Alterations and Interdependence in Self-Reported Sleep-Wake Parameters of Patient–Caregiver Dyads During Adjuvant Chemotherapy for Breast Cancer
Purpose/Objectives: To longitudinally explore changes, similarities, differences, and interrelations in the sleep-wake parameters of patient–caregiver dyads throughout adjuvant chemotherapy for breast cancer.
Design: Observational, repeated-measures, dyadic study.
Setting: Four ambulatory oncology clinics in Scotland.
Sample: 48 dyads consisting of patients and their primary informal caregivers.
Methods: Four dyadic, self-reported sleep-wake assessments took place before chemo-therapy (T0), during chemotherapy cycles 1 (T1) and 4 (T2), and after chemotherapy (T3). Dyads completed the Pittsburgh Sleep Quality Index. Multilevel hierarchical linear modeling was used to explore dyadic data.
Main Research Variables: Perceived sleep quality, sleep onset latency (SOL), total sleep time, habitual sleep efficiency, wake after sleep onset, daily disturbance, daytime napping duration, overall sleep-wake impairment.
Findings: The majority of dyads had at least one poor sleeper throughout the study; 25%–35% were dyads of concurrent poor sleepers. Curvilinear patterns of change were evident for patients’ (but not caregivers’) sleep-wake parameters, steadily deteriorating from pre- to midtreatment, then leveling off close to baseline. Average trajectories were significantly different between the dyad members but indicative of a trend for concurrent deterioration at T2. Dyad members’ perceived sleep quality, SOL, and overall sleep-wake impairment were closely interrelated; wake variables remained uncoupled.
Conclusions: Despite overall differences in magnitude, sleep problems may be concurrently present in both dyad members, covary, and peak midway through chemotherapy.
Implications for Nursing: Dyadic sleep assessments can shed light on potential areas of sleep interaction to enable interventions to support care dyads at risk of sleep distress during chemotherapy for breast cancer.
Jump to a section
Women with early-stage breast cancer are susceptible to sleep impairment and daytime dysfunction (Fiorentino & Ancoli-Israel, 2006), particularly those who receive neo-/adjuvant chemotherapy to minimize the risk of cancer recurrence (Costa et al., 2014; Kotronoulas, Wengstrom, & Kearney, 2012). Deficits in perceived sleep quality, sleep onset latency (SOL), nocturnal awakenings, and total sleep time (TST) may be present even before chemotherapy initiation, become exacerbated over treatment continuation, and persist after the last chemotherapy cycle (Ancoli-Israel et al., 2014; Berger, Wielgus, Hertzog, Fischer, & Farr, 2010; Savard et al., 2009; Savard, Ivers, Savard, & Morin, 2015; Van Onselen et al., 2013). Sleep-wake deficits in informal caregivers of patients with cancer are also prominent; at least 4 of 10 informal caregivers may report at least one sleep problem (Kotronoulas, Wengstrom, & Kearney, 2013b; Stenberg, Cvancarova, Ekstedt, Olsson, & Ruland, 2014). Diminished TST, nocturnal awakenings, prolonged wake after sleep onset (WASO), and daytime dysfunction seem to be the areas of greatest distress (Berger et al., 2005; Kotronoulas et al., 2013b). Sleep-related distress that becomes chronic may be associated with persistent fatigue, depressed mood, altered immune function, altered metabolism, and impaired memory and problem solving, collectively leading to poor functional and social well-being (Bryant, Trinder, & Curtis, 2004; Ho, Rohan, Parent, Tager, & McKinley, 2015; Liu et al., 2013; Pawl, Lee, Clark, & Sherwood, 2013; Shilling & Jenkins, 2007; Stenberg et al., 2014). When patients and caregivers experience sleep-wake problems concurrently, these adverse effects may intensify, affecting relationships and complicating joint coping with the illness (Kotronoulas, Wengstrom, & Kearney, 2013a).
Research supports the fundamental idea of dyadic interdependency in cancer care (Dorros, Card, Segrin, & Badger, 2010; Ussher, Tim Wong, & Perz, 2011). At least two theoretical models exist that propose a link between close relationships and sleep regulation. According to the attachment theory (Bowlby, 1978), interactions with caregivers and/or romantic partners generate expectations regarding the degree to which the caregiver or partner will be responsive to one’s needs. These expectations may be critical in regulating affect and arousal within close relationships, particularly because sleep is achieved when vigilance is down-regulated, a process that can be optimized when a person feels physically and emotionally safe and secure (Adams, Stoops, & Skomro, 2014; Troxel, 2010). Related to this, Crossley’s (2004) sleep ritual theory suggests that, in close relationships, both parties need to secure cooperation from one another for individual sleep needs and preferences and those of the composite relationship to be achieved. Drawing upon these theoretical frameworks, it can be argued that, in closely living people, such as patients and their informal caregivers, poor sleep may be simultaneously experienced, co-affected and co-regulated (Kotronoulas et al., 2013a). Considering the perspectives of patient and caregiver can shed light on dyadic processes that influence dyad members’ sleep-wake patterns during the natural course of caregiving, facilitate the investigation of latent interactions, and highlight potential areas that may enhance implementation and effectiveness of sleep interventions (Kotronoulas et al., 2013a; Lyons & Sayer, 2005). Despite these important implications, to date, dyadic sleep research in the context of adult cancer care remains scarce (Kotronoulas et al., 2013a).
Therefore, the authors’ aim was to longitudinally explore patterns of change, similarities and differences, and interrelations in subjective sleep-wake parameters of patient–caregiver dyads throughout adjuvant chemotherapy for early-stage breast cancer. The following research questions were posed:
• What are the trajectories of change in patients’ and caregivers’ sleep-wake parameters throughout adjuvant chemotherapy for breast cancer?
• How similar or dissimilar are patients’ and caregivers’ sleep-wake trajectories?
• Which sleep-wake parameters show the greatest interdependence across the dyads?
Methods
The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement guided reporting of this study (Vandenbroucke et al., 2014). The STROBE Statement consists of a checklist of 22 items and recommendations to provide guidance to authors about how to improve the reporting of observational studies while facilitating critical appraisal and interpretation of studies by reviewers, journal editors, and readers (Vandenbroucke et al., 2014). The Tayside Committee on Medical Research Ethics A approved the study (10/S1401/41), which was undertaken in accordance with the guidelines of the Declaration of Helsinki (World Medical Association, 2008).
Design, Participants, and Setting
An observational, repeated-measures, dyadic study was conducted at four ambulatory oncology clinics in Scotland across four NHS boards (Tayside, Grampian, Fife, and Forth Valley). A convenience sample of newly diagnosed women with early-stage breast cancer and their primary informal caregivers were possible candidates for participation.
Eligible patients were women who were recently diagnosed with clinical stage I–IIIA breast cancer, were at least two weeks after breast cancer surgery, had no previous cancer diagnosis or administration of chemotherapy, and were scheduled to receive six or more cycles of adjuvant chemotherapy. Patients were not withdrawn because of changes in chemotherapy doses or regimens, but any treatment modifications were monitored to allow seamless participation in the study. Eligible primary informal caregivers were patient-nominated people expected to offer greatest physical and emotional care and support during the treatment period. Patients and caregivers were aged 18 years or older, had adequate knowledge of English and a satisfactory level of communication, and were able to provide written informed consent.
