Risk Factors for Re-Excision Following Breast-Conserving Surgery
Purpose/Objectives: To identify previously unstudied factors predicting re-excision following breast-conserving surgery (BCS) and to assess the feasibility of obtaining data about breast density for predictive modeling.
Design: Retrospective secondary data analysis.
Setting: Data were obtained from the cancer registry and electronic health records (EHRs) at Texas Health Harris Methodist Hospital, a large, urban, private, nonprofit hospital in North Texas.
Sample: 244 patients choosing BCS from 2011–2012.
Methods: Data were subjected to univariate analyses (chi-square) followed by logistic regression.
Main Research Variables: The primary dependent variable was re-excision following BCS. Predictors of interest included lifestyle factors, time from diagnosis to surgery, surgical approach, patient age, and breast density, and controlled for covariates, such as assay results.
Findings: Three factors predicted re-excision with 87% accuracy: time from diagnosis to surgery, needle localization, and age. Missing data precluded using breast density as a predictor.
Conclusions: Women younger than 60 years whose surgery included placement of a wire for localization of tissue to be removed and who underwent surgery soon after diagnosis are the least likely to require reoperation after BCS. Data integrity is critical to the success of research using EHRs and registry information.
Implications for Nursing: Nurses may improve patient outcomes by helping women considering BCS solve problems that may delay surgery. Nurses can contribute to the success of nursing research by thoroughly and accurately recording patient information in EHRs.
Jump to a section
Breast-conserving surgeries (BCSs) are excisions of breast tissue that are not full mastectomies. Early-stage invasive breast cancer and ductal carcinoma in situ (DCIS) (i.e., cancer of the ductal elements of the breast that has not invaded beyond the ducts) (Dudley & Zuckerman, 2013) can be successfully treated with breast-conserving therapy and postoperative radiation (Hunt, Robb, Strom, & Ueno, 2008). However, some patients who undergo breast-conserving treatment may need to have a second excision or mastectomy to obtain clear margins or address complications. Clear margin refers to a margin of healthy cells surrounding the area of the tumor (Benedet & Rounsaville, 2004).
Data from diverse studies across the globe indicate that 7%–30% of patients undergo re-excision, with an average of about 20%. The re-excision rate is even higher for DCIS cases, with rates closer to 30%–60% of patients (Devouge et al., 2013; Jeevan et al., 2012; McCahill et al., 2012; Meier-Meitinger et al., 2012; Mullen et al., 2012; Unzeitig et al., 2012). Although the American Society of Breast Surgeons (2013) cautions against using re-excision rates as a quality measure, this practice is supported by the European Society of Breast Cancer Specialists and others. Using re-excision rates as a quality measure in the future could have the effect of targeting reductions in re-excision rates for healthcare reimbursement. Therefore, healthcare facilities, payers, and healthcare providers may have a strong interest in predictors of re-excision after BCS.
Review of available literature (since 1998) in MEDLINE®, CINAHL® Plus, and Health Source: Nursing/Academic Edition databases revealed disagreement about risk factors for re-excision, but some consensus exists. Most studies agreed that multifocality of tumors, infiltrative tumor borders, large tumor size, and younger age predict re-excision (Devouge et al., 2013; Jung et al., 2012; Lovrics et al., 2009; Meier-Meitinger et al., 2012). Some studies also link palpable tumors to increased risk for positive margins (Kotwall, Ranson, Stiles, & Hamann, 2007; Lovrics et al., 2009).
Increased risk associated with younger age and tumor palpability suggests that high breast density may be a risk factor for re-excision. Breast density refers to the ratio of fatty tissue to fibroglandular tissue in the breast. Mammographic density is breast density determined using mammography and is not directly related to palpable density. Breast density is an independent risk factor for breast cancer, and the sensitivity of mammography is less when breasts are dense (Mason, Yokubaitis, Howard, Shah, & Wang, 2015). Younger women have been established to have greater breast density than postmenopausal women (Checka, Chun, Schnabel, Lee, & Toth, 2012; Pearce et al., 2012). Only one published study was found that evaluated breast density as a risk factor for re-excision, finding breast density to be significant with an odds ratio (OR) of 3.2 (95% confidence interval [1.2, 11]) for women with extremely dense breasts (Jung et al., 2012).
