Physical Activity in Young Adult Cancer Survivors: A Scoping Review
Problem Identification: Physical activity, a precision health strategy, positively affects biopsychosocial health in adult cancer survivors. However, understanding its effects among young adult (YA) cancer survivors is limited. The purpose of this scoping review was to explore existing research on physical activity in YA cancer survivors.
Literature Search: CINAHL®, PubMed®, PsycINFO®, SPORTDiscus, Web of Science, and Cochrane Library were searched, producing 63 articles and 28 grey materials.
Data Evaluation: Data extraction, guided by the revised symptom management model, included research aims, sample, design, primary outcome measures, and effects of physical activity.
Synthesis: Findings of 35 review articles were reported under three main categories.
Implications for Research: Lack of clinical guidelines and limited research specific to YA cancer survivors hinders physical activity’s use as a symptom management strategy. Research is needed that addresses the development and clinical implementation of physical activity guidelines.
Jump to a section
Annually, in the United States, there are about 89,000 new cancer cases in adolescents and young adults (AYAs) (ages 15–39 years) (National Cancer Institute, 2020). Significant improvements in AYA early diagnosis and treatments have led to increased cancer survival rates that exceed 80% (Barr, 2011; Keegan et al., 2016; Roberts et al., 2017; Robison et al., 2009). However, cancer morbidity has increased during the past two decades at a rate three times higher among those aged 15–29 years than among children (Bleyer, 2007; Bleyer et al., 2017). AYA cancer survivors have a significantly increased chronic disease risk compared to those without cancer, with more than two-thirds developing at least one chronic condition by age 40 years, limiting their full life potential (Bradford & Chan, 2017; Phillips et al., 2015). Cancer and its treatments affect multiple biologic and psychosocial systems, resulting in late effects and long-lasting symptoms that can cause distress and disrupt social functioning (Dodd et al., 2001). In adults, symptom management techniques can mitigate these disruptive symptoms. The revised symptom management model (Dodd et al., 2001) encapsulates biologic, psychological, and social aspects of symptoms and symptom management. By addressing all aspects of health in young adults affected by cancer, this biopsychosocial approach offers improved function and quality of life.
There is a paucity of YA cancer research, with most of it being encapsulated within the AYA research. Therefore, this review will encompass references to both AYAs and YAs, with special attention paid to the YA population because of a critical need for research in this vulnerable cohort. Research in YAs (ages 18–39 years), a subpopulation of AYAs, is limited, with less than 2% of YA cancer survivors involved in clinical trials (Bleyer, 2007; Docherty et al., 2015; Fernandez et al., 2011; Keegan et al., 2016). Low participation in clinical trials is thought to be influenced by geographic distance, lack of insurance coverage, age, and pediatric versus nonpediatric treating oncologist (Kopp et al., 2016; Parsons et al., 2019). YAs experience an increased risk of cardiometabolic issues (Kopp et al., 2016; Nass et al., 2015; Rabin, 2011), with cardiovascular disease being the second leading cause of death in childhood and AYA cancer survivors (Armstrong et al., 2016). Cancer and its treatment often disrupt critical emotional, cognitive, and social developmental needs in AYAs in ways that can significantly impair their psychosocial health (Nass et al., 2015; Patterson et al., 2015). In addition, survivorship care is embedded in fragmented care delivery models that neglect AYAs’ unique needs, leading to more than 50% reporting unmet needs (Fernandez et al., 2011; Patterson et al., 2015).
Survivorship care focuses on the overall health, well-being, and quality of life of individuals affected by cancer (National Comprehensive Cancer Network [NCCN], 2020). Survivorship care should be evidence-based, dynamic, and responsive to the unique and evolving needs of cancer survivors, which may change rapidly in relation to ongoing changes in their health and life contexts (Howell, 2018). Despite the numerous challenges that AYAs face, this group is largely excluded from survivorship care research; research specifically involving YAs is even more limited (Bleyer, 2007; Docherty et al., 2015; Fernandez et al., 2011; Keegan et al., 2016). Clinical experts have recognized the urgent need to develop and deliver survivorship care specific to the needs and unique life circumstances of this understudied group of cancer survivors (Rabin et al., 2013). High-quality survivorship care includes support for health promotion activities designed to mitigate negative late effects and long-term impacts of cancer treatments and to reduce the risk of cancer recurrence (NCCN, 2020). However, in YAs, there are no universally accepted practice guidelines that specifically address physical activity as a precision health strategy for symptom management and improved quality of life.
