Effects of Written Information and Counseling on Illness-Related Uncertainty in Women With Vulvar Neoplasia
Objectives: To determine whether written information and/or counseling decreases illness-related uncertainty in women with vulvar neoplasia.
Sample & Setting: 49 women with vulvar neoplasia from four Swiss hospitals and one Austrian hospital.
Methods & Variables: A longitudinal, multicenter, randomized phase 2 study was performed. The written information group received a set of leaflets. The counseling group received five consultations with an advanced practice nurse (APN) from diagnosis to six months postsurgery that focused on symptom self-management, healthcare services, and decision making. Uncertainty was measured as a secondary outcome five times by the Mishel Uncertainty in Illness Scale.
Results: Total uncertainty and the subscales of ambiguity, inconsistency, and unpredictability improved significantly over time within the counseling group but not within the written information group. In addition, counseling improved inconsistency over time, and total uncertainty, inconsistency, and unpredictability at distinct time points more efficiently than written information.
Implications for Nursing: Counseling can reduce illness-related uncertainty. APNs are valuable healthcare providers who promote women’s self-management and may support them in becoming more familiar with illness-related events and common symptoms during this rare disease.
Jump to a section
Vulvar neoplasms, including vulvar intraepithelial neoplasia (VIN) as precancerous and vulvar cancer as cancerous cellular changes in the external female genitals (Bornstein et al., 2016), are rarely occurring conditions with an annual incidence of 1–7 per 100,000 in European countries and the United States (Akthtar-Denesh, Elit, & Lytwyn, 2014; HPV Information Centre, 2017; Lai et al., 2014; Meltzer-Gunnes et al., 2017). They have high recurrence rates and rising incidence rates and increasingly affect women younger than age 60 years (Akhtar-Danesh et al., 2014; Forman et al., 2012; Meltzer-Gunnes et al., 2017; Nooij et al., 2016).
The standard therapy consists of surgical procedures, which have been fundamentally improved during the past three decades (Gray, 2010; Kaushik, Pepas, Nordin, Bryant, & Dickinson, 2014). However, affected women still experience the treatment as disfiguring, particularly if it requires extensive resections of the labia or clitoris (Senn et al., 2011). The treatment leads, on average, to 20 biopsycho-social symptoms and difficulties in daily life one week postsurgery. One of the most prevalent psychosocial symptoms is uncertainty, occurring in about 83% of women (Senn et al., 2013).
This high uncertainty prevalence corresponds to the findings of several qualitative studies. They suggest that women with vulvar neoplasia experience uncertainty concerning the meaning of their disease; the disease trajectory; diagnostic results; and the meaning, likelihood, and variability of symptoms. In addition, they are uncertain about the potential for disease transmission, progression, and recurrence, as well as their reproductive and sexual capacities after treatment completion. The diagnostic process, therapy, and system of care are experienced as highly complex; sufficient information regarding the disease, treatment, common symptoms, symptom assessment, and management is missing. These uncertainties may be reinforced by the tendency of patients, healthcare providers, and others to not talk about the disease because of its location (Barlow, Hacker, Hussain, & Parmenter, 2014; Jefferies & Clifford, 2011, 2012; Kosenko, Hurley, & Harvey, 2012; Likes, Russell, & Tillmanns, 2008; Philp, Mellon, Barnett, D’Abrew, & White, 2017; Senn et al., 2011).
Illness-related uncertainty, which is an important outcome in women with vulvar neoplasia, was framed by Mishel (1988, 1990) in the theory of uncertainty in illness. Illness-related uncertainty is the inability to structure the meaning of illness-related events cognitively because of insufficient information. Uncertainty can be experienced as four dimensions: (a) ambiguity regarding the disease state, (b) complexity regarding the treatment and care system, (c) inconsistent information (inconsistency) regarding the diagnosis and seriousness of the illness, and (d) unpredictability regarding the disease course and the prognosis. The theory has three central aspects:
• Antecedents generating uncertainty (i.e., the stimuli frame [symptom pattern, event familiarity, and event congruence], cognitive capacities, and structure providers [education, social support, and credible authorities])
• Appraisal
• Coping with uncertainty
Several descriptive studies identified factors that correlate with uncertainty in women with vulvar neoplasms. Therefore, higher uncertainty correlates with older age, married family status, less education, lower income, less quality of life, less social support, and insufficient information. In addition, those with higher symptom severity, symptom distress, psychological disruption, and lower self-control and self-care behavior experience more uncertainty (Clayton, Mishel, & Belyea, 2006; Dirksen, 2000; Farren, 2010; Hagen et al., 2015; Hall, Mishel, & Germino, 2014; Kim, Lee, & Lee, 2012; Liao, Chen, Chen, & Chen, 2008; Sammarco & Konecny, 2008; Zhang, Kwekkeboom, & Petrini, 2015). Uncertainty management in acute conditions should lead to former functionality, whereas in chronic conditions like cancer (McCorkle et al., 2011), unresolvable uncertainties should be integrated as part of the disease by self-management efforts (Mishel & Clayton, 2008). Self-management means taking responsibility for health by problem solving, decision making, resource use, patient–provider partnerships, and action planning (Lorig & Holman, 2003).
Illness-related uncertainty may be reduced by information or counseling because of its generation by insufficient information. In patients with vulvar neoplasms, effects of information-based interventions have not been tested on uncertainty, but it has been shown that such interventions positively moderate uncertainty (Christman & Cain, 2004; Stiegelis et al., 2004). Patients with female-specific cancers responded with improvement in overall uncertainty (Germino et al., 2013; Gil et al., 2006; Lebel et al., 2014), inconsistency (Chow, Chan, Chan, Choi, & Siu, 2014), and ambiguity (McCorkle et al., 2009) in some intervention studies but not in others (Chow et al., 2014; Juarez, Hurria, Uman, & Ferrell, 2013). Of note, some of these studies did not measure or report all uncertainty dimensions or include survivors several years post-treatment. Women with precancerous lesions, such as cervical intraepithelial neoplasia, have not yet been investigated. Therefore, the current evidence base is inconsistent, fragmented, and not transferable to women with vulvar neoplasia because the uncertainty experience varies with type and course of cancer (e.g., survivor uncertainty focuses on long-term side effects and recurrence) (Shaha, Cox, Talman, & Kelly, 2008). Information-based interventions could be particularly relevant for women with vulvar neoplasia because they may miss information because of the rarity of the disease.