Evidence that smoking and high body mass index (BMI) are significantly associated with surgical site infection in breast surgeries has been presented (Angarita et al., 2011), but no previous study was found that evaluated the relationship between re-excision rates and BMI or tobacco use. Alcohol consumption has been linked to survival after breast cancer diagnosis (Larsen et al., 2015) but has not been examined for a relationship to re-excision after BCS.
Margin Status and Re-Excision
Because BCS preserves natural breast tissue, the entire tumor must be removed to prevent local recurrence. Margins are the rims of healthy or normal tissue surrounding the excised tumor specimen. Margins may be identified in several ways, including the use of colored inks applied to tumor edges or submission of margins as separate specimens (Lester et al., 2009). Pathology reports describe margins as positive if cancer cells are present. A negative or clear margin has no cancer-like cells present. When margins are positive, a second excision to remove any remaining cancer is warranted (Houssami et al., 2010). In the current study, re-excision was defined as a second operative procedure performed on the same or both breasts and related to either positive margins or complications, such as surgical site infections or fat necrosis, from the first breast operation.
Needle Localization
Needle localization employed during BCS may predict re-excision because it enables the surgeon to more precisely find the target tissue to be excised. A radiologist will perform a needle localization when tumors are not palpable. Using a mammogram, ultrasound, or breast magnetic resonance imaging as a guide, the radiologist inserts a very thin needle (wire) into the breast in the area of the cancer. The surgeon then uses the wire as a guide to find and remove the tumor during surgery. The wire is then removed (Oncolink, 2016).
Significance
Undergoing a second operation to treat early-stage breast cancer is unfavorable because multiple procedures result in increased costs, risks for surgical complications, and psychological stress. In North Texas, the estimated total hospital cost of a partial mastectomy (lumpectomy) is about $17,500 before physician’s fees are added (personal communication, Texas Health Resources Billing Department, 2014). If a re-excision is necessary, the cost would be equivalent to the initial operation. If the second operation is a mastectomy, costs quickly escalate. The estimated total hospital cost for a simple mastectomy is $30,700 (personal communication, Texas Health Resources Billing Department, 2014). In addition to increased costs, risks for surgical complications are experienced for each surgical procedure.
Potential complications following any breast surgery include infection, chronic pain, numbness or tingling, lymphedema, and cardiopulmonary problems (Hill-Kayser, Vachani, Hampshire, Di Lullo, & Metz, 2012; Vitug & Newman, 2007). Because standard of care for patients undergoing BCS is receiving radiation therapy after surgery, loss of skin flexibility to irradiated areas may occur (Hill-Kayser et al., 2012; Vitug & Newman, 2007). Reoperation after BCS has also been found to be significantly associated with postoperative wound complications (Lyle et al., 2014). Previous research documents the dissatisfaction of patients who experience multiple related surgeries (Janes, Stankhe, Singh, & Isgar, 2006).
Objectives
Evidence indicates that modifiable lifestyle factors are related to surgical site infections, breast cancer incidence, or mortality, but evidence linking lifestyle factors to re-excision after BCS is lacking (Angarita et al., 2011; Boone et al., 2015). Because lifestyle factors are modifiable and potentially subject to the influence of nursing intervention, the primary purpose of the current study was to assess the associations of lifestyle factors, including BMI and tobacco or alcohol use, with risk for re-excision among women choosing BCS for breast cancer. The secondary objective was to assess the feasibility of obtaining breast density data to include in modeling as a risk factor for re-excision. Based on previous literature, patients with dense breasts were expected to have a greater likelihood of re-excision. Awareness of risk factors for re-excision could be used to inform care recommendations for patients who are eligible for BCS.
Methods
The current study was a retrospective cohort study, which used cancer registry data available at a 726-bed urban, private, nonprofit hospital, Texas Health Harris Methodist Hospital, in North Texas. It was reviewed and approved by the institutional review board governing research with human participants for the healthcare system to which the target hospital belonged, Texas Health Resources.
