Identifying Barriers to Advocacy Among Patients With Lung Cancer: The Role of Stigma-Related Interpersonal Constraint
Objectives: To examine whether aspects of disease-specific stigma are barriers to advocacy among individuals with lung cancer.
Sample & Setting: 266 patients with lung cancer treated at two National Cancer Institute–designated comprehensive cancer centers in the United States.
Methods & Variables: Patients completed a cross-sectional survey. Demographic, clinical, and stigma-related correlates of advocacy were also explored.
Results: In multivariable analyses, lower levels of patient advocacy were reported by older patients and those with higher levels of constrained disclosure (i.e., avoidance of or discomfort in disclosing one’s lung cancer status).
Implications for Nursing: Avoidance of discussing lung cancer (constrained disclosure) may be a barrier to patient involvement in advocacy. Additional research is needed to examine this relationship, reduce stigma, and promote inclusivity for individuals with lung cancer.
Jump to a section
Patient advocates are crucial voices in advancing health care, research, and policy decisions. Patient advocacy can take many forms, such as supporting a cause, idea, or policy through knowledge acquisition and sharing, personal contact, or organized activities, and is essential for achieving support to make progress against the disease. Although advocacy is a broad term used to describe varied efforts, a growing body of research suggests that various forms of advocacy—from informal individual efforts to acquire and share knowledge to more organized participation in groups to promote awareness and influence policy—can help to facilitate patient benefits, including patient self-efficacy, empowerment, and social support (Brashers et al., 2002; Hagan, Cohen, et al., 2018; Molina et al., 2016; Peterson et al., 2012).
For example, patient advocates have led the way in promoting treatment advances, adequate funding, greater visibility, and increased compassion for individuals with HIV/AIDS (Dresser, 2008). These efforts have been seen in other patient advocacy movements focused on conditions such as breast cancer and Alzheimer disease (Antone et al., 2021; Dresser, 2008; Hollister et al., 2021; Osuch et al., 2012). Disease-specific organizations have emerged as important forums for patient narratives, education, fundraising, and coordinated lobbying efforts (Best, 2019). Smaller-scale self-advocacy efforts have also been shown qualitatively and quantitatively to promote better outcomes for students with disabilities and female cancer survivors by having their needs recognized and met (Hagan, Gilbertson-White, et al., 2018; Test et al., 2005).
Background
Advocacy for Patients With Lung Cancer
As the second most common cancer and leading cause of cancer-related deaths in the United States, lung cancer has a significant disease burden (U.S. Cancer Statistics Working Group, 2017). Patients with lung cancer face numerous unmet needs, including inequitable access and utilization of care (Cheville et al., 2017; Ganti et al., 2017; Kumar et al., 2012; Lim et al., 2015), significant comorbidities (Barbera et al., 2010; Zabora et al., 2001), and quality-of-life impairments (Li & Girgis, 2006). Despite a growing advocacy movement for individuals with lung cancer, the overall impact regarding visibility, funds, and patient outcomes lags behind other diseases. In a comprehensive analysis of the relationship between lost disability-adjusted life years and advocacy outcomes—an index of lobbying expenditures, organization size, and congressional testimony—lung cancer had the smallest advocacy score of any major disease (Best, 2019).
Barriers to Lung Cancer Advocacy
Because of the high mortality rate of lung cancer (overall five-year survival of 22%), there may not be enough long-term survivors to promote lung cancer awareness and advocacy (Siegel et al., 2022). Patients may also not feel well enough to participate in advocacy because of disproportionate symptom burden, poor functional status, and unmet daily living needs (Barbera et al., 2010; Li & Girgis, 2006). In addition, it is possible that stigma attenuates lung cancer advocacy. Patients diagnosed with lung cancer frequently report facing negative social attitudes because of perceptions of the disease as being self-inflicted (i.e., from smoking), with nearly all patients reporting at least one aspect of perceived stigma (Hamann et al., 2014) and about 78% endorsing clinically meaningful levels of stigma (Kaplan et al., 2022; Ostroff et al., 2019). Although tobacco use is the leading risk factor for lung cancer (Siegel et al., 2022; U.S. Department of Health and Human Services, 2014), public health efforts to denormalize smoking and highlight its negative consequences through anti-tobacco campaigns may have also contributed to stigmatizing individuals diagnosed with lung cancer (Riley et al., 2017).
Conceptual Model of Lung Cancer Stigma and Its Potential Influence on Advocacy
Informed by sociologic and social psychological theory (Goffman, 1963; Jones, 1984; Link & Phelan, 2003), lung cancer stigma can be conceptualized as a multifaceted phenomenon, in which patients’ perceive devaluation by others, internalize the experience of negative attitudes and rejection (e.g., shame, regret), and experience interpersonal constraint about disclosing their disease as a result (Hamann et al., 2014, 2017). Among patients with lung cancer, stigma is a notable stressor that is associated with negative psychosocial outcomes, including depression and anxiety (Brown Johnson et al., 2014; Ostroff et al., 2019; Williamson et al., 2018), worse physical functioning and increased symptom severity (Cataldo & Brodsky, 2013; Williamson et al., 2018), decreased relationship satisfaction, increased smoking behavior (Dirkse et al., 2014), delayed medical help-seeking (Carter-Harris et al., 2014; Scott et al., 2015), and worse patient–provider communication (Shen et al., 2016). Within the conceptual model of lung cancer stigma, although advocacy engagement may serve to empower patients and buffer against adverse outcomes, stigma may also be associated with less patient advocacy, possibly through internalized perceptions of decreased value and deserving nature, concern about hurtful responses from others, and limited comfort in disclosing one’s illness (Conlon et al., 2010). These intra- and interpersonal impacts may act as barriers to advocacy, which often require outward-facing disclosure and potential judgment from others. Despite the importance of lung cancer advocacy, to date, there is a paucity of published studies characterizing individual advocacy engagement and no empirical evaluations of the relationship between advocacy and stigma for individuals with lung cancer.