Recommendations from the National Academies of Sciences, Engineering, and Medicine (2019) on cancer control call for an increase in effective and affordable interventions for preventive, screening, and diagnostic cancer care that integrates social and behavior information to develop reliable evidence. The field of exercise oncology has grown exponentially in the past few decades (Jones & Alfano, 2013; National Cancer Institute, 2019) to develop reliable evidence on the role of physical activity in cancer care. Nurses were involved in its pioneering stage (Jones & Alfano, 2013; Winningham et al., 1989). The American College of Sports Medicine published adult physical activity guidelines for cancer survivors (Schmitz et al., 2019). However, physical activity guidelines informed by the unique preferences (e.g., psychological and environmental aspects) and developmental needs of YAs do not exist (Brunet et al., 2018; Munsie et al., 2019; Nass et al., 2015; Pugh et al., 2016; Rabin, 2011). There is a critical need to understand the state of the science on how physical activity, a modifiable precision health strategy, contributes to biopsychosocial health, well-being, and overall quality of life for YA cancer survivors and how it can be feasibly and sustainably incorporated into survivorship care. The purpose of this scoping review is to explore the existing research to date, incorporating published clinical guidelines, protocol statements, and critical work in the field, to identify the following: relationships among physical activity, cancer-related symptoms, and quality of life in YA cancer survivors, the measurement of physical activity in research involving YA cancer survivors, and guidelines for activity among YA survivors.
Methods
Literature Search and Data Evaluation
A scoping review is an iterative literature review process that allows for synthesis of the evidence with the aim of providing a broad overview of current scholarship on a topic of interest. In this sense, a scoping review may be more inclusive and sensitive to emergent areas of research and expert opinion than a more narrowly defined systematic review of the literature. This literature review process was chosen because of the complexity and lack of comprehensive research in physical activity among YA cancer survivors. This scoping review was performed using the following steps, as outlined by Arksey and O’Malley (2005):
• Stage 1: Identify the research question.
• Stage 2: Identify relevant studies.
• Stage 3: Perform study selection.
• Stage 4: Chart the data.
• Stage 5: Collate, summarize, and report the results.
The search strategy was developed in consultation with a health sciences librarian at the University of Massachusetts Amherst to include databases comprehensive enough to capture both cancer and physical activity research. The following databases were searched: CINAHL®, PubMed®, PsycINFO®, SPORTDiscus, Web of Science, and Cochrane Library. The search terms young adult AND cancer survivor AND exercise OR physical activity OR fitness OR movement OR activity AND meta-analysis OR meta-synthesis OR systematic review OR scoping review were used. The Cochrane Library was searched using the following MeSH (Medical Subject Headings) terms (all text): young adults AND cancer survivors AND exercise OR physical activity OR fitness OR movement OR activity. The search was limited to English language and peer reviewed without year limitation. The most recent search was performed in December 2019.
Inclusion Criteria
Systematic reviews, meta-analyses, meta-syntheses, or scoping research studies were considered for this scoping review if they included YAs aged 18–39 years, survivors diagnosed as a child and/or those diagnosed as YAs, and one or more of the following search terms: exercise, physical activity, fitness, movement, or activity. An a priori decision was made not to include sedentary behavior in the search terms because this is emerging science and the preliminary search yielded no results; however, attention to studies that included sedentary behavior was noted and summarized.