Patients or caregivers with a previously diagnosed primary sleep disorder (e.g., narcolepsy, sleep apnea) and/or cognitive, mental, or psychiatric impairment were excluded, as were patients with locally advanced or metastatic disease. Patients scheduled to receive neoadjuvant chemotherapy were also excluded on the premise that baseline variability in the degree of sleep-related distress (breast cancer surgery as an extra stressor for women on adjuvant chemotherapy; women on neoadjuvant chemotherapy still very close to cancer diagnosis) would reduce homogeneity of the patient group.
Procedures
Assessments took place at four distinct time points throughout chemotherapy, in line with previous research and clinical expertise. Patients were assessed about one week before initiation of chemotherapy (T0), within two weeks after initiation of the first cycle of chemotherapy (T1), within two weeks after initiation of the fourth cycle of chemotherapy (T2), and about three weeks after initiation of the final cycle of chemotherapy (i.e., end of treatment) (T3). During consultation appointments, local site collaborators checked patients’ eligibility and invited them to participate and nominate their primary informal caregivers, who were also invited to participate in the study. Patients and caregivers were contacted after no less than 24 hours, at which point they were briefed on the study’s purpose, the voluntary nature of their participation, and their liberty to withdraw at any time. Written, informed consent was obtained from all patients and caregivers.
At each assessment point, patient and caregiver case-report forms containing the set of self-reported questionnaires and forms were mailed to dyads at their home addresses. Patients and caregivers were given adequate time to complete the questionnaires, instructed to refrain from sharing their responses with each other, and asked to return completed case-report forms via prepaid mail. Brief, informal reminders were sent to all participants throughout their involvement to prevent missing data from nonresponse. Depending on the duration of chemotherapy protocol (6 or more cycles) and cycle intervals (2–4 weeks), patient participation in the study ranged from 14–24 weeks.
Measures
The Pittsburgh Sleep Quality Index (PSQI) consists of four open-ended and 19 multiple-choice questions that generate seven component scores: sleep quality (C1), SOL (C2), sleep duration (C3), HSE (C4), sleep disturbances (C5), use of sleep medications (C6), and daytime dysfunction (C7). The seven components can be added together to give an overall sleep-wake impairment (GSQI) score (Buysse, Reynolds, Monk, Berman, & Kupfer, 1989). Higher scores (range = 0–21) signify greater GSQI. GSQI scores greater than 5 indicate possible sleep pathology. Confirmatory factor analysis has supported a three-factor model with perceived sleep quality (C1, C2, C6), sleep efficiency (C3, C4), and daily disturbances (C5, C7) as separate indices of sleep-wake impairment (Casement, Harrington, Miller, & Resick, 2012). Internal consistency, test-retest reliability, and validity have been established for breast cancer (Beck, Schwartz, Towsley, Dudley, & Barsevick, 2004). In the current study, internal consistency (Cronbach alpha) was 0.78, 0.79, 0.76, and 0.69 for the patient group, and 0.75, 0.69, 0.73, and 0.73 for the caregiver group, at T0, T1, T2, and T3, respectively. To increase comprehensiveness of assessments, additional items evaluating additional sleep-wake parameters were developed based on the literature. The multiple-choice response format of the PSQI was employed to enhance consistency.
Patient and caregiver demographic and clinical characteristic forms were also developed for this study. Sociodemographic variables included patient or caregiver age, educational background, marital status, annual household income, smoking status and alcohol consumption, as well as caregiver gender, relation to patient, and duration of relationship. Patient clinical characteristics included menopausal status, comorbid diseases, perceived physical activity, time since diagnosis and/or surgery, disease stage, type of surgery, chemotherapy protocol, and use of any prescribed and/or over-the-counter medications. Caregiver clinical information included perceived physical activity, presence of concurrent diseases, and use of any prescribed and/or over-the-counter medications. At all time points, patients and caregivers reported on their level of functional capacity on the Eastern Cooperative Oncology Group (ECOG) Performance Status scale (Oken et al., 1982). At baseline, additional information was collected on sleep history; sleeping arrangements; and smoking, alcohol, and caffeine consumption.
Sample Size Estimation
Previous dyadic sleep research has shown an aggregated moderate correlation between patient and caregiver GSQI scores of r = 0.3 (Carney et al., 2011; Pal et al., 2004). According to sampling tables by Kenny, Kashy, and Cook (2006), a sample of 90 dyads (adjusted for 10% attrition rate) was required to detect this medium effect size of non-independence.
Data Analysis
A number of sleep-wake parameters were taken into consideration. To reduce the amount of statistical tests performed, only eight sleep-wake parameters were used as the main (outcome) study variables, including perceived sleep quality, habitual sleep efficiency (HSE), SOL, TST, WASO, duration of daytime naps (nap time), perceived sleep quality, daily disturbances, and GSQI scores. All additional parameters were only descriptively analyzed.
The multivariate hierarchical linear model (MHLM) for longitudinal dyadic data was employed (Kenny et al., 2006; Lyons & Sayer, 2005; Raudenbush, Brennan, & Barnett, 1995). All dyads participating in at least one wave of assessment were included in the analysis (Raudenbush & Bryk, 2002). As a first step, inter-dyad correlations for each sleep variable were examined for the presence of non-independence. Independent-groups tests were also performed to compare respondents’ and nonrespondents’ demographic characteristics and baseline and T1 scores on key outcome variables (perceived sleep quality, GSQI) at each follow-up time point to investigate patterns of missingness (Atkins, 2005). MHLM analyses produce unbiased estimates of all parameters under the assumption that data are missing at random (MAR). In such cases, no imputation of missing data is required.
Each MHLM involved two separate levels of analysis, a within-dyad model at level 1 and a between-dyad model at level 2. Two basic MHLMs were tested for each outcome variable: a baseline/unconditional MHLM, examining trajectories of change in sleep parameters, and an explanatory MHLM, exploring the relation of these trajectories with time-varying and time-invariant covariates and predictors. The current article focuses on the baseline/unconditional MHLMs. Within the baseline/unconditional MHLM, level 1 represented change over time for patients and caregivers. Univariate hypothesis testing (t ratio) indicated whether, on average, patient and caregiver intercepts and slopes differed significantly from zero. Effect sizes (rES) were calculated; values of 0.1, 0.3, and 0.5 indicated small, medium, and large effects, respectively (Cohen, 1988). Multivariate hypothesis testing indicated significant differences between average patient and caregiver trajectories (i.e., baseline status and rates of change over time) (Lyons & Sayer, 2005; Raudenbush et al., 1995). At level 2, the person-level estimates were aggregated, providing estimates for all patients and caregivers, and permitted to vary across all dyads. Tau-correlation coefficients and 95% confidence intervals indicated notable correlations representing the extent of shared variance (i.e., relationship) in each outcome variable for the members of a care dyad.
The linear and curvilinear (quadratic) effects of time were tested separately for all outcome variables to model change at level 1. The linear and quadratic MHLMs were compared with each other and with a means-only (null) model (containing only intercepts) through their deviance statistics (–2 log likelihood) to identify the most appropriate one for modeling change. Intraclass correlation coefficients (ICCs) were checked to confirm that multilevel modeling was needed in this dataset (if ICC is 0 or less, then multilevel modeling is not appropriate) (Garson, 2012). ICCs were calculated as the intercept variance component in the null model divided by the total of variance components (intercept plus residual). Preliminary analyses were performed using SPSS®, version 17; MHLM analyses were performed with restricted maximum likelihood estimation using HLM 7.