Objectives
This study involved secondary exploratory analyses that were conducted as part of a larger study investigating the concept and correlates of stigma among individuals diagnosed with lung cancer (Hamann et al., 2017). The current analyses, as part of a broader plan to explore correlates of stigma identified in previous qualitative work, aimed to (a) identify demographic and clinical correlates of patient advocacy among individuals with lung cancer and (b) determine the relationship between advocacy and aspects of stigma after accounting for relevant demographic and clinical factors identified in aim 1. The researchers predicted that patient advocacy would be negatively correlated with internalized stigma and interpersonal constraint about disclosing one’s illness because of stigma, but not with perceived stigma. This hypothesis was developed based on previous work suggesting that internalized stigma and constrained disclosure are the active mechanisms for more negative psychosocial and behavioral outcomes (Hamann et al., 2014, 2017; Ostroff et al., 2019; Williamson et al., 2018). Understanding these relationships allows a better understanding of the potential barriers to lung cancer advocacy and provides a potential road map to the benefits of reducing lung cancer stigma.
Methods
Sample and Setting
Data were analyzed from 266 patients with lung cancer who were recruited as part of a multiphase, multisite, cross-sectional evaluation study of the Lung Cancer Stigma Inventory (LCSI). This report represents secondary analyses; sample and study methods are described in greater detail elsewhere (Hamann et al., 2014; Ostroff et al., 2019). The sample size for this study was not determined a priori. Participants were recruited from two outpatient oncology settings associated with the University of Texas Southwestern Medical Center’s Simmons Cancer Center in Dallas, and Memorial Sloan Kettering Cancer Center in New York, New York. Institutional review boards at both locations approved this study. Potential participants were first identified through treating medical oncologists at outpatient clinics and then recruited by trained research staff who confirmed eligibility. Eligible patients were aged 18 years or older; were fluent in English; received a confirmed diagnosis of any stage non-small cell lung cancer or small cell lung cancer; and were either currently undergoing anticancer treatment or had undergone surgical resection, radiation therapy, or chemotherapy within the past 12 months. This time frame was chosen based on qualitative work (Hamann et al., 2014), in which individuals identified stigma as primarily an issue in the active treatment stage, with less relevance post-treatment. Additional efforts were made to ensure that varied smoking histories, particularly for those currently smoking and who had recently quit, were represented in the sample.
All patients provided informed consent prior to study participation. Study participants had the option of completing surveys on paper, on a tablet computer, or through a secure web-based portal on their own computer. All data were entered into the REDCap data management software system.
Measures
Demographics (e.g., race, marital status), smoking history, and clinical information (e.g., time since diagnosis, treatment history) were provided by patient self-report. Disease type and lung cancer stage at diagnosis were determined through self-report and abstraction of electronic health record data. Smoking status was determined by patient responses to the following two questions: “Have you smoked at least 100 cigarettes in your lifetime?” and “Do you smoke cigarettes every day, some days, or not at all?” Participants who reported smoking at least 100 cigarettes and reported currently smoking either “some days” or “every day” were considered to have current tobacco use. Participants who endorsed past lifetime smoking but reported they were not currently smoking (i.e., “not at all”) were categorized as having smoking history, and those who reported smoking fewer than 100 cigarettes in their lifetime were considered to have no smoking history.
Lung cancer advocacy: Although other self-advocacy scales exist (Brashers et al., 1999; Hagan, Cohen, et al., 2018), their validity in populations with lung cancer is not established. Therefore, advocacy was assessed using a three-item Lung Cancer Advocacy Scale based on previous qualitative interviews of patients with lung cancer (Hamann et al., 2014). In this work, a multidimensional representation of advocacy emerged, which included collective action, a desire to help others, and personal/informational sharing. These concepts were incorporated into questions that asked about the degree to which respondents endorsed engaging in each of the following behaviors since being diagnosed with lung cancer: “I have been involved in a group that promotes lung cancer awareness,” “I have wanted to help others who have lung cancer,” and “I have tried to share with others what I know about lung cancer.” Response values for each statement ranged from 1 (not at all) to 5 (extremely). Because this is a new scale measuring a multifaceted construct and was developed based on qualitative work, item-level statistics are reported in addition to scale-level statistics, although the summary scale is used in all multivariable analyses. Responses were summed to generate a total advocacy score, with total possible scores ranging from 3 to 15. This three-item advocacy scale has acceptable internal consistency, with a Cronbach’s alpha of 0.71.
Lung cancer stigma: Lung cancer stigma was assessed using the LCSI (Hamann et al., 2017), a scale focused on perceptions, internalization, and interpersonal manifestations of stigma. The 25-item LCSI consists of the following three moderately correlated subscales: perceived stigma, internalized stigma, and constrained disclosure. The LCSI was used rather than the Cataldo Lung Cancer Stigma Scale (Cataldo et al., 2011) because items were developed through thematic analysis of qualitative patient interviews and refined through multiphasic testing rather than adapted from stigma scales originally developed for use with patients with HIV and other stigmatized populations (Hamann et al., 2014, 2017). The LCSI has high internal consistency (Cronbach’s alpha = 0.89), strong stability of measurement over time, and good convergent validity with related measures, such as the Cataldo Lung Cancer Stigma Scale (Cataldo et al., 2011). Within this sample, the LCSI and each of the subscales demonstrated high internal consistency, with Cronbach’s alphas all higher than 0.78 (overall = 0.9, perceived stigma = 0.78, internalized stigma = 0.91, constrained disclosure = 0.83).