Data Sources and Search Strategy
Titles and abstracts of retrieved articles were initially screened for relevance by one reviewer (A.M.M.) using the defined inclusion and exclusion criteria and discussed with one other reviewer (R.K.W.). A total of 61 articles were identified through database searching, as noted in Figure 1. The majority of articles retrieved spanned across multiple age ranges, with only two articles specific to YAs, demanding careful consideration of what age range to include in the scoping review. To meet the aims of the research, a wider age range for inclusion needed to be considered. The authors, in consultation with the health sciences librarian, made the decision to include articles if 25% or more of the studies included sampling of YAs. Full texts of articles that met inclusion criteria (n = 20) were retrieved and reviewed by the authors using the same inclusion and exclusion criteria. Twelve peer-reviewed articles were selected for inclusion in the final analysis with consensus of all reviewers. In line with the purpose of scoping reviews, to provide literature, conceptual, and policy statements that add context and comprehensively map evidence, grey literature was added (Anderson et al., 2008; Levac et al., 2010; O’Brien et al., 2016). Grey material was identified by searching the bibliographies of the included articles and identifying related documents and statements from governing and professional bodies, and was reviewed by the authors for inclusion. A total of 23 grey materials (of 28 retrieved) related to clinical practice guidelines, expert recommendations, policy statements, and/or critical work in YA survivorship were included. In total, 35 articles were included in this scoping review. 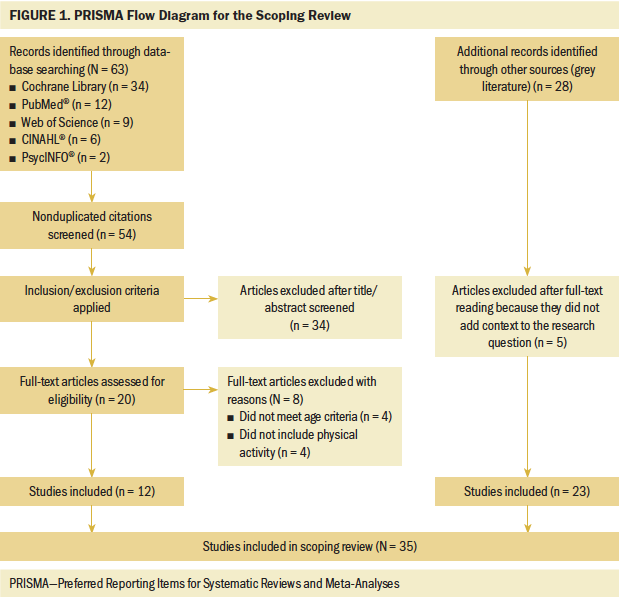
Data Extraction
Full articles were independently read by the authors. Consensus among the authors was 100%. Articles were abstracted by A.M.M. using a data extraction tool, and results of this abstraction process were placed in an organizational matrix (preformatted Microsoft® Excel spreadsheet) to allow for easy comparison of findings across all articles. The revised symptom management model (Dodd et al., 2001) informed the design of the organizational matrix. To facilitate the analysis and synthesis of resources with methodologic diversity, the data extraction tool included details about the study aims, sample and sample characteristics, study design, primary outcome measures, major findings, and the effects of physical activity on AYAs. In addition, data on variables not related to biopsychosocial health, quality of life, and measurement of practice guidelines were extracted and included to provide a more comprehensive understanding of the literature. The research in YA cancer survivors was limited, and the majority of the articles (n = 10) were published between 2016 and 2019, reflecting the emergence of this research and further warranting this scoping review. Multiple types of research studies, as well as policy and protocol statements from governing and regulatory bodies, were synthesized in this scoping review: systematic review (n = 10), systematic review and meta-analysis (n = 4), systematic review and narrative synthesis (n = 1), meta-synthesis (n = 1), scoping review (n = 1), recommendation (n = 6), and summary and review (n = 12).
Synthesis
There are inconsistencies regarding what age defines “young adult” in cancer survivorship, with many different cutoff points to describe this age cohort. Research specific to YA cancer survivorship and physical activity is very limited, with only two publications (Rabin, 2011, 2017) located for this review. Most of the reviews (Bleyer et al., 2017; Brunet et al., 2018; Coccia et al., 2018; Docherty et al., 2015; Fernandez et al., 2011; Keegan et al., 2016; Kopp et al., 2016; Nass et al., 2015; Patterson et al., 2015; Pugh et al., 2016; Smith et al., 2016; Tai et al., 2012) included in this review encompass adolescents and AYAs aged 15–39 years affected by cancer. Because of vast biologic, developmental, and psychosocial heterogeneity (Keegan et al., 2016) of YA cancer survivors, it is unclear if they are best defined by their age, cancer status, or both (Rabin, 2011). Although many studies included do not report race and ethnicity data, for those that do, study samples are primarily White and English-speaking individuals (Bradford & Chan, 2017; Kopp et al., 2016; Pugh et al., 2016). All of these factors contributed to a lack of generalizability.