Results
Accrual Rates, Attrition Rates, and Patterns of Missingness
From November 2010 to April 2012, 97 eligible women were invited to participate. Sixty-one women (63% of the eligible individuals) and 49 caregivers (94% of the nominated individuals) provided consent. The final sample consisted of 48 care dyads, who participated in at least one wave of assessment. Attrition rates were 13% (n = 6), 23% (n = 11), and 25% (n = 12) for patients, caregivers, and dyads, respectively. Across all assessment points, reasons for loss to follow-up included busy patient and caregiver schedules, family member’s illness, preliminary termination of chemotherapy because of toxicity, caregiver working abroad, and caregiver death. In all, 34 dyads had complete data.
Dyads completing all four study assessments were more likely to include retired or unemployed patients (p = 0.033), individuals in long-standing relationships (p = 0.023), spouses or partners (p = 0.003), and patients whose sleep had not been affected by the breast cancer diagnosis (p = 0.033). When T0 and T1 perceived sleep quality and GSQI scores were compared between completers and non-completers, no significant differences were found (all p > 0.05). These analyses supported the assumption of MAR data and, therefore, the use of MHLM.
Sample Characteristics
Patient and caregiver characteristics are shown in Tables 1 and 2. The sample mainly consisted of middle-aged, wife–husband dyads (n = 30). On average, care dyads had well-established relationships of about 30 years (mean = 356.1 months, SD = 161.3, range = 18–624). Forty caregivers identified as husband or partner to the patient, seven were a child of the patient, and one was a friend of the patient.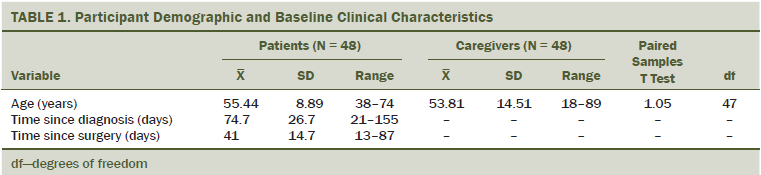
Patients varied in stage of breast cancer. Thirty patients had stage II cancer, 12 had stage IIIA, and 6 had stage I. Twenty-five patients were postmenopausal, 15 were premenopausal, and 8 were perimenopausal. Twenty-eight patients had undergone wide local excision, and 20 had a mastectomy. Thirty-one patients were on a chemotherapy regimen of fluorouracil (Efudex®), epirubicin (Ellence®), and cyclophosphamide (Cytoxan®) (FEC), nine patients were on a regimen of FEC plus taxanes, and eight patients were on another regimen.
[[{"type":"media","view_mode":"media_original","fid":"23376","attributes":{"alt":"","class":"media-image","height":"846","typeof":"foaf:Image","width":"365"}}]]
More than half of patients (65%) and caregivers (54%) were experiencing at least one chronic health condition, with patients suffering from more comorbidities compared to their caregivers (p = 0.026). Patients reported good to excellent ECOG Performance Status at baseline; most caregivers indicated excellent performance status at the same time point. Throughout the study, performance status of caregivers was consistently better compared to patients (all p < 0.001).
At least half of the patients (58%) and caregivers (50%) had experienced sleep problems in the past. Of note, 63% of the women indicated that their sleep had been affected by the diagnosis of breast cancer, whereas 40% of caregivers also indicated that. In terms of sleeping arrangements, 42 dyads indicated sharing the same house, and 37 also shared the same bedroom. Preliminary analyses showed no effect of the dyads’ cohabitation status on patients’ or caregivers’ sleep parameters (all p > 0.05); therefore, all 48 dyads were included in final analyses.
[[{"type":"media","view_mode":"media_original","fid":"23381","attributes":{"alt":"","class":"media-image","height":"761","typeof":"foaf:Image","width":"1063"}}]]
Descriptive Analysis of Sleep Variables
At all time points, mean GSQI scores for patients and caregivers exceeded the threshold of 5 (see Table 3), suggesting that 52%–77% of patients and 35%–49% of caregivers experienced clinically significant sleep-wake impairment throughout the study (see Figure 1). At baseline, 31 of the dyads consisted of one or two poor sleepers. At T2, dyads with one poor sleeper exceeded 45% (n = 21), whereas, for an additional 35% (n = 15), both the patient and the caregiver experienced poor sleep. Three weeks after the end of chemotherapy, more than 70% (n = 30) of dyads consisted of at least one poor sleeper.
On average, no major fluctuations in dyads’ self-reported bed and wake times were observed. However, compared to mean prechemotherapy values, both patients and caregivers tended to turn in earlier (patients: mean = 11–20 minutes; caregivers: mean = 2–5 minutes) and wake up later (patients: mean = 2–5 minutes; caregivers: mean = 3–12 minutes) during active treatment. Throughout chemotherapy, 35%–51% of patients and 30%–40% of caregivers experienced three or more awakenings per night; 17%–28% of patients and 13%–31% of caregivers awoke early and could not go back to sleep; and 40%–57% of patients and 30%–40% of caregivers felt minimally rested the next morning. Only 7%–23% of patients and 5%–11% of caregivers reported actual use of sleep aids, including lorazepam (Ativan®), zolpidem (Ambien®), and zopiclone, and the antidepressant drugs fluoxetine (Prozac®) and citalopram (Celexa®).
The degree of sleepiness was only moderate, with 10%–30% of patients and 8%–15% of caregivers feeling quite a bit or very sleepy during the day; however, average daytime sleepiness levels reached a peak during treatment for patients and carers compared to baseline and end of study. The need for napping in the daytime was also prominent for both groups (patients = 50%–77%; caregivers = 42%–57%). Mean nap time exceeded 20 minutes and 15 minutes for patients and caregivers, respectively. Duration of daytime naps was reported as longer than 30 minutes by 24%–44% of patients and 10%–19% of caregivers.
Preliminary Bivariate Correlation Analyses
Modest, but significant, associations between patients’ and caregivers’ sleep-wake patterns over time were found. At different time points, patient and caregiver SOL (T0 = 0.37, T2 = 0.36), perceived sleep quality (T0 = 0.3), daily disturbances (T1 = 0.37, T2 = 0.36, T3 = 0.49), WASO (T2 = –0.24), GSQI (T0 = 0.27), wake time (T0 = 0.3, T3 = 0.32), feelings upon awakening in the morning (T0 = 0.32), sleepiness (T0 = 0.3), and sleep aid use (T2 = 0.37) were found to be linearly related. Bedtime was the only variable where patient and caregiver patterns were consistently correlated throughout the study (0.35–0.41).
Multivariate Hierarchical Linear Model Analyses
Eight baseline (unconditional) MHLMs were estimated. Comparative analyses supported use of the quadratic model for perceived sleep quality, nap time, SOL, HSE, WASO, and GSQI, whereas, for daily disturbances, the linear model was a better fit to the data (see Table 4). For TST, neither model was significantly better fit than the means-only model. ICCs ranged from 0.38–0.61 (all p < 0.001) for the patient variables, and from 0.38–0.62 (all p < 0.001) for the caregiver variables, confirming that MHLM was appropriate and needed for these data.