Sample
The sample included records of any woman who underwent BCS in 2011–2012 at the study hospital. Patients with late-stage cancer (stage III or IV) or with metastases at diagnosis were excluded. The target cancer registry database uses the software OncoLog to store data and has been abstracting patient data from charts into the electronic database since 2005. Because of coding changes in the Facility Oncology Registry Data Standards in 2006 and 2010 (Commission on Cancer, 2010), and an update in cancer staging in 2010 (American Joint Commission on Cancer, 2010), the cancer registry does not have consistent data collected during those years. Because of this limited availability of certain data fields in years prior to 2010, the sample was limited to cases involving patients who received BCS from 2011–2012. From 2011–2012, 244 patients at the study hospital had an initial surgical approach of BCS; therefore, records of 244 patients were included.
Data Collection
The majority of the data evaluated for the current study was readily available in the cancer registry’s database, including tumor grade, date of diagnosis, primary tumor site, family history, International Classification of Diseases for Oncology histology/behavior, age at diagnosis, genetic testing, managing physician, biopsy procedure, tumor palpability, estrogen receptor assay, progesterone receptor assay, size of invasive tumor component, human epidermal growth factor receptor 2 (HER2) testing summary, lymph node evaluation, presence of metastasis at diagnosis, all clinical and pathologic staging, tumor size, p53 test result, height, weight, tobacco use, alcohol use, surgical margins, and surgical approach. Tobacco use and alcohol use were entered into the registry as categorical variables (current use, previous use, never used) and, therefore, were analyzed as categories. Manual chart review was required to obtain information directly from mammography reports for breast density data because the cancer registry does not collect that information.
One of the investigators queried the cancer registry’s database for patients who had BCS as the initial approach from 2011–2012. Data were extracted, de-identified, and collected in the study’s electronic database. Access to the radiologic record was only possible using the hospital’s electronic health record (EHR). An approved (by the hospital system) data manager extracted mammography reports for the patients in the study cohort.
Statistical Analysis
The variables previously mentioned were analyzed to determine which ones had a statistically significant relationship with re-excision. Factors that attained statistical significance in univariate analysis (chi-square for categorical data and t tests or Mann–Whitney U for continuous variables) were used as explanatory variables in a logistic regression. All records that fit the inclusion criteria (244 cases) were analyzed. Logistic regression was performed with SPSS®, version 20.0, using re-excision as the binary outcome variable.
Results
Characteristics of the sample are presented in Table 1. Average age in the total sample was 66 years (SD = 11.8), and the age distribution was skewed toward older women. The sample was also skewed toward those who were overweight or obese. The average BMI for the sample was 29.57, close to the boundary between categories of overweight and obese (BMI greater than 30). About 70% of the sample (n = 171) was over the threshold for overweight, and 95 women (39%) fell into the obese category. 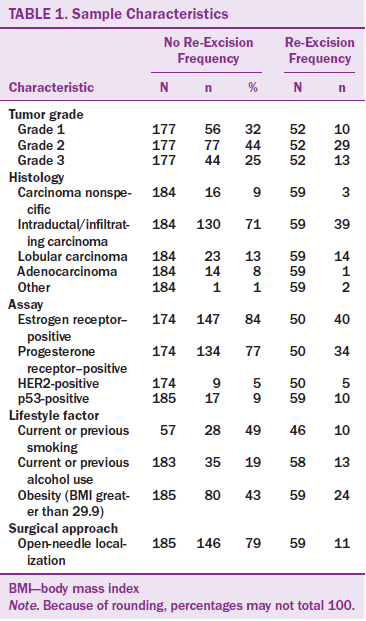
The re-excision rate for 2011–2012 at the study hospital was 25%, comparable to the re-excision rate (23%) in a study of 2,206 women who experienced partial mastectomies from three large U.S. health plans and a U.S. university hospital (McCahill et al., 2012). Forty-nine of the 60 patients who required additional surgery were subsequently treated with mastectomies rather than additional attempts at breast conservation. The high rate of treatment with mastectomies may be attributed to regional preference because more women are still treated with mastectomies rather than BCS in the southern United States, although they may be eligible for lumpectomies (Benedet & Rounsaville, 2004).
