1, 2000, to December 31, 2012, when aged younger than 18 years
• Completing cancer treatment one year or more before enrollment
• Being cancer-free and residing within 100 miles of Yale University
• Being fluent in English or Spanish
Patients who had already attended a survivorship clinic were excluded. Routine clinical practice at Yale University is to encourage all patients who have completed therapy to attend a survivorship clinic. Despite efforts, poor adherence to recommended follow-up has been described at Yale University (Zheng, Sint, Mitchell, & Kadan-Lottick, 2016) and elsewhere (Barakat, Schwartz, Szabo, Hussey, & Bunin, 2012).
Randomization
Of the 748 patients assessed for eligibility, 265 did not meet inclusion criteria, 253 refused to participate, 124 could not be contacted, and 106 enrolled. Ten withdrew before randomization (see Figure 1). From May 2011 to November 2013, 96 participants were randomized; one relapsed and one withdrew after randomization. Of the 94 participants in the larger randomized trial, 44 were 18 years or older at baseline; the current analysis was limited to this subsample. 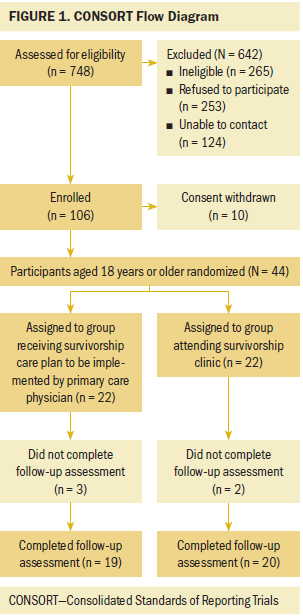
Procedures
Participants completed assessments of demographic characteristics and PTSSs prior to randomization and postintervention (12 months after randomization). The follow-up assessment included limited questions about reasons for not attending a survivorship clinic prior to the study. Under the larger trial, participants were randomized to one of two models of survivorship-focused health care: (a) a survivorship care plan to be implemented by their primary care physician (PCP) or (b) a survivorship clinic. Hospital records were reviewed by medical abstractors for each patient’s cancer diagnosis, treatment exposures, and known late complications of cancer treatment. Study staff created treatment summaries (survivorship care plans), including screening recommendations for late complications based on the Children’s Oncology Group (2013) Long-Term Follow-Up Guidelines for Survivors of Childhood, Adolescent, and Young Adult Cancers (version 4.0).
After randomization, participants in both groups were told to schedule and attend a survivorship-focused visit within the next 12 months; this visit would be either with their PCP or at the pediatric survivorship clinic at Yale University, per randomization. Participants randomized to receive a survivorship care plan to be implemented by their PCP were mailed a plan at baseline. All participants had a PCP. Participants in the survivorship clinic group were mailed a survivorship care plan following their clinic visit. Participants were considered compliant if they completed one survivorship-focused visit (with PCP or at survivorship clinic, per randomization) in the 12 months following randomization.
Measures
Post-traumatic stress symptoms: An adapted version of the 22-item University of California at Los Angeles PTSD Reaction Index (PTSD-RI) was used to assess PTS severity and frequency (Steinberg & Brymer, 2008). The PTSD-RI has demonstrated high internal consistency reliability (Cronbach alpha of 0.88–0.91 in the literature and 0.88 in the current study) (Steinberg et al., 2013), test-retest reliability (p = 0.84) (Steinberg, Brymer, Decker, & Pynoos, 2004), and convergent validity (p = 0.75) (Elhai et al., 2013; Steinberg et al., 2013). The PTSD-RI has been used in studies of young adult survivors of childhood cancer (Hobbie et al., 2000; Meeske et al., 2001; Rourke, Hobbie, Schwartz, & Kazak, 2007; Santacroce & Lee, 2006), and correlations with theoretically related variables have been in expected directions (e.g., avoidance symptoms positively correlated with nonadherence, compositive scores positively associated with functional/behavior problems) (Shemesh et al., 2000; Steinberg et al., 2013).
The PTSD-RI items reflect three symptom categories (avoidance or numbing, hyperarousal, re-experiencing). For the current study, each item was phrased to reference cancer (e.g., “I want to stay away from things . . . that make me think about the cancer”). Participants were asked to indicate the frequency with which they experienced each item on a five-point Likert-type scale ranging from 0 (none of the time) to 4 (most of the time). Responses were summed for a PTS severity composite score (range = 0–68), with higher scores indicating worse severity of PTSSs. Scores were classified as follows:
• 38 or greater: PTSD likely; screening suggests that formal diagnosis of PTSD by a clinician is likely.
• 20–37: partial PTSD likely; screening suggests that a diagnosis of PTSD is unlikely, but the patient reports many symptoms (Franklin, Piazza, Chelminski, & Zimmerman, 2015).