Data Analyses
Demographic and clinical characteristics of the sample were represented using means and standard deviations or frequencies and percentages depending on the variable and type of measurement. The significance level for all statistical tests was set at alpha < 0.05. The rate of missing data for all variables was below thresholds generally considered problematic (Bennett, 2001), and analyses used all available cases. For aim 1, bivariate correlations, independent-samples t tests, and analysis of variance were conducted to examine the associations between variables. The following variables were dichotomized to aid interpretation: lung cancer stage (I–II versus III–IV), smoking status (current tobacco use versus no current tobacco use), marital status (married or partnered versus not married or partnered), and education (high school diploma or less versus some college or more). For aim 2, a series of bivariate correlations among advocacy and lung cancer stigma components were conducted to identify significant relationships to explore with multiple regression. Based on significant relationships found, a hierarchical multiple regression model was used to assess multivariable relationships, including potential interactions. All continuous variables defining interaction terms were mean-centered. All analyses were conducted using IBM SPSS Statistics, version 24.0.
Results
Table 1 presents the demographic and clinical characteristics of all study participants, as well as mean lung cancer advocacy scores. Most participants were female (n = 170), non-Hispanic White (n = 208), and married or living with a partner (n = 162). A large proportion of the sample had an undergraduate degree (n = 87) or higher (n = 46). The majority were diagnosed with non-small cell lung cancer (n = 214) and had stage IV disease (n = 137). Regarding smoking status, 49 patients reported current tobacco use, 153 reported smoking history, and 60 reported no smoking history. Although there was some range in lung cancer advocacy scores across the entire spectrum of possible responses (mean = 7.25, SD = 3.92, range = 3–15), mean scores for each of the advocacy items were relatively low (“somewhat”). Advocacy item 1 (“I have been involved in a group that promotes lung cancer awareness”) was endorsed to a lesser degree than items 2 and 3 (“I have wanted to help others who have lung cancer” and “I have tried to share with others what I know about lung cancer”).
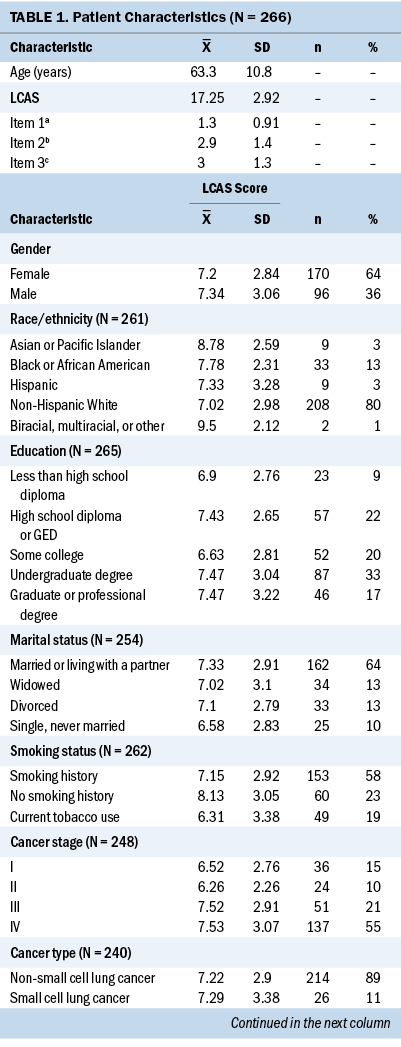
Bivariate analyses between patient-reported advocacy and demographic and clinical characteristics demonstrated higher advocacy scores among patients with stage III–IV lung cancer (t[205] = 2.54, p = 0.01), and patients currently undergoing treatment (t[214.11] = 2.45, p = 0.015). Advocacy scores correlated negatively with age, with younger patients reporting higher advocacy scores (r = –0.18, p = 0.005). Levene’s test indicated unequal variances (F = 4.35, p = 0.04), so degrees of freedom were adjusted from 254 to 214.11. Patients with current tobacco use reported less advocacy than those who reported smoking history or no smoking history (t[87.52] = 2.83, p = 0.006). Levene’s test indicated unequal variances (F = 4.56, p = 0.03), so degrees of freedom were adjusted from 254 to 87.52. No significant differences in advocacy levels were found by patients’ gender (t[256] = 0.37, p = 0.71), race or ethnicity (F[4, 249] = 1.43, p = 0.22), education (t[255] = 0.12, p = 0.9), marital status (t[247] = 0.86, p = 0.39), or lung cancer type (t[208] = –0.14, p = 0.89).
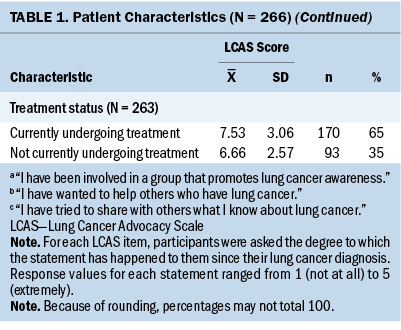
In bivariate analyses focused on advocacy and specific aspects of stigma, only the LCSI constrained disclosure subscale was correlated with patient advocacy; patients who reported higher levels of constrained disclosure related to lung cancer also reported less advocacy (r = –0.2, p = 0.002). Neither the total LCSI score nor the perceived stigma or internalized stigma subscales were significantly associated with patient advocacy (p > 0.05 for all). When the three advocacy items were analyzed separately, item 1 (“I have been involved in a group that promotes lung cancer awareness”) was not associated with constrained disclosure, but items 2 and 3 (“I have wanted to help others who have lung cancer” and “I have tried to share with others what I know about lung cancer”) were negatively correlated with constrained disclosure (see Table 2).