Variables were chosen from the cancer registry for their potential effects on the outcome of re-excision. The majority of these variables were found to have no significant effect on relevant outcomes. Significant factors are shown in Table 2. Of note, the lifestyle factors of BMI, smoking status, and alcohol consumption did not affect the target outcome in the sample. 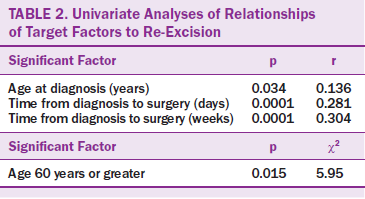
Volume of practice by breast surgeons at the study center did not influence re-excision. Surgical care was provided by 17 surgeons, with a single surgeon providing 55% of procedures. No statistically significant relationship was found between re-excision following surgeries performed by high-volume surgeons and those performed by surgeons who operated less frequently. No significant difference was seen in the re-excision rates between surgeons when surgeons were compared individually, nor were any associations made between re-excision and other (nonsurgeon) physicians.
A model predicting the odds for re-excision was developed using investigator-entered variables of interest. Target variables included all variables that were available and may plausibly link to complications resulting in re-excision. Examined variables included tumor grade, tumor size, tumor histology, tumor palpability, results of genetic assays (estrogen receptor, progesterone receptor, HER2, p53), surgical approach, surgeon, tobacco use, alcohol use, BMI, age at diagnosis, time from diagnosis to surgery, and margin status. The model is defined by three significant variables discovered in univariate analysis: surgical approach (needle localization), time from diagnosis to surgery, and age older than 60 years. Age at diagnosis became insignificant when combined with other predictors. The model explains about 30% of the variance in re-excision. Using these variables, the model has an overall accuracy for prediction of 87% but is not sensitive. The model is exceptionally specific in that it was able to predict those who would not need re-excision 99% of the time, but it only correctly identified 24% of patients who experienced re-excision. Factors excluded from the model have smaller effects than could be detected with this sample size, or, more likely, additional factors that also determine re-excision were not evaluated in this project.
Positive relationships were found between re-excision and the factors of surgical approach (needle localization) and participants aged older than 60 years. The OR for surgery with needle localization was 8.452; therefore, those who had surgery with needle localization were about 8.5 times more likely to have re-excisions than those who had surgery with no needle localization. Because needle localization is not used for palpable lesions, this variable may be a proxy for tumor size or more complicated situations. The OR for those aged older than 60 years was 1.816; therefore, women aged older than 60 years were almost twice as likely to experience re-excisions in the current sample.
The final prediction equation for this model is:
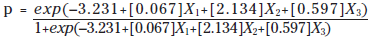
Variable X1 is the number of weeks from diagnosis to surgery. Variable X2 is whether the surgical approach included needle localization, and variable X3 is whether the patient is aged older than 60 years. Therefore, a 65-year-old woman whose surgical approach includes needle localization and who receives surgery six weeks after diagnosis has a probability of re-excision of only about 9%. Waiting 40 weeks for her surgery would result in a probability of re-excision of about 69%. Only 40 cases (16%) could be linked with breast density data. This sample of 40 breast density classifications was too small to contribute to analysis.
Discussion
The data indicate that time from surgery to diagnosis, needle localization, and age predict re-excision after BCS (i.e., women aged younger than 60 years with a needle localization surgical approach who undergo surgery soon after diagnosis are the least likely to require reoperation after BCS).
In most respects, the sample appeared to be representative of cases in private practices in the North Texas region. The majority of participants were Caucasian, and the proportion of obese women in the sample was similar to the Texas population, which was composed of 66% overweight adults and 28% obese adults during the study period (Centers for Disease Control and Prevention, 2012).
At the study center, 84% of BCSs were performed by four surgeons. A lack of relationship between surgeon experience at the center and re-excision, as well as similar re-excision rates associated with different types of physicians (e.g., surgeons, medical oncologists) at the site probably indicates substantial homogeneity in disease management and surgical approach among relevant medical care providers in the region. The pattern of practice at the center may not be representative of all breast cancer centers but is common in the region.