• 20 or less: PTSD unlikely
Previous analyses have shown that a score cutoff of 38 or greater has the greatest sensitivity and specificity for identifying PTSD (Rodriguez, Steinberg, Saltzman, & Pynoos, 2001). However, because individual symptoms can inform clinical impressions (Rourke et al., 2007), elevations in symptom categories were assessed by looking at the frequency of participants reporting the following (APA, 2000):
• Three or more avoiding or numbing symptoms (of a possible seven)
• Two or more hyperarousal symptoms (of a possible five)
• One or more re-experiencing symptoms (of a possible five)
In the current study, the PTSD-RI was a research tool administered outside of the healthcare setting and not intended for clinical evaluation; therefore, participants did not receive their scores. Participants received a telephone number for study staff to be used as a resource if they experienced psychological effects and so appropriate clinical intervention could be arranged.
Treatment intensity: Abstracted medical data were used to calculate treatment intensity using the Intensity of Treatment Rating Scale, version 2.0 (ITR-2.0) (Werba et al., 2007). The ITR-2.0 is a reliable (for inter-rater reliability, r = 0.87 and range = 0.63–1) and valid (for content validity, r = 0.95 and range = 0.71–0.91) method for characterizing intensity based on diagnosis, stage, risk, and treatment modality (e.g., radiation therapy) (Kazak et al., 2012). Treatment intensity scores range from 1 (least intensive) to 4 (most intensive).
Statistical Analysis
Primary outcomes were analyzed per the intent-to-treat approach, which in this study assumed that participants received the form of survivorship-focused health care to which they were randomized. Descriptive statistics (mean, standard deviation, frequency) were used to examine participant demographics, disease and treatment characteristics, and PTSSs. Analysis of variance tests were used to investigate the association between treatment intensity and PTSS severity. Student’s t tests and chi-square tests (or Fisher’s exact tests, as appropriate) were used to evaluate differences in participant characteristics between randomization groups and to examine changes in PTSS severity and frequency from baseline to follow-up. A two-tailed significance level of p < 0.05 was used in all analyses, and the authors had 80% power to detect a change of 4.4 points in the mean PTSD-RI composite score from baseline to follow-up. Analyses were conducted using SAS, version 9.4.
Results
Reasons for Survivorship Clinic Nonattendance
The 44 participants in this study were a mean age of 21.5 years (SD = 2.1) at baseline and 14.8 years (SD = 2.4) at diagnosis (see Table 1). Overall, 26 patients were women, and 42 were non-Hispanic White. Cancer diagnoses varied, with the most common being leukemia (n = 13, which includes diagnoses of acute lymphoblastic leukemia and acute myeloid leukemia) and lymphoma (n = 10). Treatment intensity varied, with 18 patients undergoing moderately intensive treatment and 16 undergoing very intensive treatment. There was no statistical difference in age at baseline, age at diagnosis, time since diagnosis, sex, race/ethnicity, insurance status, diagnosis, chemotherapy exposure, radiation exposure, or treatment intensity between the randomization groups. Twelve of 22 participants randomized to receive a survivorship care plan to be implemented by their PCP and 16 of 22 participants in the survivorship clinic group were compliant with the intervention (completed a survivorship-focused visit with PCP or at survivorship clinic, per randomization). 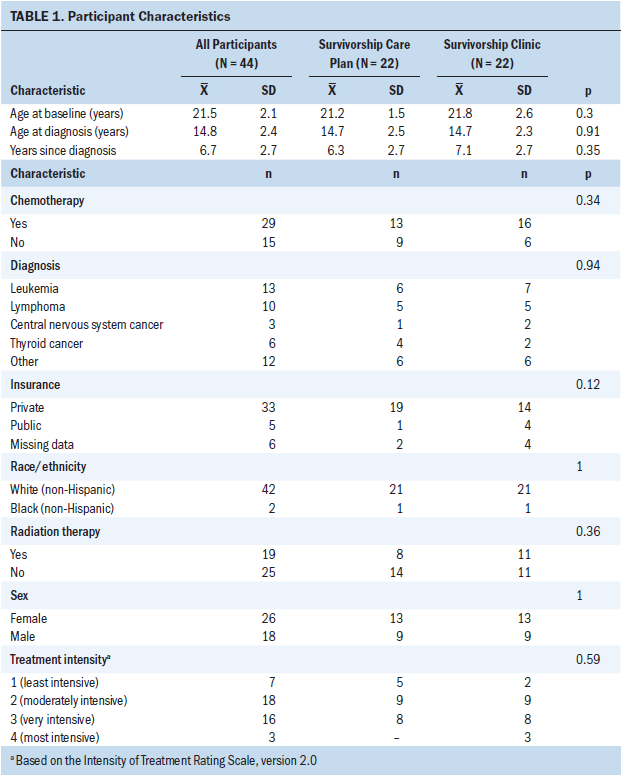
At follow-up, 20 of 39 participants endorsed at least one statement related to avoidance behavior: “I did not want to know potential complications of my past cancer” or “I wanted to put my cancer in the past and move on.” Fifteen of 39 participants endorsed at least one statement related to lack of interest or concern: “I did not think I was at increased risk for any late complications,” “I did not think I needed to know about possible future health problems,” or “I thought I was too young to have complications of my past cancer treatment.”