However, in women with vulvar neoplasia, no supportive interventions have been tested. Therefore, a set of written information materials and a counseling program (WOMAN-PRO II) were developed (Kobleder et al., 2016) and tested in a randomized, controlled trial according to the framework of the Medical Research Council of the United Kingdom (Craig et al., 2013). Its primary outcome was symptom prevalence (Raphaelis et al., 2017); however, this article focuses on the study’s secondary outcome of illness-related uncertainty and aims to do the following:
• Explore whether uncertainty decreases with each intervention from diagnosis to six months postsurgery (within-group differences over time).
• Examine whether written information or counseling is more likely to reduce uncertainty from diagnosis to six months postsurgery (between-group differences over time).
• Identify at which time points from diagnosis to six months postsurgery written information or counseling is more likely to reduce uncertainty (between-group differences at distinct time points).
Methods
A longitudinal, multicenter, randomized phase 2 study with two intervention groups was conducted to test all outcomes of the WOMAN-PRO II study (clinical trial ID: NCT01986725). A phase 2 design was selected because none of the interventions had been tested before. Phase 2 studies typically include few participants and focus primarily on changes within an intervention group. They may include a randomization to examine initially potential group differences that inform a subsequent, strictly comparative phase 3 trial (Friedman, Furberg, & DeMets, 2010).
Interventions
Written information: Women were provided with standard care during hospitalization and follow-up determined by local standards and international guidelines (Schnürch et al., 2016). In addition, they received two leaflets during the time from diagnosis to surgery in accordance to the WOMAN-PRO II symptom management guideline (Kobleder, Raphaelis, et al., 2016) (see Table 1). The first leaflet was about the early pre- and postoperative phase (e.g., surgery, recommendations for wound care) and was available in three versions depending on the planned surgery (e.g., skinning vulvectomy). The second leaflet provided information about relevant healthcare services in the hospital and community (e.g., psycho-oncologists). They were written in objective, simple, concise, nonthreatening, and noncommanding language; separated facts and recommendations for readers; and outlined actuality (when materials were written and updated) and authorship. Readability and complexity were not specifically checked by non-experts, but a review with an online tool yielded a moderate reading level (Lenhard & Lenhard, 2017), which is understandable by vocational or secondary school graduates. 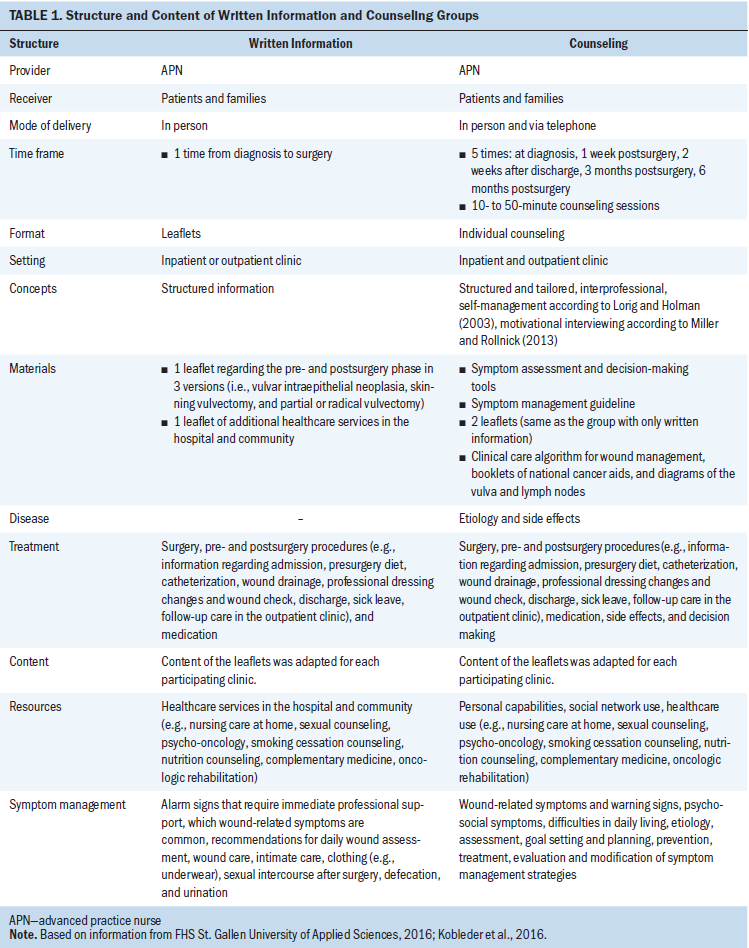
Counseling: Counseling relied on the WOMAN-PRO II symptom management guideline (Kobleder et al., 2016) and was given in addition to standard care and written information, with the difference that the written information was explained to women with consideration of their individual situations. Women received five consultations with an advanced practice nurse (APN) for 10–50 minutes after diagnosis (presurgery), one week postsurgery, two weeks after discharge, and three months and six months postsurgery complementary to physician appointments. The consultations focused on self-management (Lorig & Holman, 2003) and consisted of symptom assessment with a diary (Senn et al., 2013), symptom prevention and treatment, information materials, use of healthcare services, health-related decision making, and interprofessional coordination. Women monitored their symptoms with the diary and selected three to five symptoms they wanted to discuss with an APN. If clinically necessary, the APNs added specific symptoms (e.g., wound-related symptoms if inflammatory signs were present). The consultations were tailored to women’s needs, for example, in terms of intervention delivery (e.g., in person, via telephone) or family involvement. To ensure that all APNs applied this intervention appropriately, a 12-hour training consisting of theoretical and practical inputs was held. They were also supervised by the research team throughout the study.
For both interventions, the evidence base and development methods are published in the WOMAN-PRO II symptom management guideline (Kobleder et al., 2016).