Patient-perceived sleep quality, nap time, SOL, HSE, WASO, and GSQI deteriorated steadily from pre- (T0) to midtreatment (T1 and T2), and then leveled off three weeks postchemotherapy (T3). Modest to large effect sizes were found for the linear (rES = 0.35–0.53) and quadratic (rES = 0.28–0.49) trends (see Table 5). Somewhat differently, patient daily disturbances followed a linear pattern of change throughout chemotherapy, remaining high even after the end of treatment. Conversely, caregiver linear or quadratic trends for these parameters fell short of significance (all p < 0.05), suggesting no major disruption in caregivers’ self-reported sleep-wake parameters. However, average dyad trajectories were indicative of a trend for slight deterioration in caregiver sleep-wake patterns (particularly perceived sleep quality, nap time, HSE, WASO, GSQI) as dyads entered midtreatment; average caregiver sleep-wake impairment reached its own peak at the same time as patients’. Therefore, dyads halfway through the patient’s chemotherapy treatment were more sleep-impaired than at any other point during this period. Of note, 38% and 28% of patients and 35% and 26% of caregivers experienced reduction in their total sleep time at T1 and T2, respectively, compared to baseline. Fifty percent of dyads at T1 and 49% of dyads at T2 included one or both members whose actual sleep time during the night had been adversely affected.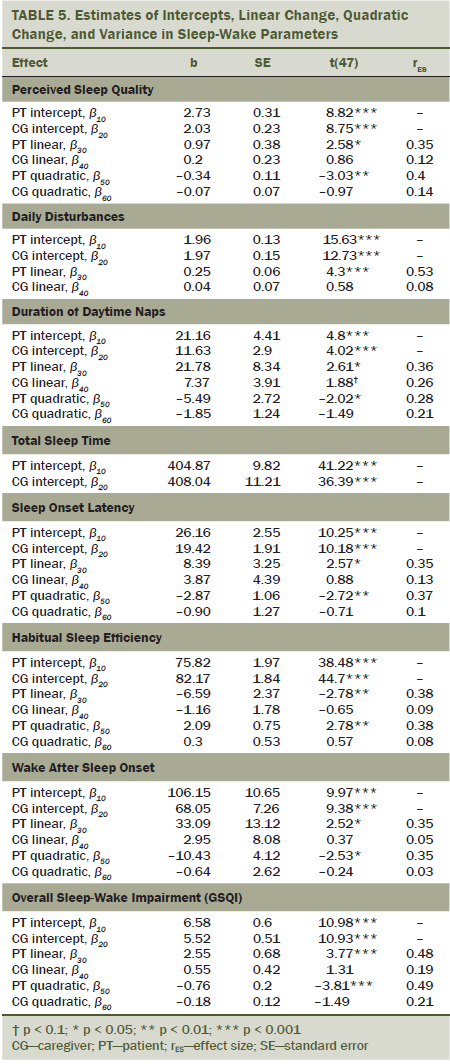
Multivariate hypothesis testing indicated that average curves for the sleep-wake parameters were significantly different between patients and caregivers (all p < 0.021) and more disrupted for patients than for caregivers throughout chemotherapy; however, at baseline, daily disturbances and GSQI scores of dyad members did not significantly differ (p > 0.05). In addition, patients’ rates of acceleration (linear trend) and deceleration (quadratic trends) were significantly different than those of caregivers; the only exceptions were nap time and SOL. At T2, prolongation of average caregiver SOL to levels close to those reported from patients (caregiver = 29 minutes versus patient = 32 minutes) and higher than their previous SOL (caregiver = 29 minutes versus 20 minutes versus 19 minutes) was evident. Early in the course of treatment, dyad members’ SOL was greater for patients than caregivers. However, as time elapsed (after the first chemotherapy cycle to end of treatment), differences in SOL patterns started to fade out; caregivers found falling asleep increasingly more difficult, assimilating patients’ own difficulty. From T2 to end of study, dyad SOL curves became parallel, showing somewhat similar patterns of deceleration and restoration of SOL to prechemotherapy levels.
Despite the differences in the magnitude of sleep impairment, within-dyad changes in dyad members’ specific sleep parameters were closely related. Cross-dyad member (random effects) tau correlations revealed a moderate patient–caregiver association in baseline GSQI scores (tau = 0.42) (see Table 6), supported by fairly strong correlations for linear (tau = 0.76) and quadratic (tau = 0.84) change. Similarly, dyad members’ average perceived sleep quality and SOL, but particularly changes in these parameters, were closely related. Conversely, no major or significant interrelations between dyad members’ daily disturbances and nap time were found. Weak tau correlation coefficients for baseline HSE (tau = 0.12) and WASO (tau = 0.19) also emerged. This dissociation was further confirmed by modest, but negative, correlations for linear and quadratic change. Within the same dyad, patient and caregiver reports of average HSE and WASO were found to be only weakly related, but steeper declines in a patient’s HSE (or WASO) correlated with steeper increases in the caregiver’s HSE (or WASO), and vice versa.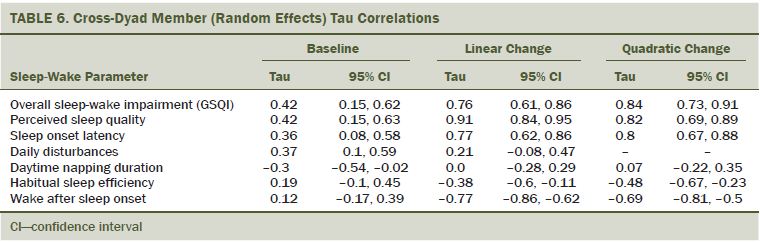
Discussion
The current study set out to investigate self-reported sleep-wake patterns of patient–caregiver dyads during adjuvant chemotherapy for breast cancer. With the exception of daily disturbances, overall trajectories of change for patients’ sleep-wake patterns followed a significant curvilinear pattern (Savard et al., 2009). Except for nap time, carers’ perceptions of their own sleep revealed no particular patterns, which is similar to previous research (Stenberg et al., 2014). The magnitude of sleep-wake disruption was significantly greater in patients compared with caregivers. However, at pretreatment, wake variables, TST, and GSQI scores were not significantly different between dyad members. After T2, there was a similar pattern for TST and SOL, potentially suggesting that patients and carers had diminished sleep duration (compared to each group’s average) and generally similar difficulty to fall asleep. Changes in perceived sleep quality, SOL, and GSQI in patients were followed closely by similar in direction changes in their caregivers, revealing interrelated sleep patterns. Daytime wake variables remained uncoupled. Although comparable with evidence from advanced cancer care dyads (Gibbins et al., 2009), alarming percentages of dyads with at least one poor sleeper were revealed before chemotherapy initiation that further increased as patient treatment progressed. With more than two-thirds of dyads still burdened by poor sleep in one or both members after the end of treatment, ongoing sleep-related distress was evident, potentially increasing the odds for adverse outcomes, including prolonged and debilitating fatigue (Broeckel, Jacobsen, Horton, Balducci, & Lyman, 1998).