Post-Traumatic Symptom Frequency and Severity and Association With Treatment Intensity
At baseline, the mean PTSS severity composite score was 18.7 (SD = 10.1) (see Table 2). Based on their composite scores, 15 participants were classified as partial PTSD likely and 3 as PTSD likely. At follow-up, the mean PTSS severity composite score was 17.8 (SD = 12.5). Based on their composite scores, 15 participants were classified as partial PTSD likely and 2 as PTSD likely. In terms of symptom categories, at baseline, 21 participants met the criteria for elevations in the avoidance or numbing category, 17 for hyperarousal, and 16 for re-experiencing. At follow-up, 16 participants met the criteria for elevations in the avoidance or numbing category, 15 for hyperarousal, and 21 for re-experiencing. Intensity of treatment rating was not associated with PTSD-RI composite scores. 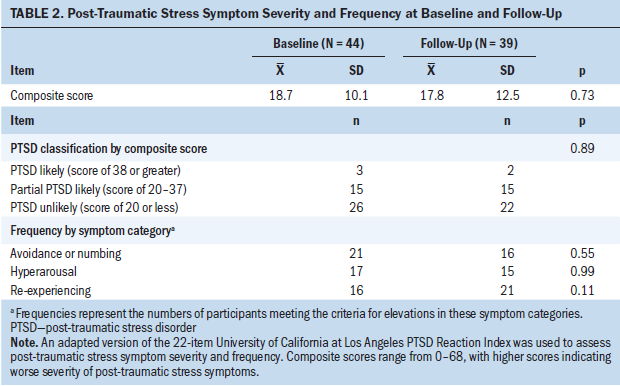
Post-Traumatic Symptoms Before and After Survivorship-Focused Healthcare Visit
No overall statistically significant change in mean PSTD-RI composite score or severity classification was detected between baseline and follow-up. The overall difference in symptom category frequency between baseline and follow-up was not statistically significant.
Survivorship Care Randomization
No statistically significant difference in PTSS severity at follow-up was detected between the randomization groups. This finding applied to the intent-to-treat analysis and when the analysis was restricted to participants deemed compliant with the intervention.
Discussion
The current study revealed considerable rates of PTSSs in a sample of young adults who had not previously attended a survivorship clinic. At baseline, almost half of the participants met the criteria for partial PTSD likely or PTSD likely. A high frequency of survivors also reported elevated avoidance or numbing, hyperarousal, and/or re-experiencing symptoms at baseline and follow-up. Results were similar following the participant’s survivorship-focused healthcare visit, suggesting that receiving survivorship care does not trigger or aggravate PTSSs and should be encouraged. However, it should be acknowledged that there was also no improvement in symptoms following the visit. The intensity of cancer treatment was not associated with PTSS severity, which is similar to findings from other studies (Bruce, 2006; Meeske et al., 2001), suggesting that the severity of the trauma did not relate to PTSSs.
In the childhood cancer survivor literature, rates of PTSSs vary by the assessment tool used and the population studied. Rates of participants in this study meeting the criteria for likely partial PTSD and likely PTSD are relatively consistent with those reported in previous studies of young adult survivors. Using the PTSD-RI, Hobbie et al. (2000) found that 24% of 78 young adult survivors met the criteria for likely partial PTSD and 8% for likely PTSD. Using the University of California at Los Angeles PTSD Index, an adaptation of the PTSD-RI, Lee and Santacroce (2007) found that 13% of 45 young adult survivors were classified as PTSD likely, and Rourke et al. (2007) observed that 22% of 182 young adult survivors met the criteria for likely partial PTSD or likely PTSD. The sample in a study by Schwartz and Drotar (2006) was most comparable to that of the current study in terms of age (range = 18–28 years). Using the Posttraumatic Stress Disorder Checklist–Civilian Version (Conybeare, Behar, Solomon, Newman, & Borkovec, 2012), Schwartz and Drotar (2006) determined that 18% of 57 young adult survivors met the criteria for PTSD.
In the current study, a large proportion of participants met the criteria for likely partial PTSD at baseline and follow-up. Evidence suggests that the prevalence of PTSSs meeting this subthreshold level of severity, where a formal diagnosis of PTSD by a clinician is unlikely but the patient reports many symptoms, may still have vast clinical implications. For example, in a study of 105 long-term adult Hodgkin lymphoma survivors by Varela, Ng, Mauch, and Recklitis (2013), 86% of survivors who met the criteria for partial PTSD reported experiencing some functional impairment because of their symptoms. It is important to note that the current sample was made up of survivors without prior attendance at a survivorship clinic, suggesting that PTSSs are present among survivors who have not sought survivorship care and, therefore, would not necessarily be regularly monitored for functional impairments related to their cancer experience. This finding suggests the existence of unmet psychosocial needs among survivors.
The current study also identified a high frequency of participants with elevations in individual symptom categories, notably avoidance or numbing, among these young adult childhood cancer survivors who had not previously attended a survivorship clinic. This suggests that a desire to avoid reminders of the cancer experience may be a contributor to the low rates (less than 20%) of survivorship care among childhood cancer survivors, particularly young adults (Nathan et al., 2008).