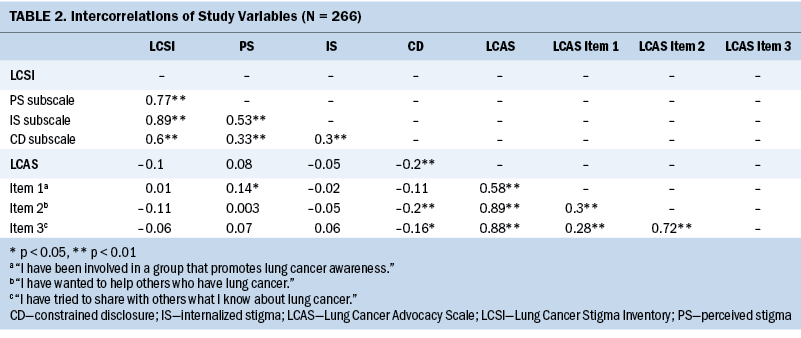
In a hierarchical regression model including significant variables from the prior bivariate analyses (age, stage, treatment status, smoking status, and constrained disclosure), the LCSI constrained disclosure subscale and age were associated with patient advocacy. None of the other demographic or clinical variables, including smoking status, remained significantly correlated with advocacy. Adding an interaction term between constrained disclosure and smoking status did not significantly improve the model, which was overall significant and explained 11% of the variance in patient advocacy (R = 0.36, adjusted R2 = 0.11, F[5, 224] = 6.43, p < 0.001) (see Table 3).
Discussion
This study focused on understanding the potential role of stigma as a barrier to advocacy among patients with lung cancer. Results indicated a negative relationship between patient advocacy and the LCSI constrained disclosure subscale, which remained after controlling for other potentially explanatory variables. Although the cross-sectional nature of the survey limits directional interpretations, the results suggest that certain aspects of stigma may limit involvement in lung cancer advocacy through an interpersonal pathway that impedes outward-facing activism. Patients who experience constrained disclosure are reluctant to share that they have been diagnosed with lung cancer, and this discomfort may contribute to being less likely to help others with lung cancer or share information about lung cancer with others. Although a more nuanced assessment of advocacy is needed to draw strong conclusions, constrained disclosure may decrease patients’ willingness to mobilize, get involved with advocacy groups, and discuss their illness with policymakers or in other large forums. This relationship may also be bidirectional in that reduced advocacy may further isolate patients, limit their communication, and hinder social support. A previous qualitative study of advocacy in the context of cancer care highlighted the fundamental need for trust in one’s healthcare provider to effectively communicate and self-advocate, which, in turn, has the potential to strengthen the patient–provider relationship (Hagan & Medberry, 2016). Although direct quantitative comparisons cannot be made given variability in samples and measurement methods, the extent to which patients in the current sample reported engaging in advocacy (particularly group participation) appears lower than in other samples, including female cancer survivors (Hagan, Gilbertson-White, et al., 2018) and people living with HIV/AIDS (Brashers et al., 2002). This aligns with previous findings that lung cancer advocacy lags behind other conditions (Best, 2019). Overall, these results suggest that constrained disclosure of lung cancer status that may emanate in a small interpersonal circle can have wide-ranging implications because it affects public awareness and attention.
Of note, other components of stigma, including perceptions of devaluation from others (perceived stigma) and negative affective/cognitive intrapersonal responses (internalized stigma), were not significantly associated with patient advocacy. Increased evidence is emerging about the potentially distinct pathways connecting internalized stigma with more intrapersonal manifestations of psychological distress (e.g., anxiety, depression) (Williamson et al., 2020) and those linking constrained disclosure with more interpersonal manifestations (e.g., limited advocacy, impaired patient–provider communication) (Shen et al., 2016). In general, these results illustrate the multifaceted nature of lung cancer stigma and the need to better understand and address these pathways and processes.
In addition, older patients reported lower levels of advocacy engagement in bivariate and multivariable analyses. This finding aligns with other literature demonstrating limited representation of older patients in cancer clinical research broadly, as well as the barriers to increased advocacy and engagement faced by this population, such as challenges accessing technology and limited health literacy (Gilmore et al., 2019). In previous studies (Rigney et al., 2021; Rose et al., 2018), younger patients with lung cancer reported greater perceived stigma. This is perhaps because of greater exposure to public health campaigns denormalizing smoking, differences in global health and activation, or a greater likelihood of never-smoking history. Therefore, it is possible that older patients with lung cancer may be less motivated to engage in advocacy because of lesser perceived need, although there was no difference in any aspect of stigma by age in the current sample (perceived stigma, internalized stigma, and constrained disclosure).