Relationships Among Physical Activity, Cancer-Related Symptoms, and Quality of Life
AYA cancer survivors’ psychosocial, medical, economic challenges (Bleyer, 2007), developmental needs (Spathis et al., 2015), and care transitions are unique and distinct from those of older adult cancer survivors (Fernandez et al., 2011). AYA cancer survivors experience higher levels of anxiety, distress, and depression (Fernandez et al., 2011; Tai et al., 2012) but are less likely to access professional mental health services (Coccia et al., 2018). There are likely multiple interconnected mechanisms driving poor psychosocial health, including sociocultural (temporary or permanent exclusion from significant socialization opportunities, such as school or group sports) and behavioral pathways (activation of the autonomic nervous system with an increase in stress hormones that increase tumor cell growth and promote angiogenesis) (Chida et al., 2008). Particular insults to self-image and bodily changes, such as hair loss, weight changes, risk of infection, altered sexuality, loss of normal activities of daily living, and financial issues, negatively affect psychosocial health (Bleyer, 2007; Fernandez et al., 2011). AYA cancer survivors also experience financial toxicity, with fewer being employed, significantly more being out of work (Nass et al., 2015), and a higher proportion not being able to seek health care, because of cost, than peers without cancer (Tai et al., 2012), which negatively affects psychosocial health (Bleyer, 2007; Fernandez et al., 2011). An estimated 75% of AYA cancer survivors experience cognitive changes that affect memory, learning, concentration, and decision-making (Nass et al., 2015; Treanor et al., 2016).
YAs have an increased risk of cardiometabolic and pulmonary issues (Kopp et al., 2016; Nass et al., 2015; Rabin, 2011; Tai et al., 2012). Treatment exposures from chemotherapy and radiation therapy increase late effects on cardiovascular health (e.g., increased risk of myocardial infarction, stroke, congestive heart failure, valvular issues, and hypertension), metabolic health (e.g., increased risk of obesity, dyslipidemia, insulin resistance, and diabetes), and pulmonary health (e.g., restrictive lung disease, pulmonary fibrosis), leading to exercise intolerance; in addition, sexual dysfunction is a prominent late effect from cancer treatments (Nass et al., 2015; Tai et al., 2012).
Fatigue is a common and often debilitating symptom for YA survivors and has the most detrimental effects on the physical and functional dimensions of quality of life (Cabilan & Hines, 2017; Rabin, 2011; Spathis et al., 2015). This review indicates that fatigue can be a barrier to physical activity (Rabin, 2011; Spathis et al., 2015) and is further complicated by the belief of survivors, family members, and healthcare clinicians that resting is the best approach to managing fatigue. However, inactivity is a maladaptive behavior that can perpetuate fatigue (Spathis et al., 2015). In adults, physical activity has been shown to mitigate the symptom of cancer-related fatigue (Schmitz et al., 2019; Spathis et al., 2015).
In adult cancer survivors, higher levels of physical activity show potential to mitigate symptom burden and positively affect quality of life, fatigue, prognostic biomarkers, secondary cancer risk, chronic disease risk, comorbidities, and mortality rates (Schmitz et al., 2019; Schwartz et al., 2017). More specifically, there is strong evidence for the efficacy of physical activity on anxiety, depressive symptoms, fatigue, quality of life, and perceived physical function; moderate evidence exists for the effect of exercise on sleep and bone health, but because of limited and poor methodologic studies, insufficient evidence exists for cardiotoxicity, chemotherapy-induced neuropathy, cognitive function, nausea, pain, sexual function, and treatment tolerance (Schmitz et al., 2019). Physical activity positively affects certain aspects of quality of life, including the following: the ability to physically manage effects of cancer and its treatment, the psychological ability to have positive self-perceptions, social aspects of interactions, and creation of purpose and meaning within a cancer diagnosis (Burke et al., 2017). Despite this, AYA cancer survivors are likely to be less physically active and have more sedentary time than their peers without cancer (Rabin, 2011; Smith et al., 2019). Research specific to the population of YAs with cancer is needed (Burke et al., 2017).