Sample and Setting
The sample size calculation was performed for the primary outcome symptom prevalence based on the symptom scores reported in a previous study (Senn et al., 2013), yielding a required sample of 90 women with a power of 80% and an expected attrition of 15%. A randomization ratio of 2:1 (written information: n = 30; counseling: n = 60) was selected because previous research favors counseling over written information. Participants were recruited in four Swiss hospitals (University Hospital of Basel, University Hospital of Berne, Lucerne Cantonal Hospital, Cantonal Hospital of St. Gallen) and in one Austrian hospital (University Hospital of Vienna). Eligible women were aged 18 years or older and undergoing surgery for vulvar neoplasia irrespective of recurrent disease. Women with linguistic, cognitive, emotional, or physical problems that inhibited participation were excluded.
Procedures
Ethical approval for the study was received from the ethics committees at Lucerne Cantonal Hospital and Cantonal Hospital of St. Gallen and from the Ethics Committee of the University Hospital of Vienna. A computerized randomization list was generated by a statistician with blocks of three and the stratification factors VIN and vulvar cancer. Recruitment took place from September 2013 to May 2015. After receipt of written informed consent and completion of baseline data, allocation concealment was implemented by sealed, opaque envelopes, which were numbered in accordance with the randomization list and were opened in front of the participating women. All data were collected by the APNs five times from diagnosis (presurgery) to six months postsurgery, concurrent with the counseling time points. During data collection, all anonymized questionnaires were stored at the participating clinics and afterward at the research institute in locked cabinets, separately from consent forms. Blinding of participants and healthcare staff was only possible regarding the differences of the two interventions. In addition, three study centers received an external quality audit in accordance with the guidelines of the National Cancer Institute (National Clinical Trials Network, 2017). A university not involved in the study reviewed the study progress, the protocol adherence, APNs’ counseling knowledge, and the study records.
Variables and Measurements
Sample characteristics: Sociodemographic data were completed by the participating women at baseline, and medical data were completed by the APNs one week postsurgery with forms pilot tested in a previous study (Senn et al., 2013).
Illness-related uncertainty: The German adult form of the Mishel Uncertainty in Illness Scale (MUIS-A) was completed by participating women at all five time points. It has an excellent internal consistency (Cronbach alpha = 0.91) (Wallner, 2015) but has not yet been tested for validity. However, the construct validity of the original English instrument has been confirmed several times (Mishel, 1981). The questionnaire contains 33 items, which are scored on a five-point Likert-type scale ranging from 1 (strongly agree) to 5 (strongly disagree). Scores can be summed to a total scale ranging from 32–160 or four subscales (i.e., ambiguity, complexity, inconsistency, and unpredictability). Higher scores indicate higher uncertainty (Mishel, 1997).
Data Analysis
For analysis, all data were recorded in a password-protected electronic databank. To detect entry failures, 10% of the questionnaires were double-entered, yielding an entry error of 0.1%. All analysis followed the intent-to-treat maxim, including missing values and dropouts, which were not replaced because they appeared to be missing nonsystematically. Hypotheses were tested on a significance level of alpha ≤ 0.05. The uncertainty scores were summed to total scales and the four subscales. Group comparisons at baseline were computed by nonparametric tests, within-group differences over time by a one-way analysis of variance (ANOVA), and between-group differences over time by a mixed-design ANOVA with time as within-subject factor and group as between-subject factor. Between-group differences at distinct time points that may have been insignificant in the longitudinal analysis were determined by the Mann–Whitney U test as a post hoc test. In addition, effect sizes were calculated for all group comparisons.
Results
Sample Characteristics
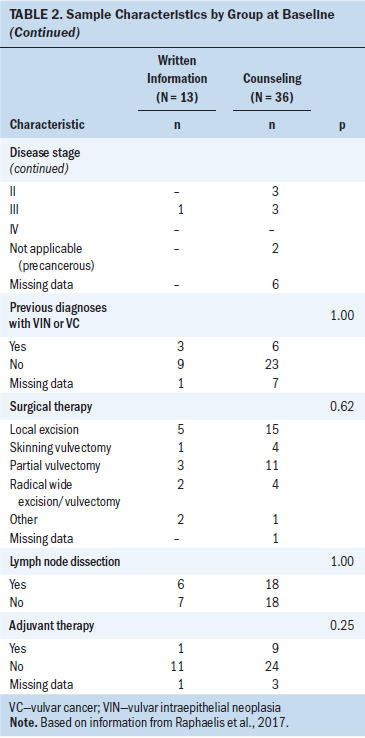