A better understanding of what keeps young adult survivors from obtaining survivorship care can help investigators design effective interventions to reach these survivors. Young adults face many barriers to receiving recommended care, including increasing independence from their parents (Nathan et al., 2009), high mobility (Oeffinger & Wallace, 2006), and the combination of lack of time, interest, guidance, and knowledge (Berg et al., 2016). However, fear and avoidance behaviors continue to be recognized in this population (Berg et al., 2016; Zeltzer et al., 2009), and these barriers are amenable to standard psychological interventions (Applebaum et al., 2015; Yehuda et al., 2015). Only 15 of 39 participants in the current study endorsed a statement suggesting lack of interest or concern about late complications and future health problems. More than half (20 of 39) endorsed a statement related to avoidance behavior. Although the healthcare decisions of young adult survivors are complex, these endorsements are further evidence that avoidance continues to play a role in the lives of survivors even in the long-term survivorship period, perhaps causing the deferment of recommended survivorship care. Results from the current study suggest the need for longitudinal research to determine if PTSSs predict avoidance of survivorship care. Regardless of whether these symptoms contribute to deferred care, PTSSs are related to negative long-term physical and psychological health effects (Ahmadi et al., 2018; Boscarino, 2004; Kroenke et al., 2007; Meeske et al., 2001) and deserve additional investigation in this population.
Limitations
The current study must also be understood in the context of several limitations. One is the use of the version of the PTSD-RI based on the fourth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-IV), which was used from 1994–2013, when it was replaced by the fifth edition (DSM-V). The DSM-V tightened the definition of a trauma (Pai, Suris, & North, 2017), and in the DSM-V, the diagnosis of a life-threatening illness, such as cancer, is no longer automatically considered a traumatic event, as it is in the DSM-IV, unless it is associated with adverse events (Cordova, Riba, & Spiegel, 2017). However, this is one of the most controversial aspects of the revision (Friedman, 2013; Pai et al., 2017), and comprehensive psychometrics are not yet available for the DSM-V (Pai et al., 2017). In addition, most individuals with childhood cancer are subjected to invasive treatments and procedures, and many experience adverse effects (Bryant, 2003; Singh, Rees, & Sander, 2007) that could be considered traumatic. Also, patients can present with PTSSs without meeting the strict definition of a traumatic event in the DSM-V (APA, 2013). Therefore, this limitation does not impede the ability of the current authors to examine PTSSs in this sample of childhood cancer survivors because the actual diagnosis of PTSD is not being discussed, only the severity of symptoms defined as the likelihood, given patient-reported symptoms, that a formal diagnosis of PTSD would be made by a clinician.
A second limitation is that all eligible participants had a PCP, so the current authors could not examine the association between having or not having a PCP and PTSSs. Also, many patients assessed for eligibility refused to participate for reasons such as lack of time, which could have affected the results; PTSSs may have differed in participants versus nonparticipants. In addition, the authors did not directly assess other reasons for deferring survivorship care (e.g., lack of information about survivorship care), and the small sample size limited the authors’ ability to detect statistically significant differences in outcomes. The lack of significant changes could also be attributable to the intervention being a one-time visit. No psychological interventions were delivered to improve PTSSs. Although education about late complications was provided during the visit, with the potential to trigger PTSSs, the current authors did not assess changes in knowledge or perception of risk before and after the visit, and it is not possible to determine what effects this one visit had on PTSSs.
However, a strength of the current study is that it was part of a larger randomized trial, ensuring that confounding variables were equally distributed between randomization groups. In addition, the sample was comprised entirely of cancer survivors who had not previously attended a survivorship clinic to receive comprehensive survivorship care, which represents the majority of childhood cancer survivors; this is an understudied group.
Implications for Nursing Practice
The current study suggests that PTSSs are present among young adult survivors of childhood cancer not receiving survivorship care. Nurses in healthcare settings that do not specialize in the care of long-term survivors should be aware that PTSSs may contribute to deferred survivorship care and should encourage survivorship care, particularly if patients express feelings indicative of PTSSs. Nurses can highlight the protective role of survivorship care in patients’ ongoing health maintenance. Nurses can also advocate for and/or provide appropriate screening for PTSSs and refer patients with PTSSs to the appropriate psychosocial support services at their medical center or in their community. Assessing PTSSs in childhood cancer survivors, and even among patients still on therapy, is critical because PTSSs can be targeted with interventions to improve quality of life and health behaviors (Karapetian, 2015). Evidence-based treatments, such as cognitive processing therapy and prolonged exposure therapy, have been tailored to and shown to be effective for individuals with medical-related trauma (Applebaum et al., 2015; Yehuda et al., 2015). An understanding of which symptoms to target can help nurses and physicians determine the most appropriate techniques to use with patients. For example, patients experiencing avoidance symptoms respond best to imaginal and in vivo exposure (Zoellner et al., 2011), which encourages patients to engage with and process their trauma. Relaxation techniques (e.g., breathing exercises) and medication can be used to manage hyperarousal (Taylor et al., 2003), including sleep problems, difficulty concentrating, and irritability. Research suggests that it may be possible for trained clinicians to effectively deliver coping skills training via telephone (Judge Santacroce, Asmus, Kadan-Lottick, & Grey, 2010). Nurses can promote and implement targeted interventions in young adults with cancer to help them transition to and actively engage in survivorship care. Additional research in a larger sample is needed to better quantify rates of PTSSs in this population and determine which patients may benefit from interventions to address PTSSs. 