Given the robust success of patient advocacy for other conditions, increased lung cancer advocacy may reduce the barrier of stigma in this patient population. Previous studies have noted that advocacy movements benefit from patient narratives, broaden the public’s understanding of causal factors, and demonstrate shared emotions and aspirations between patient and nonpatient groups (Kreuter et al., 2007). These techniques may also serve to encourage reluctant and interpersonally constrained patients to participate in activism by showing that others share their experiences, concerns, and humanity. Lung cancer advocacy is likely hampered by anti-tobacco public health campaigns that aim to increase fear by connecting tobacco use with lung cancer in graphic detail (Riley et al., 2017). Lung cancer advocates focused on humanizing the disease may face the barrier of other messaging that inadvertently dehumanizes individuals with lung cancer. Patients in the current study with current tobacco use were less likely to endorse high levels of patient advocacy in bivariate analyses. Advocacy organizations play important roles in education, supportive care, and policy change. Although greater symptom burden, worse functional status, and higher mortality rates (American Lung Association, 2014; Lehto, 2016) have traditionally limited advocacy engagement for individuals with lung cancer, treatment advances and gains in survival have allowed for a growing population of survivors who will more closely resemble survivors of other cancers, such as breast or colorectal (Howlader et al., 2020). In bivariate analyses, patients with more advanced disease and current treatment status reported greater advocacy engagement, suggesting that symptom burden and disease severity were not associated with advocacy in this sample. Lung cancer advocacy organizations can continue to make concerted inclusive efforts to engage patient advocates who represent the diverse community of patients with lung cancer. Particular attention is needed to create environments where patients feel comfortable sharing their smoking status without fear of shame or blame. Continued discussion among lung cancer advocates and public health specialists can help to develop messaging that maximizes tobacco control while minimizing stigma faced by patients with lung cancer. Further research is needed to better understand the unique barriers to advocacy among traditionally disenfranchised groups and promote efforts at inclusivity.
Limitations
Although this study provides valuable insight into patient advocacy among individuals with lung cancer, the results must be considered within the context of the study’s limitations. In addition to the limits of cross-sectional data in considering directional pathways, these analyses are also limited by the measurement of lung cancer advocacy. Although the measure was developed based on patient-reported qualitative data, it consisted of only three items reflecting a broad intent to help others with lung cancer, which may limit data interpretation. A single three-item measure is likely an imprecise way to quantify a multifaceted construct such as advocacy. Additional research is needed to more precisely define the different kinds of lung cancer advocacy that may be affected by stigma and to assess the reliability and validity of this scale.
In the current study, the model predicted only 11% of the variance in lung cancer advocacy, indicating that other unmeasured factors may be important in understanding barriers to advocacy engagement among individuals with lung cancer. Future studies should investigate other potential barriers to advocacy, some of which have been identified in previous research, such as health-related quality of life (e.g., anxiety, depression, distress, functional status, symptom burden); personal characteristics (e.g., openness, drive); communication tendencies, skills, and coping styles; and social inequities (e.g., rurality, internet access, socioeconomic status, accessibility and quality of social support, financial toxicity) (Brashers et al., 2002; Hagan & Donovan, 2013; Hagan, Gilbertson-White, et al., 2018). In addition, enrollment was restricted to individuals within 12 months of treatment. It is likely that many individuals become advocates further out from diagnosis when symptom burden and functional status may improve. Future advocacy research should focus on survivors further out from treatment.
Lastly, limited representation of individuals with current tobacco use and those from underserved racial and ethnic backgrounds hinders the ability to draw strong conclusions about differences in advocacy participation among populations who may face additional adversity and bias in health care. The limited diversity of the current sample is consistent with existing work demonstrating less frequent participation of these groups in cancer research (Aldrighetti et al., 2021) and evidencing numerous inequities in access to care (Coughlin et al., 2014; Ryan, 2018). Men were also underrepresented in this sample, perhaps because of worse survival compared to women with lung cancer (Siegel et al., 2022). Because this sample included only individuals willing to participate in research and trust is a relevant factor for research participation, it is possible that stigma might be lower and advocacy might be higher in this population than in the general population of patients with lung cancer.
Implications for Nursing
As trusted healthcare providers who are continuously engaged in advocacy efforts to enhance patient-centered cancer care, oncology nurses are important allies in advancing lung cancer advocacy efforts. Patient advocacy is a modifiable factor that can be encouraged and facilitated on an individual level by nursing professionals (Hagan, Gilbertson-White, et al., 2018) through modeling and supporting open communication regarding patients’ specific needs and symptom burden concerns. Lung cancer advocates and advocacy organizations should continue making concerted inclusive efforts to engage individuals who represent patients with lung cancer. Oncology nurses can seek to create environments where individuals feel comfortable sharing their smoking status without shame or blame. Because interactions with healthcare clinicians are a common source of stigma, oncology nurses play a critical role in mitigating lung cancer stigma and its negative consequences. Patients’ expressions of negative emotions related to lung cancer (e.g., shame, guilt, regret) represent opportunities for empathic communication, which are frequently missed or mishandled (Banerjee, Haque, Bylund, et al., 2021; Morse et al., 2008). By recognizing patients’ emotions and experiences and responding with understanding, oncology nurses can reduce stigma and build trust. Although these skills are familiar to many, they are often underused and can be cultivated through training (Banerjee, Haque, Bylund, et al., 2021; Banerjee, Haque, Schofield, et al., 2021).

Conclusion
For patients with lung cancer, reluctance to or discomfort in disclosing their diagnosis may present a barrier to engaging in lung cancer advocacy. This relationship may be bidirectional in that reduced advocacy may hinder social engagement and communication. Overall, the current analysis adds to the growing understanding of the negative impacts of lung cancer stigma and highlights the need to reduce stigma at multiple levels of intervention. Coordinated application of evidence-based strategies are crucial to reduce stigma associated with lung cancer. Further research is needed to better understand the unique barriers to patient advocacy among patients with lung cancer from traditionally disenfranchised groups and to promote efforts at inclusivity.
The authors gratefully acknowledge all study participants for their valuable contributions to this work.