Of 117 women who were assessed for eligibility in the five clinics, 24 did not meet inclusion criteria. Of 93 eligible women, 44 declined participation because they felt overburdened or had little time, interest, or trust in research, yielding a final sample of 49 women and a recruitment rate of 52.7%. Because of the 2:1 randomization, 36 women were allocated to counseling and 13 women to written information (see Figure 1). The participants did not differ significantly in sociodemographic or medical characteristics at baseline (see Table 2). In total, 13 women dropped out because they were hospitalized in another clinic, too ill, no longer available, had family problems, or otherwise declined further participation for unknown reasons. Participants of the written information group dropped out more often, but this finding was nonsignificant (p = 0.32). The participants who dropped out were, in comparison to the counseling group, more consistently older, married, and highly educated; more likely living with others and having a child; had VIN more often; had a lower tumor stage; and generally had lower uncertainty scores. However, the participants who dropped out did not differ significantly in sample characteristics or uncertainty scores by group or compared to those who completed the study (all p > 0.05). 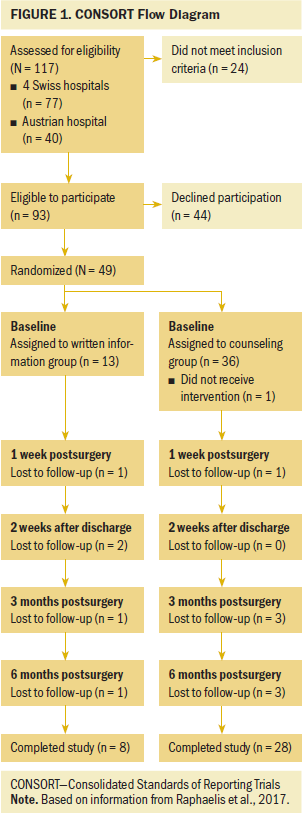
In total, 22 women were included from Austria and 27 from Switzerland. The majority of participants had vulvar cancer (n = 31), a local excision (n = 20), and inpatient surgeries (n = 43). In the written information group, women were aged an average of 57 years (median = 45, interquartile range [IQR] = 69, range = 34–78). In the counseling group, women were aged an average of 56.5 years (median = 45.5, IQR = 64.5, range = 24–81) (p = 0.62). Average length of hospitalization was 4 days for the written information group (median = 3, IQR = 7, range = 2–12) and 6.5 days for the counseling group (median = 4, IQR = 11.5, range = 1–31) (p = 0.49). 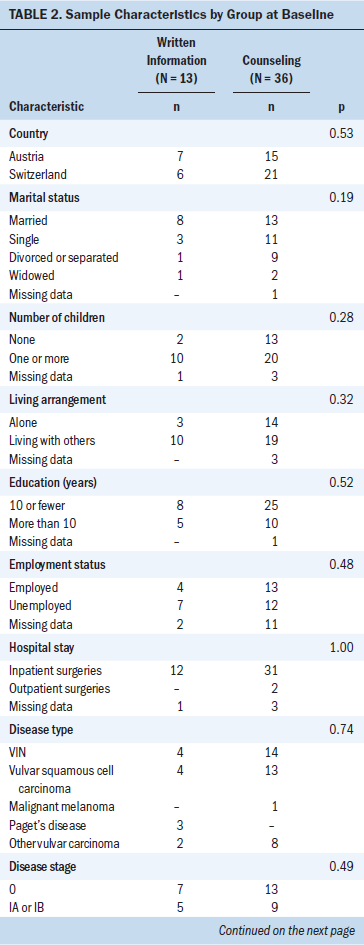
Illness-Related Uncertainty
The two groups did not differ significantly in any uncertainty scores at baseline (see Table 3). Women with written information had a total uncertainty mean of 78 (SD = 14.5), and those with counseling had a mean of 68.5 (SD = 15.6) at baseline. Women with written information experienced the lowest uncertainty at one week postsurgery (mean = 76.1, SD = 25.8) and the highest uncertainty two weeks after discharge (mean = 83, SD = 15.2). In those with counseling, the highest uncertainty was observed one week postsurgery (mean = 71.1, SD = 18.5) and the lowest was observed three (mean = 62.7, SD = 18.1) and six months postsurgery (mean = 62.8, SD = 19.6). Overall, the counseling group demonstrated a better trend of improvement in all uncertainty scales throughout the study (see Figure 2). 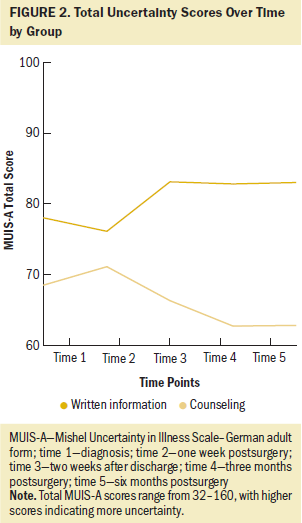
Within the counseling group, total uncertainty (F[2.02, 42.4] = 5.58, p = 0.007, eta p2 = 0.21), ambiguity (F[4, 76] = 7.76, p = 0.000, eta p2 = 0.29), inconsistency (F[4, 76] = 3.05, p = 0.022, eta p2 = 0.25), and unpredictability (F[4, 84] = 2.78, p = 0.032, eta p2 = 0.12) decreased significantly within six months. In the written information group, none of the uncertainty scales decreased significantly over time (all p > 0.05). 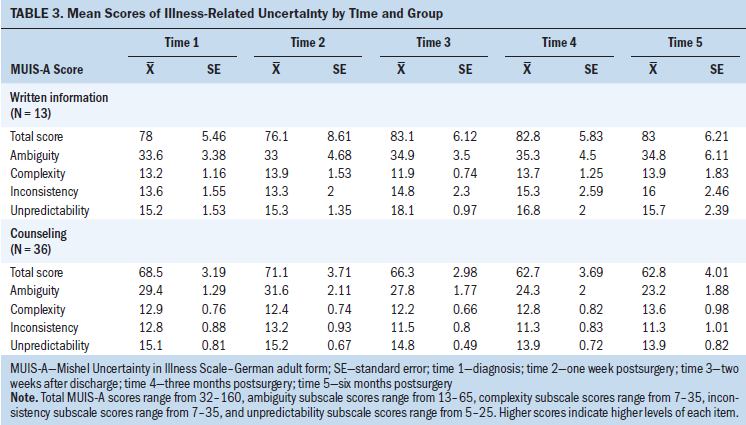
Significant group differences over time appeared in favor of counseling in the inconsistency subscale (see Table 4). In addition, counseling improved total uncertainty three months postsurgery (median = 54), inconsistency six months postsurgery (median = 8), and unpredictability two weeks after discharge (median = 14) significantly more than written information (median = 79 three months postsurgery, median = 17 six months postsurgery, median = 18 two weeks after discharge) (see Table 5). 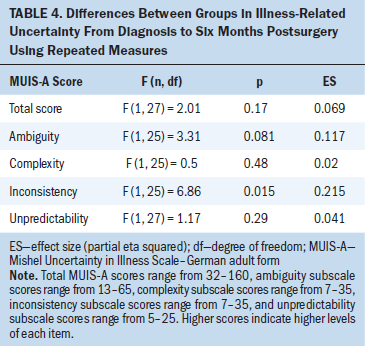
Discussion
This is the first study exploring the effect of written information and counseling on illness-related uncertainty in women with vulvar neoplasia. The authors conducted an experimental phase 2 design to discover whether uncertainty differed within intervention groups over time and between intervention groups over time and at distinct time points. 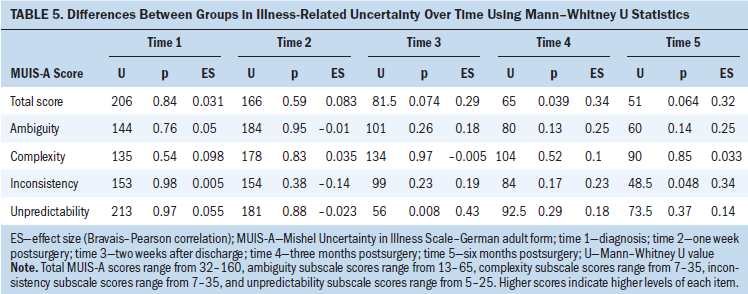
Within the counseling group, total uncertainty, ambiguity, inconsistency, and unpredictability decreased significantly from diagnosis to six months postsurgery. The written information group did not show any significant changes over time. Significant differences between the two groups appeared in favor of counseling on inconsistency over time, total uncertainty three months postsurgery, unpredictability two weeks after discharge, and inconsistency six months postsurgery.