Conclusion
This study suggests that young adult survivors of childhood cancer who have not attended a survivorship clinic are at risk for PTSSs. Avoidance or numbing symptoms were most prevalent, which could explain, in part, deferral of survivorship care. This study should be replicated in a larger longitudinal sample to examine patterns of change in PTSSs during the years following treatment. Studies should examine interventions aimed at engaging young adult survivors who may be inhibited by avoidance symptoms in lifelong survivorship care.
About the Author(s)
Wilhelmenia L. Ross, MPH, is a research coordinator in the School of Medicine at Yale University in New Haven, CT; Hannah-Rose Mitchell, MPH, is a graduate student in the Department of Psychology at the University of Miami in Florida; Neel S. Iyer, DO, MPH, is a resident in the Department of Obstetrics and Gynecology at Cooper University Hospital in Camden, NJ; Sheila Judge Santacroce, PhD, RN, CPNP, FAANP, is an associate professor and Beerstecher-Blackwell Distinguished Scholar in the School of Nursing at the University of North Carolina in Chapel Hill; and Nina S. Kadan-Lottick, MD, MSPH, is an associate professor in the School of Medicine at Yale University. This research was funded by a grant (119700-RSGHP-10-107-01-CPHPS) from the American Cancer Society. Mitchell, Santacroce, and Kadan-Lottick contributed to the conceptualization and design. Mitchell, Iyer, and Kadan-Lottick completed the data collection. Ross, Mitchell, and Iyer provided statistical support. All authors provided the analysis and contributed to the manuscript preparation. Ross can be reached at wilhelmenia.ross@yale.edu, with copy to ONFEditor@ons.org. (Submitted January 2018. Accepted June 20, 2018.)
References
Ahmadi, N., Hajsadeghi, F., Nabavi, V., Olango, G., Molla, M., Budoff, M., . . . Yehuda, R. (2018). The long-term clinical outcome of posttraumatic stress disorder with impaired coronary distensibility. Psychosomatic Medicine, 80, 294–300.
Allen, J., Willard, V.W., Klosky, J.L., Li, C., Srivastava, D.K., Robison, L.L., . . . Phipps, S. (2018). Posttraumatic stress-related psychological functioning in adult survivors of childhood cancer. Journal of Cancer Survivorship, 12, 216–223. https://doi.org/10.1007/s11764-017-0660-x
American Psychiatric Association. (2000). Diagnostic and statistical manual of mental disorders (4th ed.). Washington, DC: Author.
American Psychiatric Association. (2013). Diagnostic and statistical manual of mental disorders (5th ed.). Arlington, VA: Author.
Applebaum, A.J., Bedoya, C.A., Hendriksen, E.S., Wilkinson, J.L., Safren, S.A., & O’Cleirigh, C. (2015). Future directions for interventions targeting PTSD in HIV-infected adults. Journal of the Association of Nurses in AIDS Care, 26, 127–138. https://doi.org/10.1016/j.jana.2014.11.001
Arnett, J.J. (2000). Emerging adulthood: A theory of development from the late teens through the twenties. American Psychologist, 55, 469–480. https://doi.org/10.1037/0003-066X.55.5.469
Barakat, L.P., Schwartz, L.A., Szabo, M.M., Hussey, H.M., & Bunin, G.R. (2012). Factors that contribute to post-treatment follow-up care for survivors of childhood cancer. Journal of Cancer Survivorship, 6, 155–162. https://doi.org/10.1007/s11764-011-0206-6
Berg, C., Stratton, E., Esiashvili, N., Mertens, A., & Vanderpool, R.C. (2016). Providers’ perspectives of survivorship care for young adult survivors of childhood cancer. Journal of Cancer Education, 31, 31–38. https://doi.org/10.1007/s13187-015-0848-6
Boscarino, J.A. (2004). Posttraumatic stress disorder and physical illness: Results from clinical and epidemiologic studies. Annals of the New York Academy of Sciences, 1032, 141–153. https://doi.org/10.1196/annals.1314.011
Brinkman, T.M., Recklitis, C.J., Michel, G., Grootenhuis, M.A., & Klosky, J.L. (2018). Psychological symptoms, social outcomes, socioeconomic attainment, and health behaviors among survivors of childhood cancer: Current state of the literature. Journal of Clinical Oncology, 36, 2190–2197. https://doi.org/10.1200/jco.2017.76.5552
Bruce, M. (2006). A systematic and conceptual review of posttraumatic stress in childhood cancer survivors and their parents. Clinical Psychology Review, 26, 233–256. https://doi.org/10.1016/j.cpr.2005.10.002
Bryant, R. (2003). Managing side effects of childhood cancer treatment. Journal of Pediatric Nursing, 18, 113–125. https://doi.org/10.1053/jpdn.2003.11
Children’s Oncology Group. (2013). Long-term follow-up guidelines for survivors of childhood, adolescent, and young adult cancers [v.4.0]. Retrieved from http://www.survivorshipguidelines.org/pdf/LTFUGuidelines_40.pdf