Comparability in frequencies of poor self-reported sleep between patients and caregivers has been suggested (Carney et al., 2011; Celik, Annagur, Yilmaz, Demir, & Kara, 2012; Gibbins et al., 2009; Happe & Berger, 2002; Smith, Ellgring, & Oertel, 1997), but it was only confirmed for pretreatment data in the current article. Except for TST, the significant differences in average curves of all outcome variables were indicative of a generally greater impact on patients’ rather than caregivers’ sleep throughout chemotherapy, which persisted into the initial postchemotherapy period. Relative discrepancies may be because of contextual effects of ever-changing challenges experienced during chemotherapy. Rates of acceleration in the deterioration of perceived sleep quality, HSE, WASO, daily disturbances, and GSQI were also consistently steeper for patients. These findings were complemented by consistent clinical differences in the frequency of sleep aid use and nocturnal awakenings, and daytime napping and sleepiness between dyad members. Despite inherent differences in the nature and course of illness when compared to early-stage breast cancer, prospective studies in the context of dementia (McCurry, Pike, Vitiello, Logsdon, & Teri, 2008) and advanced cancer care (Gibbins et al., 2009) have also suggested greater problems with sleep maintenance (McCurry et al., 2008) and daytime functioning (Gibbins et al., 2009) for patients than for caregivers. For example, Gibbins et al. (2009) found that activity levels were consistently higher for caregivers, and time immobile in the daytime was greater for patients with advanced cancer. In the current study, however, physical activity levels were similar for dyad members overall. It is more possible that nocturnal sleep loss and physical symptom burden urged women to have more prolonged napping in the daytime. In addition, in this largely independent patient population, overnight caregiving demands might have been kept to a minimum. In contrast, other caregiving populations may be more heavily involved in care and required to actively respond to patients’ needs at night, becoming more sleep deprived (Carlsson, 2012; Carter, 2003; Hearson, McClement, McMillan, & Harlos, 2011; Pawl et al., 2013). Still, caregivers in the current study went to sleep at night an average of 4–18 minutes later than patients and woke up in the morning an average of 39–50 minutes earlier throughout the study period, suggesting that caregivers tended to have generally later bedtimes and earlier awakening times (Carney et al., 2011; Castro et al., 2009; Lee, Morgan, & Lindesay, 2007). An alternative explanation might be that, potentially because of adverse chemotherapy effects, women with breast cancer simply felt the need to rest earlier than usual and extended the time they spent lying on the bed, even if they were not actually sleeping. Even with minimal caregiving involvement, it is still possible that, in the current study, certain chemotherapy-related symptoms experienced during the night (e.g., pain, cough, breathlessness, hot flushes) and the associated arousal, agitation, and distress have been the cause of multiple nocturnal awakenings and prolonged WASO not only for patients, but also for caregivers, and not only for bed or bedroom sharers (through direct interactions or indirect sleep-disrupting cues, such as noise or light), but also for non-cohabiting dyads, perhaps because of patients telephoning or texting their caregivers in the night for help and support.
At specific time points and for certain variables, dyad members’ sleep patterns tended to converge. At baseline, daily disturbances, nap time, TST, and GSQI were no different between patients and caregivers, suggesting that, to a certain extent, dyads shared similar degrees of sleep-wake impairment before chemotherapy. These findings confirm similar evidence from a sample of 102 patient–caregiver dyads evaluated one week prior to primary or adjuvant radiation therapy for diverse non-metastatic cancers (Carney et al., 2011). Among the unique findings of this study is that, during treatment continuation, rates of change in SOL and nap time were not significantly different between patients and caregivers. Particularly with regard to SOL, average patient and caregiver trajectories almost coincided at midtreatment, where difficulty to fall asleep became equivalent for patients and caregivers. In addition, one of the most clinically important observations was that deterioration in caregiver sleep-wake variables reached its highest as dyads reached midtreatment, a time point where patient sleep-wake impairment also peaked. Despite differences in the magnitude of sleep deficit, this trend gives credence to the hypothesis that patients with breast cancer (Savard et al., 2009) and caregivers might show progressive impairments in their sleep-wake patterns, and dyad members concurrently experience sleep deficits at specific time points during treatment.
Even in the presence of a small number of non-cohabiting dyads in this sample (n = 6), moderate positive correlations also emerged between patient and caregiver perceived sleep quality, SOL, and GSQI at pretreatment. Patterns of change in these variables were strongly correlated between dyad members throughout the study, indicating significant interdependence. Two cross-sectional studies conducted with dyads in the context of cancer (Carney et al., 2011) and Parkinson’s disease (Pal et al., 2004) reported relationships of a similar magnitude in the dyad members’ perceived sleep quality. Particularly in cohabiting and bed-sharing dyads with characteristics similar to the ones of those participated in the current study (i.e., spouses or partners in well-established relationships), this finding underpins the notion of dyadic sleep rituals (Crossley, 2004), such as common bedtimes and complementary sleep hygiene practices (e.g., caregiver abstinence from alcohol consumption as a sleep-inducing method, joint avoidance of activities other than sleep in the bed or bedroom) that potentially allowed for closely correlated sleep initiation and quality, or conversely led to closely correlated sleep disruption in both dyad members if these sleep rituals were disturbed or not met. Although sleep rituals may be absent in non-cohabiting care dyads, it can still be postulated that patient sleeplessness in the night might have led to caregiver wakefulness because patients may have sought help and consolation for symptoms. In addition, MHLM analyses indicated only minimal, or even inverse, relationships between average patient and caregiver TST, HSE, and WASO curves; this finding stands in contrast with existing evidence suggesting moderate to strong positive relationships, depending on objective recordings (Carney et al., 2011; Castro et al., 2009; Lee et al., 2007), but it does support similar weak associations based on self-report (Carney et al., 2011). It can be hypothesized that these results reflect differing degrees of burden posed to dyad members during chemotherapy that have ultimately blurred or weakened such relationships. Finally, trajectories of daytime behaviors (i.e., need for daytime napping and daytime napping duration) of dyad members remained uncoupled throughout this study, supporting previous evidence (Carney et al., 2011; Lee et al., 2007; Pal et al., 2004; Pollak & Stokes, 1997; Pollak, Stokes, & Wagner, 1997). The reason for this outcome can possibly be differences in daytime schedules in this sample of young and employed dyads, compared to older adults who may share their daytime activities more closely.
Strengths and Limitations
This study has a number of strengths. First, the authors employed a dyadic methodology, introducing a novel way to examine sleep-wake disturbances in the context of cancer care (Carney et al., 2011; Gibbins et al., 2009) and reveal sleep-related processes and sources or triggers of sleep problems in patients and their caregivers who were co-affected within the cancer-related circumstances (Kotronoulas et al., 2013a). Second, this study employed a longitudinal, repeated-measures design (Berger et al., 2005, 2010; Swore Fletcher, Dodd, Schumacher, & Miaskowski, 2008). Given the lack of longitudinal sleep research of cancer caregivers, the study makes a significant addition to the knowledge regarding sleep-wake deficits over time in this population (Berger et al., 2005; Kim & Rose, 2011; Swore Fletcher et al., 2008). Finally, the current study used multilevel modeling techniques to analyze dyadic data (Raudenbush et al., 1995), an approach that has only recently been employed in sleep research with patients with breast cancer (Liu et al., 2012, 2013; Rumble et al., 2010).
This study also has several limitations. A challenging recruitment resulted in a smaller than required sample size. Post-hoc power analyses revealed a statistical power ranging from less than 50% (WASO) to 80% (GSQI) to detect small to medium effect sizes of non-independence (tau correlation coefficients = 0.12–0.42) in patient and caregiver pretreatment sleep-wake parameters (Kenny et al., 2006). Similarly, power to detect a significant difference between GSQI of patients and caregivers ranged from 50% to greater than 80% (effect sizes d = 0.42–1.15) (Kenny et al., 2006). Relying on subjective sleep measures, which are prone to introducing recall bias because of their retrospective nature, might have affected measurement accuracy. Care dyads’ sleeping arrangements were assessed only at pretreatment, but it cannot be ruled out that some of the dyads changed their sleeping arrangements at a later point during the study. This might have led to patients and caregivers spending more or less time together at night. Demographic homogeneity limits generalizability of the current findings to female caregivers, non-Caucasian patient–caregiver dyads, or dyads other than spouses or long-term partners. Gender might have played a significant role in how dyadic sleep patterns were represented in this study. Had the gender synthesis of care dyads been different (for example, in the context of prostate cancer, patients are male, but the majority of caregivers are female), dyadic sleep profiles and the extent of interdependence might have been different. Finally, caregivers providing additional care to other family members were not excluded from this study; therefore, sleep impairment might have been magnified by concurrent parenting and caregiving responsibilities.