The authors assumed that counseling was more likely to reduce uncertainty because the APNs functioned as important structure providers. Structure providers are mentioned in the theory of uncertainty in illness as the available resources for individuals in the form of education, social support, and healthcare providers. These providers support them in interpreting and managing the stimuli frame of uncertainty. The APNs as structure providers addressed two important components of the stimuli frame: symptom pattern and event familiarity. The pattern of a symptom is characterized by the frequency, intensity, duration, and localization of a symptom (Mishel & Braden, 1988). Individuals who know the usual symptom pattern can potentially anticipate the possible symptom range and required management strategies and, therefore, experience less uncertainty. During counseling in the current study, women learned about common symptoms to expect, alarm signs that indicate complications or recurrent disease, and appropriate management strategies, which may have resulted in less uncertainty. In addition, counseling may have reduced uncertainty because it established familiarity with unfamiliar illness-related events. Theoretically, individuals reach event familiarity if they share their opinions and views about unfamiliar events with others, and healthcare providers are key people in this regard according to previous research (Clayton et al., 2006; Mishel & Braden, 1988; Mishel & Clayton, 2008). This is supported by interviews with women who received counseling in the current study. Participants stated that the APNs were a key point of support—people who were there for any concerns that were difficult to talk about with a social network or healthcare providers (Kobleder, Mayer, & Senn, 2017). In particular, APNs provided discussions about the disease, the specialized healthcare system, routines, and treatments and, therefore, may have been a main source to create event familiarity. Finally, counseling may have been more likely to reduce uncertainty because it supported women’s self-management (Lorig & Holman, 2003), which represents a main focus of uncertainty management in chronic conditions (Mishel, 1990; Mishel & Clayton, 2008) and has been reported as a valuable factor in reducing uncertainty (Dirksen, 2000; Liao et al., 2008; Zhang et al., 2015). The current results add to previous research where positive effects of counseling on uncertainty were reported in women with female-specific cancers (Germino et al., 2013; Lebel et al., 2014).
When considering the results of the first study aim, the authors suggest regarding the observed significant improvements within the counseling group with caution because within-group effects generally lack strong internal validity. By contrast, the analysis corresponding to the second aim yielded the most robust finding that counseling was, over repeated measures until six months postsurgery, more efficient than written information to reduce inconsistent information. This may be because women with counseling received tailored healthcare information, which was kept consistent with other sources by documentation and APN participation on physician appointments. In contrast, women with written information received standardized material that was not consistently adjusted with other information sources. In particular, these women did not have the same chance to reach consistency by discussing remaining questions on a planned basis during counseling and joint appointments between APNs and physicians. This finding corresponds with previous research in women with female-specific cancers in which inconsistency decreased by counseling as well (Chow et al., 2014). Concerning the third study aim, the authors do not have a firm explanation for the differing time effects depending on the uncertainty scale under consideration. Total uncertainty may have been significant six months postsurgery in a larger sample because the p value was nearly significant (p = 0.064). This assumption would mean that women’s total uncertainty and experience of inconsistent information responded primarily after three to six months to counseling. The fact that unpredictability was not any more significant beyond two weeks after discharge may be because women with written information adapted increasingly to this uncertainty dimension (e.g., risk for disease recurrence). In this regard, the theory proposes that people who live with ongoing uncertainty and/or intolerable uncertainty appraise their uncertainty over time less severely by a positively oriented illusion (Mishel, 1990).
Limitations
First, the results are based on a small sample even though the authors extended recruitment for another six months and in another clinic. The project schedule did not allow a further extension, but a statistician affirmed that within-group effects in counseling could be determined by the accrued sample. Particularly, the current sample may have introduced type II errors concerning the mixed-design ANOVA and within-group analysis in the written information group. Large confidence intervals and the 9.5 difference between groups in total MUIS-A scores at baseline, although not significant, indicate that the mean estimates of the written information group are imprecise. Small samples are a widespread problem in research concerning women with vulvar neoplasia. In previous randomized, controlled trials, the average number of participants ranged from 32–62 (Manci et al., 2009; Mathiesen, Buus, & Cramers, 2007; Naik et al., 2006; Tristram et al., 2014). Although experimental research with individuals affected by rare diseases may be easily underpowered, it is important to generate some insights. This is also acknowledged in the Oncology Nursing Society research agenda, which defines the development and evaluation of self-management interventions for minority groups as a research priority (Knobf et al., 2015). Second, different clinical care in the participating clinics may have affected the findings, but intervention delivery in various settings gives the results more generalizability. Finally, the APNs might have introduced contamination bias because they performed the study for both groups.
Implications for Nursing
The results do not support providing written information alone to affected women. At a minimum, information should be explained to patients with respect to their individual situations. In contrast, the results support counseling with consideration to the following points. APNs should perform counseling because they may support patients to alter the pattern of symptoms and to create event familiarity. The intensity of counseling should be increased in the early treatment phase by another session close to and/or after surgery because uncertainty was highest at this time. During follow-up, a non–time-consuming mode of delivery, such as telephone counseling, may be sufficient because uncertainty may stabilize during later treatment. Tailoring of counseling under close interprofessional cooperation is valuable to keep it consistent with women’s needs and other healthcare measures. In particular, tailored information concerning the disease, treatment, and system of care seems to be beneficial to create familiarity with this rare disease. Specific information and psychosocial support on the risk for disease recurrence should be included to ensure that women do not adapt to the unpredictability of their disease in a dysfunctional manner. In addition, the focus on self-management should be maintained because this seems to be an important factor in reducing uncertainty. Proactive symptom management should be emphasized because it may influence the pattern of symptoms and, therefore, uncertainty.