Conybeare, D., Behar, E., Solomon, A., Newman, M.G., & Borkovec, T.D. (2012). The PTSD Checklist—Civilian Version: Reliability, validity, and factor structure in a nonclinical sample. Journal of Clinical Psychology, 68, 699–713. https://doi.org/10.1002/jclp.21845
Cordova, M.J., Riba, M.B., & Spiegel, D. (2017). Post-traumatic stress disorder and cancer. Lancet Psychiatry, 4, 330–338. https://doi.org/10.1016/s2215-0366(17)30014-7
Docherty, S.L., Kayle, M., Maslow, G.R., & Santacroce, S.J. (2015). The adolescent and young adult with cancer: A developmental life course perspective. Seminars in Oncology Nursing, 31, 186–196. https://doi.org/10.1016/j.soncn.2015.05.006
Elhai, J.D., Layne, C.M., Steinberg, A.M., Brymer, M.J., Briggs, E.C., Ostrowski, S.A., & Pynoos, R.S. (2013). Psychometric properties of the UCLA PTSD Reaction Index. Part II: Investigating factor structure findings in a national clinic-referred youth sample. Journal of Traumatic Stress, 26, 10–18. https://doi.org/10.1002/jts.21755
Franklin, C.L., Piazza, V., Chelminski, I., & Zimmerman, M. (2015). Defining subthreshold PTSD in the DSM-IV literature: A look toward DSM-5. Journal of Nervous and Mental Disease, 203, 574–577.
Frederick, N.N., Bober, S.L., Berwick, L., Tower, M., & Kenney, L.B. (2017). Preparing childhood cancer survivors for transition to adult care: The young adult perspective. Pediatric Blood and Cancer, 64(10), e26544. https://doi.org/10.1002/pbc.26544
French-Rosas, L.N., Moye, J., & Naik, A.D. (2011). Improving the recognition and treatment of cancer-related posttraumatic stress disorder. Journal of Psychiatric Practice, 17, 270–276. https://doi.org/10.1097/01.pra.0000400264.30043.ae
Friedman, M.J. (2013). Finalizing PTSD in DSM-5: Getting here from there and where to go next. Journal of Traumatic Stress, 26, 548–556. https://doi.org/10.1002/jts.21840
Hobbie, W.L., Stuber, M., Meeske, K., Wissler, K., Rourke, M.T., Ruccione, K., . . . Kazak, A.E. (2000). Symptoms of posttraumatic stress in young adult survivors of childhood cancer. Journal of Clinical Oncology, 18, 4060–4066. https://doi.org/10.1200/jco.2000.18.24.4060
Jones, C. (2010). Post-traumatic stress disorder in ICU survivors. Journal of the Intensive Care Society, 11(Suppl.), 12–14. https://doi.org/10.1177/17511437100112s105
Judge Santacroce, S., Asmus, K., Kadan-Lottick, N., & Grey, M. (2010). Feasibility and preliminary outcomes from a pilot study of coping skills training for adolescent–young adult survivors of childhood cancer and their parents. Journal of Pediatric Oncology Nursing, 27, 10–20.
Kadan-Lottick, N.S., Ross, W.L., Mitchell, H.-R., Rotatori, J., Gross, C.P., & Ma, X. (2018). Randomized trial of the impact of empowering childhood cancer survivors with survivorship care plans. Journal of the National Cancer Institute. Advance online publication. https://doi.org/10.1093/jnci/djy057
Karapetian, G. (2015). Trauma-focused cognitive behavioral therapy (TFCBT). Retrieved from https://www.cibhs.org/trauma-focused-cognitive-behavioral-therapy-tfcbt
Kazak, A.E., Abrams, A.N., Banks, J., Christofferson, J., DiDonato, S., Grootenhuis, M.A., . . . Kupst, M.J. (2015). Psychosocial assessment as a standard of care in pediatric cancer. Pediatric Blood and Cancer, 62 (Suppl. 5), S426–S459. https://doi.org/10.1002/pbc.25730
Kazak, A.E., Alderfer, M., Rourke, M.T., Simms, S., Streisand, R., & Grossman, J.R. (2004). Posttraumatic stress disorder (PTSD) and posttraumatic stress symptoms (PTSSS) in families of adolescent childhood cancer survivors. Journal of Pediatric Psychology, 29, 211–219. https://doi.org/10.1093/jpepsy/jsh022
Kazak, A.E., Hocking, M.C., Ittenbach, R.F., Meadows, A.T., Hobbie, W., DeRosa, B.W., . . . Reilly, A. (2012). A revision of the Intensity of Treatment Rating Scale: Classifying the intensity of pediatric cancer treatment. Pediatric Blood and Cancer, 59, 96–99. https://doi.org/10.1002/pbc.23320
Koutná, V., Jelínek, M., Blatný, M., & Kepák, T. (2017). Predictors of posttraumatic stress and posttraumatic growth in childhood cancer survivors. Cancers, 9(3), E26. https://doi.org/10.3390/cancers9030026
Kroenke, K., Spitzer, R.L., Williams, J.B., Monahan, P.O., & Löwe, B. (2007). Anxiety disorders in primary care: Prevalence, impairment, comorbidity, and detection. Annals of Internal Medicine, 146, 317–325. https://doi.org/10.7326/0003-4819-146-5-200703060-00004
Lee, Y.-L., & Santacroce, S.J. (2007). Posttraumatic stress in long-term young adult survivors of childhood cancer: A questionnaire survey. International Journal of Nursing Studies, 44, 1406–1417. https://doi.org/10.1016/j.ijnurstu.2006.07.002
Meeske, K.A., Ruccione, K., Globe, D.R., & Stuber, M.L. (2001). Posttraumatic stress, quality of life, and psychological distress in young adult survivors of childhood cancer. Oncology Nursing Forum, 28, 481–489.