About the Authors
Sarah N. Price, PhD, is a postdoctoral fellow in the Department of Social Sciences and Health Policy in the School of Medicine at Wake Forest University in Winston-Salem, NC; Megan Shen, PhD, is an associate professor in the Clinical Research Division at the Fred Hutchinson Cancer Center in Seattle, WA; Maureen Rigney, LICSW, is the senior director of support initiatives at the GO2 Foundation for Lung Cancer in Washington, DC; Jamie S. Ostroff, PhD, is the chief of behavioral sciences service at Memorial Sloan Kettering Cancer Center in New York, NY; and Heidi A. Hamann, PhD, is an associate professor in the Department of Psychology at the University of Arizona in Tuscon. This work was supported, in part, by grant awards from the National Cancer Institute (T32CA122061, Price; R03CA154016, Hamann; R03193986, Shen; T32CA009461, Ostroff), the National Lung Cancer Partnership and its North Carolina affiliate (Hamann), and the Lung Cancer Research Foundation (Ostroff), as well as a National Institutes of Health Support Grant (P30CA008748). Price, Ostroff, and Hamann contributed to the conceptualization and design. Ostroff and Harmann completed the data collection. Price provided statistical support and analysis. All authors contributed to the manuscript preparation. Price can be reached at sarahpr@wakehealth.edu, with copy to ONFEditor@ons.org. (Submitted December 2021. Accepted March 20, 2022.)
References
Aldrighetti, C.M., Niemierko, A., Van Allen, E., Willers, H., & Kamran, S.C. (2021). Racial and ethnic disparities among participants in precision oncology clinical studies. JAMA Network Open, 4(11), e2133205. https://doi.org/10.1001/JAMANETWORKOPEN.2021.33205
American Lung Association. (2014). Addressing the stigma of lung cancer. https://www.lung.org/getmedia/54eb0979-4272-4f43-be79-9bef36fab30b/ala-…
Antone, N., Kizub, D., Gralow, J., Zujewski, J.A., & Dvaladze, A. (2021). Advancing breast cancer advocacy in eastern Europe and central Asia: Findings from Women’s Empowerment Cancer Advocacy Network (WE CAN) Summits. JCO Global Oncology, 7(2021), 585–592. https://doi.org/10.1200/GO.20.00532
Banerjee, S.C., Haque, N., Bylund, C.L., Shen, M.J., Rigney, M., Hamann, H.A., . . . Ostroff, J.S. (2021). Responding empathically to patients: A communication skills training module to reduce lung cancer stigma. Translational Behavioral Medicine, 11(2), 613–618. https://doi.org/10.1093/tbm/ibaa011
Banerjee, S.C., Haque, N., Schofield, E.A., Williamson, T.J., Martin, C.M., Bylund, C.L., . . . Ostroff, J.S. (2021). Oncology care provider training in empathic communication skills to reduce lung cancer stigma. Chest, 159(5), 2040–2049. https://doi.org/10.1016/J.CHEST.2020.11.024
Barbera, L., Seow, H., Howell, D., Sutradhar, R., Earle, C., Liu, Y., . . . Dudgeon, D. (2010). Symptom burden and performance status in a population-based cohort of ambulatory cancer patients. Cancer, 116(24), 5767–5776. https://doi.org/10.1002/cncr.25681
Bennett, D.A. (2001). How can I deal with missing data in my study? Australian and New Zealand Journal of Public Health, 25(5), 464–469. https://doi.org/10.1111/J.1467-842X.2001.TB00294.X
Best, R.K. (2019). Common enemies. Oxford University Press. https://doi.org/10.1093/oso/9780190918408.001.0001
Brashers, D.E., Haas, S.M., & Neidig, J.L. (1999). The Patient Self-Advocacy Scale: Measuring patient involvement in health care decision-making interactions. Health Communication, 11(2), 97–121. https://doi.org/10.1207/s15327027hc1102_1
Brashers, D.E., Haas, S.M., Neidig, J.L., & Rintamaki, L.S. (2002). Social activism, self-advocacy, and coping with HIV illness. Journal of Social and Personal Relationships, 19(1), 113–133. https://doi.org/10.1177/0265407502191006
Brown Johnson, C.G., Brodsky, J.L., & Cataldo, J.K. (2014). Lung cancer stigma, anxiety, depression, and quality of life. Journal of Psychosocial Oncology, 32(1), 59–73. https://doi.org/10.1080/07347332.2013.855963
Carter-Harris, L., Hermann, C.P., Schreiber, J., Weaver, M.T., & Rawl, S.M. (2014). Lung cancer stigma predicts timing of medical help-seeking behavior. Oncology Nursing Forum, 41(3), E203–E210. https://doi.org/10.1188/14.ONF.E203-E210
Cataldo, J.K., & Brodsky, J.L. (2013). Lung cancer stigma, anxiety, depression and symptom severity. Oncology, 85(1), 33–40. https://doi.org/10.1159/000350834
Cataldo, J.K., Slaughter, R., Jahan, T.M., Pongquan, V.L., & Hwang, W.J. (2011). Measuring stigma in people with lung cancer: Psychometric testing of the Cataldo Lung Cancer Stigma Scale. Oncology Nursing Forum, 38(1), E46–E54. https://doi.org/10.1188/11.ONF.E46-E54
Cheville, A.L., Rhudy, L., Basford, J.R., Griffin, J.M., & Flores, A.M. (2017). How receptive are patients with late stage cancer to rehabilitation services and what are the sources of their resistance? Archives of Physical Medicine and Rehabilitation, 98(2), 203–210. https://doi.org/10.1016/j.apmr.2016.08.459
Conlon, A., Gilbert, D., Jones, B., & Aldredge, P. (2010). Stacked stigma: Oncology social workers’ perceptions of the lung cancer experience. Journal of Psychosocial Oncology, 28(1), 98–115. https://doi.org/10.1080/07347330903438982
Coughlin, S.S., Matthews-Juarez, P., Juarez, P.D., Melton, C.E., & King, M. (2014). Opportunities to address lung cancer disparities among African Americans. Cancer Medicine, 3(6), 1467–1476. https://doi.org/10.1002/cam4.348
Dirkse, D., Lamont, L., Li, Y., Simonič, A., Bebb, G., & Giese-Davis, J. (2014). Shame, guilt, and communication in lung cancer patients and their partners. Current Oncology, 21(5), e718–e722. https://doi.org/10.3747/co.21.2034
Dresser, R. (2008). The role of patient advocates and public representatives in research. In E.J. Emanuel, C. Grady, R.A. Crouch, R.K. Lie, F.G. Miller, & D. Wendler (Eds.), Oxford textbook of clinical research ethics (pp. 231–241). Oxford University Press.