Measurement of Physical Activity in Research Involving Young Adult Cancer Survivors
YA cancer survivors have been vastly underrepresented in research (Bradford & Chan, 2017), with less than 2% of YA survivors aged 20–29 years and less than 15% of adolescents aged 15–19 years participating in clinical trials (Bleyer, 2007; Docherty et al., 2015; Fernandez et al., 2011). This is further compounded by limited studies related to physical activity and cancer survivorship (Cabilan & Hines, 2017). In addition, there are methodologic deficiencies, including modest effect sizes, lack of validated measures in interventional trials, heterogeneity in study designs and outcome measures, lack of control groups, lack of longitudinal studies, lack of randomized controlled trials, lack of control groups, and risk of bias, resulting in a lack of robust evidence and affecting clinical effects and statistical significance (Braam et al., 2016; Bradford & Chan, 2017; Brunet et al., 2018; Cabilan & Hines, 2017; Kopp et al., 2016; Munsie et al., 2019; Neufer et al., 2015; Rabin, 2011, 2017; Roberts et al., 2017; Smith et al., 2016; Spathis et al., 2015; Speck et al., 2010; Treanor et al., 2016; Wurz & Brunet, 2016).
There are issues on how or if physical activity is operationalized (Brunet et al., 2018), making it difficult to compare its effect across studies. Physical activity has been described as any form of activity that has the potential to promote health (Hagger, 2019). It has also been described as movement that elicits skeletal muscle contractions, resulting in increased caloric requirements that exceed resting energy expenditure (Caspersen et al., 1985; Rochmis & Blackburn, 1971). There is also a wide range of physical activity outcome measures, including self-report (Cabilan & Hines, 2017), accelerometer and pedometer data (Brunet et al., 2018; Kopp et al., 2016), minutes per day or metabolic equivalent per week of moderate to vigorous exercise or calories burned (Barbaric et al., 2010; Pugh et al., 2016), functional fitness outcome measures (Braam et al., 2016; Munsie et al., 2019), or a combination of such. Physical activity is often clustered with other health behaviors (Pugh et al., 2016) or is measured with inadequate study design to capture correlations, making it difficult to articulate its effects. Sedentary behavior, a separate risk factor from physical activity (Rabin, 2011), was discussed in some of the reviews, noting that increased sedentary time increases cancer risk and fatigue (Brunet et al., 2018; Kopp et al., 2016; Rabin, 2011; Roberts et al., 2017; Schmitz et al., 2019; Schwartz et al., 2017; Wolin et al., 2012). However, specific measurement of sedentary behavior was limited in the literature, other than noting that YA survivors spend more time in sedentary behavior than their peers without cancer (Rabin, 2011).
Guidelines for Physical Activity and Clinical Considerations Among Young Adult Survivors
Lifelong survivorship care is the recommended standard for AYA cancer survivors. The use of physical activity in YAs during and after treatment is an emerging strategy to decrease mortality and mitigate negative biopsychosocial effects of cancer treatment. However, there is limited scientific research in this age cohort compared to younger pediatric and adult cancer cohorts (Munsie et al., 2019; Roberts et al., 2017; Wurz & Brunet, 2016). In late 2019, the first set of national physical activity guidelines specifically designed to help adults prevent and treat cancer-related symptoms (e.g., fatigue, anxiety, depression, physical function, quality of life) was released (Schmitz et al., 2019). Research involving adult survivors indicates that home and community-based activity interventions before, during, and after cancer treatment improve physical functioning, role functioning, and social functioning aspects of health-related quality of life (Burke et al., 2017; Mishra et al., 2015; Wurz & Brunet, 2016).
The timing of the physical activity intervention should begin at the time of diagnosis and continue during treatment and the post-treatment period to maximize the restoration or enhancement of physical and functional status and promote well-being and normalcy (Cabilan & Hines, 2017). A study of YA hematopoietic stem cell transplantation survivors indicated that individuals exercised 30 minutes a day for 76% of their 24-day hospitalization, with the majority performing walking (Munsie et al., 2019), highlighting the feasibility of physical activity during treatment and hospitalization. Physical activity interventions in AYA cancer survivors are highly acceptable to study volunteers, with retention rates greater than 70% (Kopp et al., 2016). There are few behavioral interventions for YA survivors (Rabin, 2011), but of those that exist, physical activity is the most commonly targeted behavior (Pugh et al., 2016; Wurz & Brunet, 2016). Tailored physical activity programs have shown improvements in physical function and endurance (Munsie et al., 2019), highlighting the need to assess physical activity preferences that are influenced by unique personal preferences and demographic factors (Nass et al., 2015; Patterson et al., 2015).