Nathan, P.C., Ford, J.S., Henderson, T.O., Hudson, M.M., Emmons, K.M., Casillas, J.N., . . . Oeffinger, K.C. (2009). Health behaviors, medical care, and interventions to promote healthy living in the Childhood Cancer Survivor Study cohort. Journal of Clinical Oncology, 27, 2363–2373. https://doi.org/10.1200/jco.2008.21.1441
Nathan, P.C., Greenberg, M.L., Ness, K.., Hudson, M.M., Mertens, A.C., Mahoney, M.C., . . . Oeffinger, K.C. (2008). Medical care in long-term survivors of childhood cancer: A report from the Childhood Cancer Survivor Study. Journal of Clinical Oncology, 26, 4401–4409. https://doi.org/10.1200/jco.2008.16.9607
Noel, M., Wilson, A.C., Holley, A.L., Durkin, L., Patton, M., & Palermo, T.M. (2016). Posttraumatic stress disorder symptoms in youth with vs without chronic pain. Pain, 157, 2277–2284. https://doi.org/10.1097/j.pain.0000000000000642
Oeffinger, K.C., Mertens, A.C., Sklar, C.A., Kawashima, T., Hudson, M.M., Meadows, A.T., . . . Robison, L.L. (2006). Chronic health conditions in adult survivors of childhood cancer. New England Journal of Medicine, 355, 1572–1582. https://doi.org/10.1056/NEJMsa060185
Oeffinger, K.C., & Wallace, W.H. (2006). Barriers to follow-up care of survivors in the United States and the United Kingdom. Pediatric Blood and Cancer, 46, 135–142. https://doi.org/10.1002/pbc.20614
Osterman, J.E., Hopper, J., Heran, W.J., Keane, T.M., & van der Kolk, B.A. (2001). Awareness under anesthesia and the development of posttraumatic stress disorder. General Hospital Psychiatry, 23, 198–204. https://doi.org/10.1016/S0163-8343(01)00142-6
Pai, A., Suris, A.M., & North, C.S. (2017). Posttraumatic stress disorder in the DSM-5: Controversy, change, and conceptual considerations. Behavioral Sciences, 7(1), E7. https://doi.org/10.3390/bs7010007
Price, J., Kassam-Adams, N., Alderfer, M.A., Christofferson, J., & Kazak, A.E. (2016). Systematic review: A reevaluation and update of the integrative (trajectory) model of pediatric medical traumatic stress. Journal of Pediatric Psychology, 41, 86–97. https://doi.org/10.1093/jpepsy/jsv074
Rodriguez, N., Steinberg, A.S., Saltzman, W.S., & Pynoos, R.S. (2001, December 6). PTSD Index: Psychometric analyses of the adolescent version. Paper presented at the annual meeting of the International Society for Traumatic Stress Studies, New Orleans, LA.
Rourke, M.T., Hobbie, W.L., Schwartz, L., & Kazak, A.E. (2007). Posttraumatic stress disorder (PTSD) in young adult survivors of childhood cancer. Pediatric Blood and Cancer, 49, 177–182. https://doi.org/10.1002/pbc.20942
Santacroce, S.J., & Lee, Y.-L. (2006). Uncertainty, posttraumatic stress, and health behavior in young adult childhood cancer survivors. Nursing Research, 55, 259–266. https://doi.org/10.1097/00006199-200607000-00006
Schwartz, L., & Drotar, D. (2006). Posttraumatic stress and related impairment in survivors of childhood cancer in early adulthood compared to healthy peers. Journal of Pediatric Psychology, 31, 356–366. https://doi.org/10.1093/jpepsy/jsj018
Shemesh, E., Lurie, S., Stuber, M.L., Emre, S., Patel, Y., Vohra, P.,. . . Shneider, B. L. (2000). A pilot study of posttraumatic stress and nonadherence in pediatric liver transplant recipients. Pediatrics, 105, E29.