Ganti, A.K., Hirsch, F.R., Wynes, M.W., Ravelo, A., Ramalingam, S.S., Ionescu-Ittu, R., . . . Borghaei, H. (2017). Access to cancer specialist care and treatment in patients with advanced stage lung cancer. Clinical Lung Cancer, 18(6), 640–650.e2. https://doi.org/10.1016/j.cllc.2017.04.010
Gilmore, N.J., Canin, B., Whitehead, M., Sedenquist, M., Griggs, L., Finch, L., . . . Mohile, S.G. (2019). Engaging older patients with cancer and their caregivers as partners in cancer research. Cancer, 125(23), 4124–4133. https://doi.org/10.1002/CNCR.32402
Goffman, E. (1963). Stigma: Notes on the management of spoiled identity. Penguin Books.
Hagan, T.L., Cohen, S.M., Rosenzweig, M.Q., Zorn, K., Stone, C.A., & Donovan, H.S. (2018). The Female Self-Advocacy in Cancer Survivorship Scale: A validation study. Journal of Advanced Nursing, 74(4), 976–987. https://doi.org/10.1111/jan.13498
Hagan, T.L., & Donovan, H.S. (2013). Self-advocacy and cancer: A concept analysis. Journal of Advanced Nursing, 69(10), 2348–2359. https://doi.org/10.1111/JAN.12084
Hagan, T.L., Gilbertson-White, S., Cohen, S.M., Temel, J.S., Greer, J.A., & Donovan, H.S. (2018). Symptom burden and self-advocacy: Exploring the relationship among female cancer survivors. Clinical Journal of Oncology Nursing, 22(1), E23–E30. https://doi.org/10.1188/18.CJON.E23-E30
Hagan, T.L., & Medberry, E. (2016). Patient education vs. patient experiences of self-advocacy: Changing the discourse to support cancer survivors. Journal of Cancer Education, 31(2), 375–381. https://doi.org/10.1007/s13187-015-0828-x
Hamann, H.A., Ostroff, J.S., Marks, E.G., Gerber, D.E., Schiller, J.H., & Craddock Lee, S.J. (2014). Stigma among patients with lung cancer: A patient-reported measurement model. Psycho-Oncology, 23(1), 81–92. https://doi.org/10.1002/pon.3371
Hamann, H.A., Shen, M.J., Thomas, A.J., Craddock Lee, S.J., & Ostroff, J.S. (2017). Development and preliminary psychometric evaluation of a patient-reported outcome measure for lung cancer stigma: The Lung Cancer Stigma Inventory (LCSI). Stigma and Health, 3(3), 195–203. https://doi.org/10.1037/sah0000089
Hollister, B.A., Yeh, J., Ross, L., Schlesinger, J., & Cherry, D. (2021). Building an advocacy model to improve the dementia-capability of health plans in California. Journal of the American Geriatric Society, 69(12), 3641–3649. https://doi.org/10.1111/jgs.17429
Howlader, N., Forjaz, G., Mooradian, M.J., Meza, R., Kong, C.Y., Cronin, K.A., . . . Feuer, E.J. (2020). The effect of advances in lung-cancer treatment on population mortality. New England Journal of Medicine, 383(7), 640–649. https://doi.org/10.1056/NEJMOA1916623
Jones, E.E. (1984). Social stigma: The psychology of marked relationships. W.H. Freeman.