Implications for Research
The findings of the current study justify testing counseling further in a strictly comparative phase 3 trial to estimate its whole potential. Recruitment should be extended to 18 months in more participating clinics, and data collection should be simplified by online completion and reduction of measurement points from five to three (at diagnosis, one week postsurgery, and six months postsurgery). A 1:1 randomization is essential to ensure equal groups for a minimal analysis if the required sample is not reachable. The results do not support testing written information again in a phase 3 study, but a less rigorous study design may clarify its concrete benefit. 
Conclusion
These results imply that counseling can most efficiently reduce inconsistent information in women with vulvar neoplasia and might improve total uncertainty and unpredictability at specific time points. In addition, the results guide the further development of counseling as described previously so that the best version is available for clinical practice. Future research would benefit from the development of an evidence-based symptom diary in the form of a mobile application to ensure consistency and ease of use, which would require actualizing counseling materials and optimizing readability.
About the Author(s)
Silvia Raphaelis, Mag., RN, PhD, is a student research assistant, and Hanna Mayer, Mag., RN, PhD, is a professor, both in the Department of Nursing Science at the University of Vienna in Austria; Stefan Ott, Dr. oec. HSG, Dipl.-Math. oec., is a lecturer of business mathematics and statistics at the FHS St. Gallen University of Applied Sciences in Switzerland; Rene Hornung, PhD, is a chief physician in the Department of Obstetrics and Gynecology at the Cantonal Hospital of St. Gallen in Switzerland; and Beate Senn, PhD, RN, is a professor in the Institute for Applied Nursing Sciences at the FHS St. Gallen University of Applied Sciences. This research was funded by a grant from Swiss Cancer Research (KFS-3160-02-2013). Raphaelis, Mayer, Hornung, and Senn contributed to the conceptualization and design. Raphaelis, Hornung, and Senn completed the data collection. Raphaelis, Ott, and Senn provided statistical support and the analysis. Raphaelis, Mayer, Ott, and Senn contributed to the manuscript preparation. Raphaelis can be reached at silvia.mona.raphaelis@univie.ac.at, with copy to ONFEditor@ons.org. (Submitted December 2017. Accepted May 15, 2018.)
References
Akhtar-Danesh, N., Elit, L., & Lytwyn, A. (2014). Trends in incidence and survival of women with invasive vulvar cancer in the United States and Canada: A population-based study. Gynecologic Oncology, 134, 314–318. https://doi.org/10.1016/j.ygyno.2014.05.014
Barlow, E.L., Hacker, N.F., Hussain, R., & Parmenter, G. (2014). Sexuality and body image following treatment for early-stage vulvar cancer: A qualitative study. Journal of Advanced Nursing, 70, 1856–1866. https://doi.org/10.1111/jan.12346
Bornstein, J., Bogliatto, F., Haefner, H.K., Stockdale, C.K., Preti, M., Bohl, T.G., & Reutter, J. (2016). The 2015 International Society for the Study of Vulvovaginal Disease (ISSVD) terminology of vulvar squamous intraepithelial lesions. Obstetrics and Gynecology, 127, 264–268. https://doi.org/10.1097/aog.0000000000001285
Chow, K.M., Chan, C.W., Chan, J.C., Choi, K.K., & Siu, K.Y. (2014). A feasibility study of a psychoeducational intervention program for gynecological cancer patients. European Journal of Oncology Nursing, 18, 385–392. https://doi.org/10.1016/j.ejon.2014.03.011
Christman, N.J., & Cain, L.B. (2004). The effects of concrete objective information and relaxation on maintaining usual activity during radiation therapy [Online exclusive]. Oncology Nursing Forum, 31, E39–E45. https://doi.org/10.1188/04.ONF.E39-E45
Clayton, M.F., Mishel, M.H., & Belyea, M. (2006). Testing a model of symptoms, communication, uncertainty, and well-being in older breast cancer survivors. Research in Nursing and Health, 29, 18–39. https://doi.org/10.1002/nur.20108
Craig, P., Dieppe, P., Macintyre, S., Michie, S., Nazareth, I., & Petticrew, M. (2013). Developing and evaluating complex interventions: The new Medical Research Council guidance. International Journal of Nursing Studies, 50, 587–592. https://doi.org/10.1016/j.ijnurstu.2012.09.010
Dirksen, S.R. (2000). Predicting well-being among breast cancer survivors. Journal of Advanced Nursing, 32, 937–943. https://doi.org/10.1046/j.1365-2648.2000.01559.x
Farren, A.T. (2010). Power, uncertainty, self-transcendence, and quality of life in breast cancer survivors. Nursing Science Quarterly, 23, 63–71. https://doi.org/10.1177/0894318409353793
FHS St. Gallen University of Applied Sciences. (2016). Nursing advice for women with vulvar neoplasias and surgical intervention: Evidence-based guideline [Article in German]. Retrieved from https://www.fhsg.ch/fhs.nsf/files/swissEBN_Leitlinie/$FILE/Leitlinie_IPW.pdf
Forman, D., de Martel, C., Lacey, C.J., Soerjomataram, I., Lortet-Tieulent, J., Bruni, L., . . . Franceschi, S. (2012). Global burden of human papillomavirus and related diseases. Vaccine, 30(Suppl. 5), F12–F23. https://doi.org/10.1016/j.vaccine.2012.07.055
Friedman, L.M., Furberg, C.D., & DeMets, D.L. (2010). Fundamentals of clinical trials (4th ed.). New York, NY: Springer.