Shemesh, E., Rudnick, A., Kaluski, E., Milovanov, O., Salah, A., Alon, D., . . . Cotter, G. (2001). A prospective study of posttraumatic stress symptoms and nonadherence in survivors of a myocardial infarction (MI). General Hospital Psychiatry, 23, 215–222. https://doi.org/10.1016/S0163-8343(01)00150-5
Singh, G., Rees, J.H., & Sander, J.W. (2007). Seizures and epilepsy in oncological practice: Causes, course, mechanisms and treatment. Journal of Neurology, Neurosurgery, and Psychiatry, 78, 342–349. https://doi.org/10.1136/jnnp.2006.106211
Steinberg, A.M., & Brymer, M.J. (2008). The UCLA PTSD Reaction Index. In G. Reyes, J.D. Elhai, & J.D. Ford (Eds.), Encyclopedia of psychological trauma (pp. 673–674). Hoboken, NJ: John Wiley and Sons.
Steinberg, A.M., Brymer, M.J., Decker, K.B., & Pynoos, R.S. (2004). The University of California at Los Angeles Post-Traumatic Stress Disorder Reaction Index. Current Psychiatry Reports, 6, 96–100. https://doi.org/10.1007/s11920-004-0048-2
Steinberg, A.M., Brymer, M.J., Kim, S., Briggs, E.C., Ippen, C.G., Ostrowski, S.A., . . . Pynoos, R.S. (2013). Psychometric properties of the UCLA PTSD Reaction Index: Part I. Journal of Traumatic Stress, 26, 1–9. https://doi.org/10.1002/jts.21780
Stuber, M.L., Kazak, A.E., Meeske, K., & Barakat, L. (1998). Is posttraumatic stress a viable model for understanding responses to childhood cancer? Child and Adolescent Psychiatric Clinics of North America, 7, 169–182.
Stuber, M.L., Meeske, K.A., Krull, K.R., Leisenring, W., Stratton, K., Kazak, A.E., . . . Zeltzer, L.K. (2010). Prevalence and predictors of posttraumatic stress disorder in adult survivors of childhood cancer. Pediatrics, 125, e1124–e1134. https://doi.org/10.1542/peds.2009-2308
Supelana, C., Annunziato, R.A., Kaplan, D., Helcer, J., Stuber, M.L., & Shemesh, E. (2016). PTSD in solid organ transplant recipients: Current understanding and future implications. Pediatric Transplantation, 20, 23–33. https://doi.org/10.1111/petr.12628
Taylor, S., Thordarson, D.S., Maxfield, L., Fedoroff, I.C., Lovell, K., & Ogrodniczuk, J. (2003). Comparative efficacy, speed, and adverse effects of three PTSD treatments: Exposure therapy, EMDR, and relaxation training. Journal of Consulting and Clinical Psychology, 71, 330–338. https://doi.org/10.1037/0022-006X.71.2.330
Vannatta, K., & Gerhardt, C.A. (2003). Pediatric oncology: Psychosocial outcomes for children and families. In M.C. Roberts (Ed.), Handbook of pediatric psychology (3rd ed., pp. 342–357). New York, NY: Guilford Press.
Varela, V.S., Ng, A., Mauch, P., & Recklitis, C.J. (2013). Posttraumatic stress disorder (PTSD) in survivors of Hodgkin’s lymphoma: Prevalence of PTSD and partial PTSD compared with sibling controls. Psycho-Oncology, 22, 434–440. https://doi.org/10.1002/pon.2109
Vuotto, S.C., Procidano, M.E., & Annunziato, R.A. (2015). Understanding the health behaviors of survivors of childhood and young-adult cancer: Preliminary analysis and model development. Children, 2, 174–190. https://doi.org/10.3390/children2020174
Werba, B.E., Hobbie, W., Kazak, A.E., Ittenbach, R.F., Reilly, A.F., & Meadows, A.T. (2007). Classifying the intensity of pediatric cancer treatment protocols: The intensity of treatment rating scale 2.0 (ITR-2). Pediatric Blood and Cancer, 48, 673–677. https://doi.org/10.1002/pbc.21184
Yehuda, R., Hoge, C.W., McFarlane, A.C., Vermetten, E., Lanius, R.A., Nievergelt, C.M., . . . Hyman, S.E. (2015). Post-traumatic stress disorder. Nature Reviews Disease Primers, 1, 15057. https://doi.org/10.1038/nrdp.2015.57
Zeltzer, L.K., Recklitis, C., Buchbinder, D., Zebrack, B., Casillas, J., Tsao, J.C., . . . Krull, K. (2009). Psychological status in childhood cancer survivors: A report from the Childhood Cancer Survivor Study. Journal of Clinical Oncology, 27, 2396–2404. https://doi.org/10.1200/jco.2008.21.1433
Zheng, D.J., Sint, K., Mitchell, H.-R., & Kadan-Lottick, N.S. (2016). Patterns and predictors of survivorship clinic attendance in a population-based sample of pediatric and young adult childhood cancer survivors. Journal of Cancer Survivorship, 10, 505–513. https://doi.org/10.1007/s11764-015-0493-4
Zoellner, L.A., Feeny, N.C., Bittinger, J.N., Bedard-Gilligan, M.A., Slagle, D.M., Post, L.M., & Chen, J.A. (2011). Teaching trauma-focused exposure therapy for PTSD: Critical clinical lessons for novice exposure therapists. Psychological Trauma, 3, 300–308. https://doi.org/10.1037/a0024642