Kaplan, D.M., Hamann, H.A., Price, S.N., Williamson, T.J., Ver Hoeve, E.S., McConnell, M.H., . . . Ostroff, J.S. (2022). Developing an ACT-based intervention to address lung cancer stigma: Stakeholder recommendations and feasibility testing in two NCI-designated cancer centers. Journal of Psychosocial Oncology, 6, 1–17. https://doi.org/10.1080/07347332.2022.2033377
Kreuter, M.W., Green, M.C., Cappella, J.N., Slater, M.D., Wise, M.E., Storey, D., . . . Woolley, S. (2007). Narrative communication in cancer prevention and control: A framework to guide research and application. Annals of Behavioral Medicine, 33(3), 221–235. https://doi.org/10.1007/BF02879904
Kumar, P., Casarett, D., Corcoran, A., Desai, K., Li, Q., Chen, J., . . . Mao, J.J. (2012). Utilization of supportive and palliative care services among oncology outpatients at one academic cancer center: Determinants of use and barriers to access. Journal of Palliative Medicine, 15(8), 923–930. https://doi.org/10.1089/jpm.2011.0217
Lehto, R.H. (2016). Symptom burden in lung cancer: Management updates. Lung Cancer Management, 5(2), 61–78. https://doi.org/10.2217/LMT-2016-0001
Li, J., & Girgis, A. (2006). Supportive care needs: Are patients with lung cancer a neglected population? Psycho-Oncology, 15(6), 509–516. https://doi.org/10.1002/pon.983
Lim, C., Tsao, M., Le, L., Shepherd, F., Feld, R., Burkes, R., . . . Leighl, N. (2015). Biomarker testing and time to treatment decision in patients with advanced nonsmall-cell lung cancer. Annals of Oncology, 26(7), 1415–1421. https://doi.org/10.1093/annonc/mdv208
Link, B.G., & Phelan, J.C. (2003). Conceptualizing stigma. Annual Review of Sociology, 27(1), 363–385. https://doi.org/10.1146/ANNUREV.SOC.27.1.363
Molina, Y., Scherman, A., Constant, T.H., Hempstead, B., Thompson-Dodd, J., Richardson, S., . . . Ceballos, R.M. (2016). Medical advocacy among African-American women diagnosed with breast cancer: From recipient to resource. Supportive Care in Cancer, 24(7), 3077–3084. https://doi.org/10.1007/s00520-016-3123-4
Morse, D.S., Edwardsen, E.A., & Gordon, H.S. (2008). Missed opportunities for interval empathy in lung cancer communication. Archives of Internal Medicine, 168(17), 1853–1858. https://doi.org/10.1001/ARCHINTE.168.17.1853
Ostroff, J.S., Riley, K.E., Shen, M.J., Atkinson, T.M., Williamson, T.J., & Hamann, H.A. (2019). Lung cancer stigma and depression: Validation of the Lung Cancer Stigma Inventory. Psycho-Oncology, 28(5), 1011–1017. https://doi.org/10.1002/pon.5033
Osuch, J.R., Silk, K., Price, C., Barlow, J., Miller, K., Hernick, A., & Fonfa, A. (2012). A historical perspective on breast cancer activism in the United States: From education and support to partnership in scientific research. Journal of Women’s Health, 21(3), 355–362. https://doi.org/10.1089/jwh.2011.2862
Peterson, J.L., Rintamaki, L.S., Brashers, D.E., Goldsmith, D.J., & Neidig, J.L. (2012). The forms and functions of peer social support for people living with HIV. Journal of the Association of Nurses in AIDS Care, 23(4), 294–305. https://doi.org/10.1016/j.jana.2011.08.014
Rigney, M., Rapsomaniki, E., Carter-Harris, L., & King, J.C. (2021). A 10-year cross-sectional analysis of public, oncologist, and patient attitudes about lung cancer and associated stigma. Journal of Thoracic Oncology, 16(1), 151–155. https://doi.org/10.1016/J.JTHO.2020.09.011
Riley, K.E., Ulrich, M.R., Hamann, H.A., & Ostroff, J.S. (2017). Decreasing smoking but increasing stigma? Anti-tobacco campaigns, public health, and cancer care. AMA Journal of Ethics, 19(5), 475–485. https://doi.org/10.1001/journalofethics.2017.19.5.msoc1-1705
Rose, S., Kelly, B., Boyes, A., Cox, M., Palazzi, K., & Paul, C. (2018). Impact of perceived stigma in people newly diagnosed with lung cancer: A cross-sectional analysis. Oncology Nursing Forum, 45(6), 737–747. https://doi.org/10.1188/18.ONF.737-747
Ryan, B.M. (2018). Lung cancer health disparities. Carcinogenesis, 39(6), 741–751. https://doi.org/10.1093/carcin/bgy047
Scott, N., Crane, M., Lafontaine, M., Seale, H., & Currow, D. (2015). Stigma as a barrier to diagnosis of lung cancer: Patient and general practitioner perspectives. Primary Health Care Research and Development, 16(6), 618–622. https://doi.org/10.1017/S1463423615000043
Shen, M.J., Hamann, H.A., Thomas, A.J., & Ostroff, J.S. (2016). Association between patient–provider communication and lung cancer stigma. Supportive Care in Cancer, 24(5), 2093–2099. https://doi.org/10.1007/s00520-015-3014-0
Siegel, R.L., Miller, K.D., & Jemal, A. (2022). Cancer statistics, 2022. CA: A Cancer Journal for Clinicians, 72(1), 7–33. https://doi.org/10.3322/caac.21708
Test, D.W., Fowler, C.H., Wood, W.M., Brewer, D.M., & Eddy, S. (2005). A conceptual framework of self-advocacy for students with disabilities. Remedial and Special Education, 26(1), 43–54. https://doi.org/10.1177/07419325050260010601
U.S. Cancer Statistics Working Group. (2017). United States cancer statistics: 1999–2014 incidence and mortality web-based report. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Cancer Institute. https://apha.confex.com/apha/2017/mediafile/Handout/Paper385940/USCS_Fa…
U.S. Department of Health and Human Services. (2014). The health consequences of smoking: 50 years of progress. A report of the Surgeon General. Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health. https://www.ncbi.nlm.nih.gov/books/NBK179276
Williamson, T.J., Choi, A.K., Kim, J.C., Garon, E.B., Shapiro, J.R., Irwin, M.R., . . . Stanton, A.L. (2018). A longitudinal investigation of internalized stigma, constrained disclosure, and quality of life across 12 weeks in lung cancer patients on active oncologic treatment. Journal of Thoracic Oncology, 13(9), 1284–1293. https://doi.org/10.1016/j.jtho.2018.06.018
Williamson, T.J., Kwon, D.M., Riley, K.E., Shen, M.J., Hamann, H.A., & Ostroff, J.S. (2020). Lung cancer stigma: Does smoking history matter? Annals of Behavioral Medicine, 54(7), 535–540. https://doi.org/10.1093/abm/kaz063
Zabora, J., Brintzenhofeszoc, K., Curbow, B., Hooker, C., & Piantadosi, S. (2001). The prevalence of psychological distress by cancer site. Psycho-Oncology, 10(1), 19–28. https://doi.org/10.1002/1099-1611(200101/02)10:1<19::AID-PON501>3.0.CO;…