Because age is one of the most important risk factors for breast cancer (Hunt et al., 2008), the sample being skewed toward older women was not surprising, with 163 patients (67%) being aged older than 60 years. The findings in the current study are not consistent with findings in previous studies that document a greater risk of re-excision in younger women after BCS (Bani et al., 2009; Janes et al., 2006; Jung et al., 2012). Contrary to some findings (Aziz et al., 2006; Jung et al., 2012), the data from this project do not strongly support using age as an independent risk factor for re-excision.
Age may instead reflect the different types of cancer experienced by younger and older women, as younger women often experience more serious breast cancers than older women (Anders, Johnson, Litton, Phillips, & Bleyer, 2009). Age could possibly be related to menopausal status as well, which would support the idea that breast density may be at play because menopausal status readily affects breast density (Kerlikowske et al., 2010). However, evidence of the relationship between breast density and re-excision, if any, must wait on additional investigations. In the meantime, age is also likely to be a proxy for other, more precise predictors, including frailty, immunosuppression, comorbidities, and depression.
The relationship between length of time (in weeks) from diagnosis and surgery to re-excision was small (OR = 1.069). However, in the current sample, some patients waited as long as a year after diagnosis for their BCS. For each week that passed from diagnosis to surgery, the odds of needing re-excision increased about 7%. Over time, the effect became substantial. An interview with the breast cancer nurse navigator in the study center indicated that the most common reasons for postponing surgery included cultural and language barriers, lack of understanding of risk, and financial problems, usually lack of adequate insurance benefits (personal communication, Dana McQuirk, 2013). For instance, patients with high-deductible insurance may be insured but unable to pay the deductible for breast surgery. Although the study center has funding to assist with paying for screening mammograms and diagnostic workup for abnormal findings, accessing care for surgical biopsies and subsequent treatment can cause delays.
The model presented in this article could be used to predict lack of need for re-excision in selected patients with confidence, which may be reassuring to patients. If re-excision were predicted using this model, it could cue patients and care providers to consider surgical options other than BCS or to use the approach with caution. The prediction of re-excision is quite poor in this model; therefore, additional factors must be considered in future predictive models.
Evaluating the impact of breast density on re-excision should be easier in Texas than in some other states because Henda’s Law was enacted in Texas in 2011 (Green, 2016). This legislation requires information about breast density to be included in mammogram reports. However, breast density data were disappointing. Some women in the sample obtained mammograms at agencies outside the study healthcare system, and their mammogram reports were not available in the registry or hospital EHR. Also, not every mammogram report contained breast density data because full compliance with Henda’s Law was not required until January 2012. Therefore, feasibility of collecting breast density data remains questionable. Although these data should always appear on mammogram reports generated after January 2012 in Texas, difficulties obtaining reports from different healthcare systems may remain a problem.
Limitations
The current study highlights problems with the use of hospital cancer registry data for research. For instance, patients who were initially diagnosed and received BCS at the study hospital but subsequently received re-excision surgery elsewhere would not have their re-excision documented in the data gathered. Although the estimated number of patients having re-excision elsewhere in this population is low, the actual proportion of patients who go on to have reoperations outside the study system is unknown. Additional research on this topic would benefit from use of regional, rather than hospital or hospital system, registries.
Integrity of self-reported data may also be an issue. Reporting of tobacco and alcohol use is based on patient honesty and, therefore, may not be accurate. In addition, registry data used in the current study are imprecise regarding dosage of lifestyle factors. For instance, patients report if they ever smoked, currently smoke, or never smoked. No dose measures, such as pack-years, are recorded, and the same situation exists for use of alcohol. In addition, although breast density was a variable of great interest, the authors were unable to include it in the model because of missing data.
One notable limitation of the current study is sampling. As with many convenience samples, the current study may have a selection bias unique to women who choose to have surgery at the particular hospital being studied. A chance exists that factors related to choosing the study hospital as a treatment center may influence re-excision. A greater issue with the sampling is the size of the sample available for evaluation. If the true relationship between the factors not included in the model and re-excision is small but still significant, it will not likely be shown in the authors’ analysis because of lack of power created by the small sample size and lack of data availability.