Implications for Research and Practice
Future dyadic studies relying on sleep interaction data will be required to validate associations, confirm similarities (e.g., perceived sleep quality, SOL) and differences (e.g., nap time, daily disturbances), and resolve ambiguity (e.g., TST, WASO) with regard to dyads’ sleep-wake trajectories, as well as to reinvestigate the magnitude of dyads’ sleep-wake impairment throughout treatment for breast cancer. Longitudinal sleep studies that make use of predictive models of statistical associations are highly advisable (Berger et al., 2010) to facilitate clarification of relationships between patient and caregiver sleep parameters. Structural equation modeling can be used in dyadic research as an alternative to MHLM data analytic strategy (Kenny et al., 2006). Latent class growth analysis (Jung & Wickrama, 2008) could facilitate identification of subgroups of dyads (both poor sleepers versus both good sleepers versus one poor sleeper) and allow comparative investigation of changes over time in sleep-wake patterns. In addition, studies integrating quantitative and qualitative data (Ostlund, Kidd, Wengstrom, & Rowa-Dewar, 2011) could be particularly useful in clarifying underlying mechanisms in the development of dyadic sleep disturbances.
With pressing requirements for improved psychosocial cancer care (Jacobsen, Holland, & Steensma, 2012; Jacobsen & Wagner, 2012; Northouse, Katapodi, Song, Zhang, & Mood, 2010), healthcare teams are now expected to view the patient–caregiver dyad as the unit of care (Northouse et al., 2010), provide support that meets concurrent and interdependent needs, and promote the combined well-being of the dyad. Together with the latest approaches to patient and caregiver support that urge for early identification of potential sleep disturbance (Berger et al., 2005, 2010; Lee & Thomas, 2011), the finding that at least 25%–48% of care dyads might meet criteria for sleep-wake impairment at the same time throughout chemotherapy for early-stage breast cancer calls for a proactive management of sleep complaints in these populations. Evaluation of sleep from a dyadic perspective requires engaging in a systematic investigation that goes beyond the individual; data are simultaneously taken into account, synthesized, and contrasted to establish a dyad’s levels of sleep quality. Ongoing sleep assessments are required to ensure optimal results. Complemented by additional information regarding past sleep problems, present sleeping arrangements, sleep hygiene behaviors, and current use of sleep aids, such data can help clinicians identify potentially vulnerable dyads for sleep problems. Incorporation of screening tools for organized sleep assessments in routine clinical practice is recommended (Lowery, 2014). Once problems are suspected and/or complaints are raised, evaluation of the onset, duration, and severity of sleep deficits, as well as daytime dysfunction can reveal potentially evolving, co-occurring, and/or interrelated problems. A plan of practical suggestions to reduce disturbing nocturnal interactions of cohabiting dyads can be usefully devised, including use of twin beds or separate rooms for sleep, separation of sleeping quarters, the use of alarms, readjustment of the patient’s caregiving routines, and synchronization of positive sleep hygiene behaviors (Pollak et al., 1997). Joint provision of psychobehavioral interventions to patients and caregivers may have an additive effect to the benefit that they can gain from them as individuals (Matthews et al., 2014). Because cognitive-behavioral therapy for insomnia (CBT-I) can be conducted either one-on-one or in groups of individuals (Langford, Lee, & Miaskowski, 2012), devising a plan to offer CBT-I to dyads should be considered. The goal would be to ensure ongoing and intensive intervention provision during at least the first four chemotherapy cycles to achieve maximum benefit when dyads reach midtreatment (major sleep-wake disturbance), and promote individualized and combined goal-setting for patients and caregivers (Kotronoulas et al., 2013a).
Conclusion
The current study has taken dyadic sleep research a step forward by examining the interrelations in the sleep-wake patterns of women receiving chemotherapy for early-stage breast cancer and their informal caregivers. However, the bidirectional associations in the sleep-wake patterns of patients with cancer and their caregivers are not yet mapped and understood. Unraveling the complex underlying pathways that lead to the development of sleep-wake impairments in these dyads and exploring how interventions can support people affected by cancer in everyday practice are questions that need to be addressed in an attempt to find innovative and more effective ways to provide better care to patients and their caregivers.
The authors gratefully acknowledge Dougal J.A. Adamson, MD, John Dewar, MD, Ravi Sharma, MBBS, MRCP, MSc, FRCR, FRCP Edin, Radha Todd, MBBS, PhD, FRCP, Angela Bowman, MCChB, MRCP, Maureen Devaney, MSC, BSN, RN, and Fiona Galbraith, BSN, RN, for their assistance in the recruitment of dyads at the participating sites, as well as Karen Lyons, PhD, for her detailed feedback on an early version of the data analytic strategy. The authors also acknowledge the patients and caregivers for their invaluable contribution to the study.
References
Adams, G.C., Stoops, M.A., & Skomro, R.P. (2014). Sleep tight: Exploring the relationship between sleep and attachment style across the life span. Sleep Medicine Reviews, 18, 495–507. doi:10.1016/j.smrv.2014.03.002
Ancoli-Israel, S., Liu, L., Rissling, M., Natarajan, L., Neikrug, A.B., Palmer, B.W., . . . Maglione, J. (2014). Sleep, fatigue, depression, and circadian activity rhythms in women with breast cancer before and after treatment: A 1-year longitudinal study. Supportive Care in Cancer, 22, 2535–2545. doi:10.1007/s00520-014-2204-5
Atkins, D.C. (2005). Using multilevel models to analyze couple and family treatment data: Basic and advanced issues. Journal of Family Psychology, 19, 98–110. doi:10.1037/0893-3200.19.1.98
Beck, S.L., Schwartz, A.L., Towsley, G., Dudley, W., & Barsevick, A. (2004). Psychometric evaluation of the Pittsburgh Sleep Quality Index in cancer patients. Journal of Pain and Symptom Management, 27, 140–148. doi:10.1016/j.jpainsymman.2003.12.002
Berger, A.M., Parker, K.P., Young-McCaughan, S., Mallory, G.A., Barsevick, A.M., Beck, S.L., . . . Hall, M. (2005). Sleep wake disturbances in people with cancer and their caregivers: State of the science [Online exclusive]. Oncology Nursing Forum, 32, E98–E126. doi:10.1188/05.ONF.E98-E126
Berger, A.M., Wielgus, K., Hertzog, M., Fischer, P., & Farr, L. (2010). Patterns of circadian activity rhythms and their relationships with fatigue and anxiety/depression in women treated with breast cancer adjuvant chemotherapy. Supportive Care in Cancer, 18, 105–114. doi:10.1007/s00520-009-0636-0
Bowlby, J. (1978). Attachment theory and its therapeutic implications. Adolescent Psychiatry, 6, 5–33.
Broeckel, J.A., Jacobsen, P.B., Horton, J., Balducci, L., & Lyman, G.H. (1998). Characteristics and correlates of fatigue after adjuvant chemotherapy for breast cancer. Journal of Clinical Oncology, 16, 1689–1696.