No expert consensus or guidelines specific to YA cancer survivors exist on physical activity. The NCCN clinical practice guidelines call for an evaluation of exercise needs, hobbies, and recreational activities but offer no supportive care services or interventions (Coccia et al., 2018). Courneya et al. (2015) compiled a list of research questions related to physical activity and cancer survivorship to guide researchers in investigating the most critical areas. The American College of Sports Medicine proposed using the Exercise is Medicine® initiative to address practice gaps by clinicians in assessing, advising, and referring cancer survivors for activity and called on key stakeholders to create the necessary infrastructure to promote activity for individuals with cancer (Schmitz et al., 2019). Advancements in AYA survivorship care have lagged behind compared to other age cohorts (Bleyer et al., 2017).
Discussion
There are currently no expert consensus reports or formal clinical guidelines to guide recommendations specific to assessment, prescription, and implementation of physical activity as a symptom management strategy to mitigate biopsychosocial changes associated with cancer and cancer treatment in YAs. This lack of consensus hinders further research and efforts related to integrating physical activity into supportive care survivorship practices. The lack of activity in YA survivors is an issue that affects healthcare delivery worldwide, and consensus is needed to guide clinicians (Spathis et al., 2015).
Survivorship care focuses on the overall health, well-being, and quality of life of individuals affected by cancer (NCCN, 2020). Biopsychosocial health outcomes of cancer can be influenced by modifiable precision health lifestyle behaviors, such as diet, exercise, and inactivity, affecting quality of life (Dulaney et al., 2017; Nass et al., 2015; Rabin, 2011). Physical activity has emerged as one of the most promising foci for research and interventions to mitigate the negative effects of cancer and cancer treatment on biopsychosocial health. Physical activity is a safe, feasible, and viable option throughout the cancer continuum in YA cancer survivors, with retention rates greater than 70% (Bradford & Chan, 2017; Kopp et al, 2016; Munsie et al., 2019; Pugh et al., 2016; Rabin, 2011; Wurz & Brunet, 2016). In addition, there is a strong interest in receiving information regarding physical activity, with greater than 75% of those surveyed expressing interest and greater than 85% feeling able to participate in an activity program (Bélanger et al., 2012; Gupta et al., 2013; Rabin, 2011). Despite YAs articulating preferences, many clinical and research facets of physical activity (e.g., measurement, intervention design and delivery) specific to YA survivors have not been articulated.
Nurses have multiple opportunities to support the biopsychosocial health of YA cancer survivors at every stage in the cancer continuum (Dodd et al., 2001). Nursing scientists like Victoria Mock, PhD, RN, FAAN, were early pioneers in the investigation of hospital-based and home-based physical activity interventions on the sleep quality, emotional well-being, and fatigue levels of cancer survivors in active treatment and noted increased vigor with lower levels of emotional stress and fatigue in those who exercised (Jones & Alfano, 2013; Wenzel et al., 2013; Winningham et al., 1989). Professional oncology nursing associations, such as the Oncology Nursing Society, have also launched national practice initiatives designed to promote physical activity among cancer survivors. However, in a study by Schmitz et al. (2019), less than 9% of nurses discuss physical activity with cancer survivors.
Physical activity is an emerging precision health strategy. However, there are challenges regarding how to measure physical activity and sedentary behavior in the literature. The Nursing Science Precision Health Model is useful when considering a starting point for addressing the gaps in measurement, intervention design, and delivery. The Nursing Science Precision Health Model has the following four precision concepts: measurement; characterization of (psychosocial) phenotype, including influences from lifestyle and environment; characterization of genotype (biological markers); and intervention target, design, and delivery, with an emphasis on self-management techniques (Hickey et al., 2019).