Implications for Nursing
Results of the current study highlight two critically important implications for oncology nurses. First, all nurses who interact with EHRs play an important, often underappreciated, role in guaranteeing the integrity of data that appear in EHRs and registries. Generating new knowledge in nursing is critically important for the profession, but missing data in EHRs and registries often preclude answering research questions.
The data from this sample highlight the importance of reducing time from diagnosis to BCS. Nurses can help promote positive outcomes for patients by assessing reasons for delaying BCS and intervening when they can be influential to ensure as little delay as possible from diagnosis to surgery. Oncology nurse navigators and oncology nurses in ambulatory or screening settings have the opportunity to affect this goal greatly by helping patients understand risks and the importance of prompt treatment, overcome healthcare and insurance system barriers to surgery, and close gaps in compliance behaviors because of cultural and language barriers or lack of knowledge. These activities involve a high degree of nurse awareness and excellent patient education tailored to individual patient characteristics. Among other educational activities available to nurses, the model generated by the current study can be used to show patients how surgery delay relates to other factors to influence the risk for reoperation. 
Conclusion
Results from the current study indicate that time from diagnosis to surgery, needle localization, and age are predictive of the need for re-excision following BCS. In the absence of randomized, controlled trials, which would be unethical in this situation, a thorough understanding of predictors for re-excision will require multiple associational studies. The popularity of registry and EHR data for associational study purposes will increase as analytic methods continue to improve, but the quality of findings depends heavily on the quality of the data recorded. The current study demonstrates that, although simple predictive models can be constructed using almost any registry data, more sensitive models await the inclusion of improved measures. Including diagnostic reports, images, and more precise measures of already existing variables, such as lifestyle activities, could improve predictive capacity. For breast cancer specifically, measures of breast density would be a welcome addition to registry variables. Nurses who record patient information electronically have a substantial influence on data integrity and directly affect the validity of associational research that uses precollected data, such as the current study.
About the Author(s)
Rodriguez was a cancer registry supervisor at the Dignity Health Marian Regional Medical Center in Santa Maria, CA; Wilkins is a clinical education specialist and Newcomb is a nurse scientist, both at the Texas Health Harris Methodist Hospital in Fort Worth; Gwirtz is a professor and associate dean of the Graduate School of Biomedical Sciences at the University of North Texas in Fort Worth; and Skrine is a general surgeon at the Texas Health Physicians Group in Fort Worth. No financial relationships to disclose. Gwirtz and Skrine contributed to the conceptualization and design. Rodriguez and Skrine completed the data collection. Wilkins, Newcomb, and Skrine provided statistical support. Newcomb, Gwirtz, and Skrine provided the analysis. All of the authors contributed to the manuscript preparation. Newcomb can be reached at patricianewcomb@texashealth.org, with copy to editor at ONFEditor@ons.org. Submitted May 2016. Accepted for publication August 25, 2016.
References
American Joint Commission on Cancer. (2010). AJCC cancer staging manual (7th ed.). New York, NY: Springer.
American Society of Breast Surgeons. (2013). Position statement on breast cancer lumpectomy margins. Retrieved from https://www.breastsurgeons.org/new_layout/about/statements/PDF_Statemen…
Anders, C.K., Johnson, R., Litton, J., Phillips, M., & Bleyer, A. (2009). Breast cancer before age 40 years. Seminars in Oncology, 36, 237–249. doi:10.1053/j.seminoncol.2009.03.001
Angarita, F.A., Acuna, S.A., Torregrosa, L., Tawil, M., Escallon, J., & Ruiz, Á. (2011). Perioperative variables associated with surgical site infection in breast cancer surgery. Journal of Hospital Infection, 79, 328–332. doi:10.1016/j.jhin.2011.08.006
Aziz, D., Rawlinson, E., Narod, S.A., Sun, P., Lickley, H.L., McCready, D.R., & Holloway, C.M. (2006). The role of reexcision for positive margins in optimizing local disease control after breast-conserving surgery for cancer. Breast Journal, 12, 331–337. doi:10.1111/j.1075-122X.2006.00271.x
Bani, M.R., Lux, M.P., Heusinger, K., Wenkel, E., Magener, A., Schulz-Wendtland, R., . . . Fasching, P.A. (2009). Factors correlating with reexcision after breast-conserving therapy. European Journal of Surgical Oncology, 35, 32–37. doi:10.1016/j.ejso.2008.04.008
Benedet, R., & Rounsaville, M.C. (2004). Understanding lumpectomy. Omaha, NE: Addicus Books.