Bryant, P.A., Trinder, J., & Curtis, N. (2004). Sick and tired: Does sleep have a vital role in the immune system? Nature Reviews. Immunology, 4, 457–467. doi:10.1038/nri1369
Buysse, D.J., Reynolds, C.F., III, Monk, T.H., Berman, S.R., & Kupfer, D.J. (1989). The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research. Psychiatry Research, 28, 193–213. doi:10.1016/0165-1781(89)90047-4
Carlsson, M.E. (2012). Sleep disturbance in relatives of palliative patients cared for at home. Palliative and Supportive Care, 10, 165–170. doi:10.1017/s1478951511000836
Carney, S., Koetters, T., Cho, M., West, C., Paul, S.M., Dunn, L., . . . Miaskowski, C. (2011). Differences in sleep disturbance parameters between oncology outpatients and their family caregivers. Journal of Clinical Oncology, 29, 1001–1006.
Carter, P.A. (2003). Family caregivers’ sleep loss and depression over time. Cancer Nursing, 26, 253–259.
Casement, M.D., Harrington, K.M., Miller, M.W., & Resick, P.A. (2012). Associations between Pittsburgh Sleep Quality Index factors and health outcomes in women with posttraumatic stress disorder. Sleep Medicine, 13, 752–758. doi:10.1016/j.sleep.2012.02.014
Castro, C.M., Lee, K.A., Bliwise, D.L., Urizar, G.G., Woodward, S.H., & King, A.C. (2009). Sleep patterns and sleep-related factors between caregiving and non-caregiving women. Behavioral Sleep Medicine, 7, 164–179. doi:10.1080/15402000902976713
Celik, G., Annagur, B.B., Yilmaz, M., Demir, T., & Kara, F. (2012). Are sleep and life quality of family caregivers affected as much as those of hemodialysis patients? General Hospital Psychiatry, 34, 518–524. doi:10.1016/j.genhosppsych.2012.01.013
Cohen, J. (1988). Statistical power analysis for the behavioral sciences (2nd ed.). Hillsdale, NJ: Erlbaum.
Costa, A.R., Fontes, F., Pereira, S., Goncalves, M., Azevedo, A., & Lunet, N. (2014). Impact of breast cancer treatments on sleep disturbances—A systematic review. Breast, 23, 697–709. doi:10.1016/j.breast.2014.09.003
Crossley, N. (2004, December). Sleep, reflexive embodiment and social networks. Paper presented at the Economic and Social Research Council “Sleep and society: Critical themes, future agendas” seminar, Coventry, England. Retrieved from http://www2.warwick.ac.uk/fac/cross_fac/healthatwarwick/news/sleepandso…
Dorros, S.M., Card, N.A., Segrin, C., & Badger, T.A. (2010). Interdependence in women with breast cancer and their partners: An interindividual model of distress. Journal of Consulting and Clinical Psychology, 78, 121–125. doi:10.1037/a0017724
Fiorentino, L., & Ancoli-Israel, S. (2006). Insomnia and its treatment in women with breast cancer. Sleep Medicine Reviews, 10, 419–429. doi:10.1016/j.smrv.2006.03.005
Garson, G.D. (2012). Introductory guide to HLM with HLM 7 software. In G.D. Garson (Ed.), Hierarchical linear modeling: Guide and applications (pp. 55–96). Austin, TX: Sage.
Gibbins, J., McCoubrie, R., Kendrick, A.H., Senior-Smith, G., Davies, A.N., & Hanks, G.W. (2009). Sleep-wake disturbances in patients with advanced cancer and their family carers. Journal of Pain and Symptom Management, 38, 860–870. doi:10.1016/j.jpainsymman.2009.04.025
Happe, S., & Berger, K. (2002). The association between caregiver burden and sleep disturbances in partners of patients with Parkinson’s disease. Age and Ageing, 31, 349–354. doi:10.1093/ageing/31.5.349
Hearson, B., McClement, S., McMillan, D.E., & Harlos, M. (2011). Sleeping with one eye open: The sleep experience of family members providing palliative care at home. Journal of Palliative Care, 27, 69–78.
Ho, S.Y., Rohan, K.J., Parent, J., Tager, F.A., & McKinley, P.S. (2015). A longitudinal study of depression, fatigue, and sleep disturbances as a symptom cluster in women with breast cancer. Journal of Pain and Symptom Management, 49, 707–715. doi:10.1016/j.jpainsymman.2014.09.009
Jacobsen, P.B., Holland, J.C., & Steensma, D.P. (2012). Caring for the whole patient: The science of psychosocial care. Journal of Clinical Oncology, 30, 1151–1153. doi:10.1200/jco.2011.41.4078
Jacobsen, P.B., & Wagner, L.I. (2012). A new quality standard: The integration of psychosocial care into routine cancer care. Journal of Clinical Oncology, 30, 1154–1159. doi:10.1200/jco.2011.39.5046
Jung, T., & Wickrama, K.A.S. (2008). An introduction to latent class growth analysis and growth mixture modeling. Social and Personality Psychology Compass, 2(1), 302–317. doi:10.1111/j.1751-9004.2007.00054.x
Kenny, D.A., Kashy, D.A., & Cook, W.L. (2006). Dyadic data analysis. London, England: Guilford Press.
Kim, H., & Rose, K. (2011). Sleep disturbances in family caregivers: An overview of the state of the science. Archives of Psychiatric Nursing, 25, 456–468. doi:10.1016/j.apnu.2011.02.007
Kotronoulas, G., Wengstrom, Y., & Kearney, N. (2012). A critical review of women’s sleep-wake patterns in the context of neo-/adjuvant chemotherapy for early-stage breast cancer. Breast, 21, 128–141. doi:10.1016/j.breast.2011.12.004
Kotronoulas, G., Wengstrom, Y., & Kearney, N. (2013a). Sleep and sleep-wake disturbances in care recipient-caregiver dyads in the context of a chronic illness: A critical review of the literature. Journal of Pain and Symptom Management, 45, 579–594. doi:10.1016/j.jpainsymman.2012.03.013
Kotronoulas, G., Wengstrom, Y., & Kearney, N. (2013b). Sleep patterns and sleep-impairing factors of persons providing informal care for people with cancer: A critical review of the literature. Cancer Nursing, 36, E1–E15. doi:10.1097/NCC.0b013e3182456c38
Langford, D.J., Lee, K., & Miaskowski, C. (2012). Sleep disturbance interventions in oncology patients and family caregivers: A comprehensive review and meta-analysis. Sleep Medicine Reviews, 16, 397–414. doi:10.1016/j.smrv.2011.07.002
Lee, D., Morgan, K., & Lindesay, J. (2007). Effect of institutional respite care on the sleep of people with dementia and their primary caregivers. Journal of the American Geriatrics Society, 55, 252–258. doi:10.1111/j.1532-5415.2007.01036.x
Lee, D.R., & Thomas, A.J. (2011). Sleep in dementia and caregiving—Assessment and treatment implications: A review. International Psychogeriatrics, 23, 190–201. doi:10.1017/s1041610210001894
Liu, L., Fiorentino, L., Rissling, M., Natarajan, L., Parker, B.A., Dimsdale, J.E., . . . Ancoli-Israel, S. (2013). Decreased health-related quality of life in women with breast cancer is associated with poor sleep. Behavioral Sleep Medicine, 11, 189–206. doi:10.1080/15402002.2012.660589
Liu, L., Rissling, M., Natarajan, L., Fiorentino, L., Mills, P. J., Dimsdale, J.E., . . . Ancoli-Israel, S. (2012). The longitudinal relationship between fatigue and sleep in breast cancer patients undergoing chemotherapy. Sleep, 35, 237–245. doi:10.5665/sleep.1630
Lowery, A.E. (2014). Insomnia screening and treatment in cancer care: Current practices, barriers and future directions. JSM Clinical Oncology and Research, 2, 1026.