Germino, B.B., Mishel, M.H., Crandell, J., Porter, L., Blyler, D., Jenerette, C., & Gil, K.M. (2013). Outcomes of an uncertainty management intervention in younger African American and Caucasian breast cancer survivors. Oncology Nursing Forum, 40, 82–92. https://doi.org/10.1188/13.ONF.82-92
Gil, K.M., Mishel, M.H., Belyea, M., Germino, B., Porter, L.S., & Clayton, M. (2006). Benefits of the uncertainty management intervention for African American and White older breast cancer survivors: 20-month outcomes. International Journal of Behavioral Medicine, 13, 286–294. https://doi.org/10.1207/s15327558ijbm1304_3
Gray, H.J. (2010). Advances in vulvar and vaginal cancer treatment. Gynecologic Oncology, 118, 3–5. https://doi.org/10.1016/j.ygyno.2010.04.011
Hagen, K.B., Aas, T., Lode, K., Gjerde, J., Lien, E., Kvaløy, J.T., . . . Lind, R. (2015). Illness uncertainty in breast cancer patients: Validation of the 5-item short form of the Mishel Uncertainty in Illness Scale. European Journal of Oncology Nursing, 19, 113–119. https://doi.org/10.1016/j.ejon.2014.10.009
Hall, D.L., Mishel, M.H., & Germino, B.B. (2014). Living with cancer-related uncertainty: Associations with fatigue, insomnia, and affect in younger breast cancer survivors. Supportive Care in Cancer, 22, 2489–2495. https://doi.org/10.1007/s00520-014-2243-y
HPV Information Centre. (2017). Human papillomavirus and related diseases report. Retrieved from http://www.hpvcentre.net/statistics/reports/XWX.pdf
Jefferies, H., & Clifford, C. (2011). Aloneness: The lived experience of women with cancer of the vulva. European Journal of Cancer Care, 20, 738–746. https://doi.org/10.1111/j.1365-2354.2011.01246.x
Jefferies, H., & Clifford, C. (2012). Invisibility: The lived experience of women with cancer of the vulva. Cancer Nursing, 35, 382–389. https://doi.org/10.1097/NCC.0b013e31823335a1
Juarez, G., Hurria, A., Uman, G., & Ferrell, B. (2013). Impact of a bilingual education intervention on the quality of life of Latina breast cancer survivors [Online exclusive]. Oncology Nursing Forum, 40, E50–60. https://doi.org/10.1188/13.ONF.E50-E60
Kaushik, S., Pepas, L., Nordin, A., Bryant, A., & Dickinson, H.O. (2014). Surgical interventions for high-grade vulval intraepithelial neoplasia. Cochrane Database of Systematic Reviews, 3, CD007928. https://doi.org/10.1002/14651858.CD007928.pub3
Kim, S.H., Lee, R., & Lee, K.S. (2012). Symptoms and uncertainty in breast cancer survivors in Korea: Differences by treatment trajectory. Journal of Clinical Nursing, 21, 1014–1023. https://doi.org/10.1111/j.1365-2702.2011.03896.x
Knobf, M.T., Cooley, M.E., Duffy, S., Doorenbos, A., Eaton, L., Given, B., . . . Mallory, G. (2015). The 2014–2018 Oncology Nursing Society research agenda. Oncology Nursing Forum, 42, 450–465. https://doi.org/10.1188/15.ONF.450-465
Kobleder, A., Mayer, H., & Senn, B. (2017). ‘Feeling someone is there for you’—Experiences of women with vulvar neoplasia with care delivered by an advanced practice nurse. Journal of Clinical Nursing, 26, 456–465. https://doi.org/10.1111/jocn.13434
Kobleder, A., Raphaelis, S., Glaus, A., Fliedner, M., Mueller, M.D., Gafner, D., . . . Senn, B. (2016). Recommendations for symptom management in women with vulvar neoplasms after surgical treatment: An evidence-based guideline. European Journal of Oncology Nursing, 25, 68–76. https://doi.org/10.1016/j.ejon.2016.10.003
Kosenko, K.A., Hurley, R.J., & Harvey, J.A. (2012). Sources of the uncertainty experienced by women with HPV. Qualitative Health Research, 22, 534–545. https://doi.org/10.1177/1049732311424404
Lai, J., Elleray, R., Nordin, A., Hirschowitz, L., Rous, B., Gildea, C., & Poole, J. (2014). Vulval cancer incidence, mortality and survival in England: Age-related trends. BJOG, 121, 728–739. https://doi.org/10.1111/1471-0528.12459
Lebel, S., Maheu, C., Lefebvre, M., Secord, S., Courbasson, C., Singh, M., . . . Catton, P. (2014). Addressing fear of cancer recurrence among women with cancer: A feasibility and preliminary outcome study. Journal of Cancer Survivorship, 8, 485–496. https://doi.org/10.1007/s11764-014-0357-3
Lenhard, W., & Lenhard, A. (2017). Calculation of the readability index LIX according to Björnson. Retrieved from https://www.psychometrica.de/lix.html
Liao, M.N., Chen, M.F., Chen, S.C., & Chen, P.L. (2008). Uncertainty and anxiety during the diagnostic period for women with suspected breast cancer. Cancer Nursing, 31, 274–283. https://doi.org/10.1097/01.NCC.0000305744.64452.fe
Likes, W.M., Russell, C., & Tillmanns, T. (2008). Women’s experiences with vulvar intraepithelial neoplasia. Journal of Obstetric, Gynecologic, and Neonatal Nursing, 37, 640–646. https://doi.org/10.1111/j.1552-6909.2008.00291.x
Lorig, K.R., & Holman, H. (2003). Self-management education: History, definition, outcomes, and mechanisms. Annals of Behavioral Medicine, 26, 1–7. https://doi.org/10.1207/S15324796ABM2601_01
Manci, N., Marchetti, C., Esposito, F., De Falco, C., Bellati, F., Giorgini, M., . . . Panici, P.B. (2009). Inguinofemoral lymphadenectomy: Randomized trial comparing inguinal skin access above or below the inguinal ligament. Annals of Surgical Oncology, 16, 721–728. https://doi.org/10.1245/s10434-008-0216-4
Mathiesen, O., Buus, S.K., & Cramers, M. (2007). Topical imiquimod can reverse vulvar intraepithelial neoplasia: A randomised, double-blinded study. Gynecologic Oncology, 107, 219–222. https://doi.org/10.1016/j.ygyno.2007.06.003
McCorkle, R., Dowd, M., Ercolano, E., Schulman-Green, D., Williams, A.L., Siefert, M.L., . . . Schwartz, P. (2009). Effects of a nursing intervention on quality of life outcomes in post-surgical women with gynecological cancers. Psycho-Oncology, 18, 62–70. https://doi.org/10.1002/pon.1365
McCorkle, R., Ercolano, E., Lazenby, M., Schulman-Green, D., Schilling, L.S., Lorig, K., & Wagner, E.H. (2011). Self-management: Enabling and empowering patients living with cancer as a chronic illness. CA: A Cancer Journal of Clinicians, 61, 50–62. https://doi.org/10.3322/caac.20093
Meltzer-Gunnes, C.J., Småstuen, M.C., Kristensen, G.B., Tropé, C.G., Lie, A.K., & Vistad, I. (2017). Vulvar carcinoma in Norway: A 50-year perspective on trends in incidence, treatment and survival. Gynecologic Oncology, 145, 543–548. https://doi.org/10.1016/j.ygyno.2017.03.008
Miller, W.R., & Rollnick, S. (2013). Motivational interviewing: Helping people change (3rd ed.). New York, NY: Guildford Press.