Boone, S.D., Baumgartner, K.B., Baumgartner, R.N., Connor, A.E., John, E.M., Giuliano, A.R., . . . Slattery, M.L. (2015). Active and passive cigarette smoking and mortality among Hispanic and non-Hispanic white women diagnosed with invasive breast cancer. Annals of Epidemiology, 25, 824–831. doi:10.1016/j.annepidem.2015.08.007
Centers for Disease Control and Prevention. (2012). Overweight and obesity. Texas: State nutrition, physical activity, and obesity profile. Retrieved from http://www.cdc.gov/obesity/stateprograms/fundedstates/pdf/texas-state-p…
Checka, C.M., Chun, J.E., Schnabel, F.R., Lee, J., & Toth, H. (2012). The relationship of mammographic density and age: Implications for breast cancer screening. AJR: American Journal of Roentgenology, 198(3), W292–W295. doi:10.2214/AJR.10.6049
Commission on Cancer. (2010). FORDS: Facility Oncology Registry Data Standards revised for 2010. Retrieved from https://www.facs.org/~/media/files/quality%20programs/cancer/coc/fords/…
Devouge, P., Phalippou, J., Martin de Beauce, S., Kerdraon, O., Prolongeau, J.F., Collinet, P., . . . Boulanger, L. (2013). Reexcision for positive margins in the surgery of ductal carcinoma in situ: Are there any risk factors? Gynécologie, Obstétrique and Fertilité, 41, 228–234. doi:10.1016/j.gyobfe.2013.02.007
Dudley, S., & Zuckerman, D. (2013). DCIS, LCIS, pre-cancer and other “stage zero” breast conditions: What kind of treatment—if any—is needed? Retrieved from http://www.stopcancerfund.org/p-breast-cancer/dcis-lcis-pre-cancer-and-…
Green, V.L. (2016). Mammographic breast density and breast cancer risk: Implications of the breast density legislation for health care practitioners. Clinical Obstetrics and Gynecology, 59, 419–438. doi:10.1097/GRF.0000000000000192
Hill-Kayser, C.E., Vachani, C., Hampshire, M.K., Di Lullo, G.A., & Metz, J.M. (2012). Cosmetic outcomes and complications reported by patients having undergone breast-conserving treatment. International Journal of Radiation Oncology, Biology, Physics, 83, 839–844. doi:10.1016/j.ijrobp.2011.08.013
Houssami, N., Macaskill, P., Marinovich, M.L., Dixon, J.M., Irwig, L. Brennan, M.E., & Solin, L.J. (2010). Meta-analysis of the impact of surgical margins on local recurrence in women with early-stage invasive breast cancer treated with breast-conserving therapy. European Journal of Cancer, 46, 3219–3232. doi:10.1016/j.ejca.2010.07.043
Hunt, K.K., Robb, G.L., Strom, E.A., & Ueno, N.T. (2008). Breast cancer (2nd ed.). New York, NY: Springer Science+Business Media.
Janes, S.E., Stankhe, M., Singh, S., & Isgar, B. (2006). Systematic cavity shaves reduces close margins and re-excision rates in breast conserving surgery. Breast, 15, 326–330. doi:10.1016/j.breast.2005.10.006
Jeevan, R., Cromwell, D.A., Trivella, M., Lawrence, G., Kearins, O., Pereira, J., . . . van der Meulen, J.H. (2012). Reoperation rates after breast conserving surgery for breast cancer among women in England: Retrospective study of hospital episode statistics. BMJ, 345, e4505. doi:10.1136/bmj.e4505
Jung, W., Kang, E., Kim, S.M., Kim, D., Hwang, Y., Sun, Y., . . . Kim, S.W. (2012). Factors associated with re-excision after breast-conserving surgery for early-stage breast cancer. Journal of Breast Cancer, 15, 412–419. doi:10.4048/jbc.2012.15.4.412
Kerlikowske, K., Cook, A.J., Buist, D.S., Cummings, S.R., Vachone, C., Vacek, P., & Miglioretti, D.L. (2010). Breast cancer risk by breast density, menopause, and postmenopausal hormone therapy use. Journal of Clinical Oncology, 28, 3830–3837. doi:10.1200/JCO.2009.26.4770
Kotwall, C., Ranson, M., Stiles, A., & Hamann, M.S. (2007). Relationship between initial margin status for invasive breast cancer and residual carcinoma after re-excision. American Surgeon, 73, 337–343.