Lyons, K.S., & Sayer, A.G. (2005). Using multilevel modeling in caregiving research. Aging and Mental Health, 9, 189–195. doi:10.1080/13607860500089831
Matthews, E.E., Berger, A.M., Schmiege, S.J., Cook, P.F., McCarthy, M.S., Moore, C.M., & Aloia, M.S. (2014). Cognitive behavioral therapy for insomnia outcomes in women after primary breast cancer treatment: A randomized, controlled trial. Oncology Nursing Forum, 41, 241–253. doi:10.1188/14.onf.41-03ap
McCurry, S.M., Pike, K.C., Vitiello, M.V., Logsdon, R.G., & Teri, L. (2008). Factors associated with concordance and variability of sleep quality in persons with Alzheimer’s disease and their caregivers. Sleep, 31, 741–748.
Northouse, L.L., Katapodi, M.C., Song, L., Zhang, L., & Mood, D.W. (2010). Interventions with family caregivers of cancer patients: Meta-analysis of randomized trials. CA: A Cancer Journal for Clinicians, 60, 317–339. doi:10.3322/caac.20081
Oken, M.M., Creech, R.H., Tormey, D.C., Horton, J., Davis, T.E., McFadden, E.T., & Carbone, P.P. (1982). Toxicity and response criteria of the Eastern Cooperative Oncology Group. American Journal of Clinical Oncology, 5, 649–655. doi:10.1097/00000421-198212000-00014
Ostlund, U., Kidd, L., Wengstrom, Y., & Rowa-Dewar, N. (2011). Combining qualitative and quantitative research within mixed method research designs: A methodological review. International Journal of Nursing Studies, 48, 369–383. doi:10.1016/j.ijnurstu.2010.10.005
Pal, P.K., Thennarasu, K., Fleming, J., Schulzer, M., Brown, T., & Calne, S.M. (2004). Nocturnal sleep disturbances and daytime dysfunction in patients with Parkinson’s disease and in their caregivers. Parkinsonism and Related Disorders, 10, 157–168. doi:10.1016/j.parkreldis.2003.11.002
Pawl, J.D., Lee, S.Y., Clark, P.C., & Sherwood, P.R. (2013). Sleep characteristics of family caregivers of individuals with a primary malignant brain tumor. Oncology Nursing Forum, 40, 171–179. doi:10.1188/13.onf.171-179
Pollak, C.P., & Stokes, P.E. (1997). Circadian rest-activity rhythms in demented and nondemented older community residents and their caregivers. Journal of the American Geriatrics Society, 45, 446–452. doi:10.1111/j.1532-5415.1997.tb05169.x
Pollak, C.P., Stokes, P.E., & Wagner, D.R. (1997). Nocturnal interactions between community elders and caregivers, as measured by cross-correlation of their motor activity. Journal of Geriatric Psychiatry and Neurology, 10, 168–173. doi:10.1177/089198879701000408
Raudenbush, S.W., Brennan, R.T., & Barnett, R.C. (1995). A multivariate hierarchical model for studying psychological change within married couples. Journal of Family Psychology, 9(2), 161–174. doi:10.1037/0893-3200.9.2.161
Raudenbush, S.W., & Bryk, A.S. (2002). Hierarchical linear models: Applications and data analysis methods. Thousand Oaks, CA: Sage.
Rumble, M.E., Keefe, F.J., Edinger, J.D., Affleck, G., Marcom, P.K., & Shaw, H.S. (2010). Contribution of cancer symptoms, dysfunctional sleep related thoughts, and sleep inhibitory behaviors to the insomnia process in breast cancer survivors: A daily process analysis. Sleep, 33, 1501–1509.
Savard, J., Ivers, H., Savard, M.H., & Morin, C.M. (2015). Cancer treatments and their side effects are associated with aggravation of insomnia: Results of a longitudinal study. Cancer, 121, 1703–1711. doi:10.1002/cncr.29244
Savard, J., Liu, L., Natarajan, L., Rissling, M.B., Neikrug, A.B., He, F., . . . Ancoli-Israel, S. (2009). Breast cancer patients have progressively impaired sleep-wake activity rhythms during chemotherapy. Sleep, 32, 1155–1160.
Shilling, V., & Jenkins, V. (2007). Self-reported cognitive problems in women receiving adjuvant therapy for breast cancer. European Journal of Oncology Nursing, 11, 6–15.
Smith, M.C., Ellgring, H., & Oertel, W.H. (1997). Sleep disturbances in Parkinson’s disease patients and spouses. Journal of the American Geriatrics Society, 45, 194–199.
Stenberg, U., Cvancarova, M., Ekstedt, M., Olsson, M., & Ruland, C. (2014). Family caregivers of cancer patients: Perceived burden and symptoms during the early phases of cancer treatment. Social Work in Health Care, 53, 289–309. doi:10.1080/00981389.2013.873518
Swore Fletcher, B.A., Dodd, M.J., Schumacher, K.L., & Miaskowski, C. (2008). Symptom experience of family caregivers of patients with cancer [Online exclusive]. Oncology Nursing Forum, 35, E23–E44. doi:10.1188/08.ONF.E23-E44
Troxel, W.M. (2010). It’s more than sex: Exploring the dyadic nature of sleep and implications for health. Psychosomatic Medicine, 72, 578–586. doi:10.1097/PSY.0b013e3181de7ff8
Ussher, J.M., Tim Wong, W.K., & Perz, J. (2011). A qualitative analysis of changes in relationship dynamics and roles between people with cancer and their primary informal carer. Health, 15, 650–667.
Vandenbroucke, J.P., von Elm, E., Altman, D.G., Gotzsche, P.C., Mulrow, C.D., Pocock, S.J., . . . Egger, M. (2014). Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): Explanation and elaboration. International Journal of Surgery, 12, 1500–1524. doi:10.1016/j.ijsu.2014.07.014
Van Onselen, C., Paul, S.M., Lee, K., Dunn, L., Aouizerat, B.E., West, C., . . . Miaskowski, C. (2013). Trajectories of sleep disturbance and daytime sleepiness in women before and after surgery for breast cancer. Journal of Pain and Symptom Management, 45, 244–260.
World Medical Association. (2008). WMA Declaration of Helsinki: Ethical principles for medical research involving human subjects. Retrieved from http://www.wma.net/en/30publications/10policies/b3/index.html
About the Author(s)
Kotronoulas is a research fellow in the School of Health Sciences at the University of Surrey in Glasgow, Scotland; Wengström is a professor of nursing in the Department of Neurobiology, Care Science, and Society at the Karolinska Institutet in Huddinge, Sweden; and Kearney is professor emeritus in the School of Health Sciences at the University of Surrey in Guildford, England. This research was funded by a PhD scholarship from the Alexander S. Onassis Public Benefit Foundation and an education grant from the Hellenic Society of Medical Oncology in Athens, Greece. Mention of specific products and opinions related to those products do not indicate or imply endorsement by the Oncology Nursing Forum or the Oncology Nursing Society. Kotronoulas, Wengström, and Kearney contributed to the conceptualization and design. Kotronoulas completed the data collection, provided statistical support, and contributed to the analysis. Kotronoulas, Wengström, and Kearney contributed to the manuscript preparation. Kotronoulas can be reached at g.kotronoulas@surrey.ac.uk, with copy to editor at ONFEditor@ons.org. Submitted April 2015. Accepted for publication June 15, 2015.