There are limited reviews on the benefits of physical activity in YA cancer survivors; therefore, this scoping review adds to the field of physical activity and cancer research by addressing YAs affected by cancer. The scoping review methodology allowed for broad-lens data acquisition and synthesis from methodologically diverse data sources to address the critical gap of understanding the state of the science of physical activity in YA cancer survivors.
The limited operationalization of physical activity in the existing research, the breadth of definitions for what constitutes YAs, and the vast heterogeneity of developmental stages, psychosocial challenges, and life contexts that the age range of 18–39 years spans posed challenges to this review. In addition, the limited research specific to YAs, warranting inclusion of literature across multiple age ranges, limits the generalizability of this review. The use of a theory to guide this review and the use of a data extraction tool to describe the effects of physical activity on symptoms, function, and aspects of quality of life add to the strength of this review.
Implications for Nursing
Nurses can make a significant impact by exploring personal and structural facilitators and barriers to physical activity; providing tailored assessments and education; referring to cancer exercise specialists, when needed, to promote the integration of physical activity into survivorship; and advocating for equity-based policies and resources that increase access to such opportunities. Understanding the effects of physical activity in YA cancer survivors as a precision health strategy will provide a starting point for nurses to collaborate with survivors to enhance tailored symptom management, resulting in improved biopsychosocial health. This scoping review highlights the need for nursing scientists to explore areas of consensus on the effects of physical activity on biopsychosocial aspects of survivors to better articulate interventions that improve overall health, well-being, and quality of life among YA cancer survivors. 
Conclusion
Cancer survivorship rates in YAs are greater than 80%, but late and long-term symptoms reside. These “consequences of the cure” (Kopp et al., 2016, p. 13), along with fragmented models of care, leave more than 50% of YAs affected by cancer with unmet needs that affect biopsychosocial health, well-being, and quality of life. YAs experience elevated health risks because of the longevity of symptom burden and profound disruptions of normal psychosocial and developmental milestones. In adults, strong evidence exists on the efficacy of physical activity, a modifiable precision health behavior, as a symptom management strategy for anxiety, depressive symptoms, fatigue, and perceived physical function (Schmitz et al., 2019). In addition, it positively affects the ability to physically manage many effects of cancer and its treatment, the psychological ability to have positive self-perceptions, social aspects of interactions, and creation of purpose and meaning within a cancer diagnosis (Burke et al., 2017).
Physical activity, a precision health strategy, in YA cancer survivors has shown improvements in physical function; however, there is a paucity of research dedicated to this vulnerable population. Compounding this is the fact that most literature that includes YAs also straddles the AYA population, despite biologic, developmental, and psychosocial heterogeneity in those age groups. In addition, there are issues with how physical activity is operationalized and measured and a lack of clinical guidelines and recommendations specific to the unique needs of YAs.
This review elevates the call for evidence-based research and clinical guidelines, building on the American College of Sports Medicine’s Exercise is Medicine® (Schmitz et al., 2019) initiative to address practice gaps in assessing, advising, and referring cancer survivors for activity, and calls on key stakeholders to create the necessary infrastructure to promote physical activity that is tailored to the unique needs of YAs. Collectively, results from this scoping review add to the existing literature of the use of physical activity among cancer survivors by addressing the YA population. In addition, it highlights the need for follow-up with key stakeholders in YA survivorship and physical activity to gain insight into the state of the science and next steps in policy, clinical, and research endeavors on physical activity as a symptom management strategy in YA cancer survivors.
The authors gratefully acknowledge health sciences librarian Ellen Lutz, BS, MLS, for her input and advice during the search strategy.
About the Author(s)
Ann Marie Moraitis, PhD, MS, RN, is a research assistant, Memnun Seven, PhD, RN, is an assistant professor, and Rachel K. Walker, PhD, RN, FAAN, is an associate professor and PhD program director, all in the College of Nursing at the University of Massachusetts Amherst. This work was partially funded by a grant from the Beta Zeta at-Large Chapter of Sigma. Moraitis and Walker contributed to the conceptualization and design. Moraitis completed the data collection and analysis. All authors contributed to the manuscript preparation. Moraitis can be reached at amoraitis@umass.edu, with copy to ONFEditor@ons.org. (Submitted June 2020. Accepted September 21, 2020.)