Larsen, S.B., Kroman, N., Ibfelt, E.H., Christensen, J. Tjonneland, A., & Dalton, S.O. (2015). Influence of metabolic indicators, smoking, alcohol, and socioeconomic position on mortality after breast cancer. Acta Oncologica, 54, 780–788. doi:10.3109/0284186X.2014.998774
Lester, S.C., Bose, S., Chen, Y.Y., Connolly, J.L., de Baca, M.E., Fitzgibbons, P.L., . . . Winer, E. (2009). Protocol for the examination of specimens from patients with invasive carcinoma of the breast. Archives of Pathology and Laboratory Medicine, 133, 1515–1538.
Lovrics, P.J., Cornacchi, S.D., Farrokhyar, F., Garnett, A., Chen, V., Franic, S., & Simunovic, M. (2009). The relationship between surgical factors and margin status after breast-conservation surgery for early stage breast cancer. American Journal of Surgery, 197, 740–746. doi:10.1016/j.amjsurg.2008.03.007
Lyle, B.A., Landercasper, J., Johnson, J.M., Al-Hamadani, M., Vang, C.A., Zutavern, K., . . . Linebarger, J. (2014). Wound complications following breast conserving surgery (BCS)–A quality improvement study. 2014 Official Proceedings of Annual 15th Meeting of the American Society of Breast Surgeons, 78.
Mason, C., Yokubaitis, K., Howard, E., Shah, Z., & Wang, J. (2015). Impact of Henda’s law on the utilization of screening breast magnetic resonance imaging. Proceedings, 28, 7–9.
McCahill, L.E., Single, R.M., Aiello Bowles, E.J., Feigelson, H.S., James, T.A., Barney, T., . . . Onitilo, A.A. (2012). Variability in reexcision following breast conservation surgery. JAMA, 307, 467–475.
Meier-Meitinger, M., Rauh, C., Adamietz, B., Fasching, P.A., Schwab, S.A., Haeberle, L., . . . Heuginger, K. (2012). Accuracy of radiological tumour size assessment and the risk for re-excision in a cohort of primary breast cancer patients. European Journal of Surgical Oncology, 38, 44–51. doi:10.1016/j.ejso.2011.10.008
Mullen, R., Macaskill, E.J., Khalil, A., Elseedawy, E., Brown, D.C., Lee, A.C., . . . Thompson, A.M. (2012). Involved anterior margins after breast conserving surgery?: Is re-excision required? European Journal of Surgical Oncology, 38, 302–306. doi:10.1016/j.ejso.2012.01.004
Oncolink. (2016). Surgical procedures: Needle localization partial mastectomy. Retrieved from http://www.oncolink.org/types/article.cfm?c=781&id=6972
Pearce, M.S., Tennant, P.W., Mann, K.D., Pollard, T.M., Mclean, L., Kaye, B. & Parker, L., (2012). Lifecourse predictors of mammographic density: The Newcastle Thousand Families cohort study. Breast Cancer Research and Treatment, 131, 187–195. doi:10.1007/s10549-011-1708-7
Unzeitig, A., Kobbermann, A., Xie, X.J., Yan, J., Euhus, D., Peng, Y., . . . Rao, R. (2012). Influence of surgical technique on mastectomy and reexcision rates in breast-conserving therapy for cancer. Retrieved from https://www.hindawi.com/journals/ijso/2012/725121/
Vitug, A.F., & Newman, L.A. (2007). Complications in breast surgery. Surgical Clinics of North America, 87, 431–451. doi:10.1016/j.suc.2007.01.005