Mishel, M.H. (1981). The measurement of uncertainty in illness. Nursing Research, 30, 258–263. https://doi.org/10.1097/00006199-198109000-00002
Mishel, M.H. (1988). Uncertainty in illness. Image: The Journal of Nursing Scholarship, 20, 225–232. https://doi.org/10.1111/j.1547-5069.1988.tb00082.x
Mishel, M.H. (1990). Reconceptualization of the uncertainty in illness theory. Image: The Journal of Nursing Scholarship, 22, 256–262.
Mishel, M.H. (1997). Uncertainty in illness scale. Retrieved from https://nursing.unc.edu/files/2012/12/mishel_uncertainty_scales.pdf
Mishel, M.H., & Braden, C.J. (1988). Finding meaning: Antecedents of uncertainty in illness. Nursing Research, 37, 98–103.
Mishel, M.H., & Clayton, M. (2008). Theories of uncertainty in illness. In M.J. Smith & P.R. Liehr (Eds.), Middle range theory for nursing (2nd ed., pp. 55–84). New York, NY: Springer.
Naik, R., Nixon, S., Lopes, A., Godfrey, K., Hatem, M.H., & Monaghan, J.M. (2006). A randomized phase II trial of indole-3-carbinol in the treatment of vulvar intraepithelial neoplasia. International Journal of Gynecological Cancer, 16, 786–790. https://doi.org/10.1111/j.1525-1438.2006.00386.x
National Clinical Trials Network. (2017). NCI guidelines for auditing clinical trials for the NCI national clinical trials network (NCTN) program including NCI community oncology research program (NCORP) and NCORP research bases. Retrieved from http://ctep.cancer.gov/branches/ctmb/clinicalTrials/docs/ctmb_audit_gui…
Nooij, L.S., Brand, F.A., Gaarenstroom, K.N., Creutzberg, C.L., de Hullu, J.A., & van Poelgeest, M.I. (2016). Risk factors and treatment for recurrent vulvar squamous cell carcinoma. Critical Reviews in Oncology/Hematology, 106, 1–13. https://doi.org/10.1016/j.critrevonc.2016.07.007
Philp, S., Mellon, A., Barnett, C., D’Abrew, N., & White, K. (2017). The road less travelled: Australian women’s experiences with vulval cancer. European Journal of Cancer Care, 26. https://doi.org/10.1111/ecc.12465
Raphaelis, S., Mayer, H., Ott, S., Mueller, M.D., Steiner, E., Joura, E., & Senn, B. (2017). The impact of written information and counseling (WOMAN-PRO II Program) on symptom outcomes in women with vulvar neoplasia: A multicenter randomized controlled phase II study. Gynecologic Oncology, 146, 114–122. https://doi.org/10.1016/j.ygyno.2017.04.024
Sammarco, A., & Konecny, L.M. (2008). Quality of life, social support, and uncertainty among Latina breast cancer survivors. Oncology Nursing Forum, 35, 844–849. https://doi.org/10.1188/08.ONF.844-849
Schnürch, H.G., Ackermann, S., Alt, C.D., Barinoff, J., Böing, C., Dannecker, C., . . . Hampl, M. (2016). Diagnosis, therapy and follow-up care of vulvar cancer and its precursors. Guideline of the DGGG and DKG (S2k-level, AWMF registry number 015/059, November 2015). Geburtshilfe und Frauenheilkunde, 76, 1035–1049. https://doi.org/10.1055/s-0042-103728
Senn, B., Eicher, M., Mueller, M.D., Hornung, R., Fink, D., Baessler, K., . . . Engberg, S. (2013). A patient-reported outcome measure to identify occurrence and distress of post-surgery symptoms of WOMen with vulvAr Neoplasia (WOMAN-PRO)—A cross sectional study. Gynecologic Oncology, 129, 234–240. https://doi.org/10.1016/j.ygyno.2012.12.038
Senn, B., Gafner, D., Happ, M.B., Eicher, M., Mueller, M.D., Engberg, S., & Spirig, R. (2011). The unspoken disease: Symptom experience in women with vulval neoplasia and surgical treatment: A qualitative study. European Journal of Cancer Care, 20, 747–758. https://doi.org/10.1111/j.1365-2354.2011.01267.x
Shaha, M., Cox, C.L., Talman, K., & Kelly, D. (2008). Uncertainty in breast, prostate, and colorectal cancer: Implications for supportive care. Journal of Nursing Scholarship, 40, 60–67. https://doi.org/10.1111/j.1547-5069.2007.00207.x
Stiegelis, H.E., Hagedoorn, M., Sanderman, R., Bennenbroek, F.T., Buunk, B.P., van den Bergh, A.C., . . . Ranchor, A.V. (2004). The impact of an informational self-management intervention on the association between control and illness uncertainty before and psychological distress after radiotherapy. Psycho-Oncology, 13, 248–259. https://doi.org/10.1002/pon.738
Tristram, A., Hurt, C.N., Madden, T., Powell, N., Man, S., Hibbitts, S., . . . Griffiths, G. (2014). Activity, safety, and feasibility of cidofovir and imiquimod for treatment of vulval intraepithelial neoplasia (RT3VIN): A multicentre, open-label, randomised, phase 2 trial. Lancet Oncology, 15, 1361–1368. https://doi.org/10.1016/s1470-2045(14)70456-5
Wallner, M. (2015). Disease-related uncertainty in colorectal cancer patients undergoing chemotherapy treatment: A quantitative cross-sectional study (Master’s thesis). University of Vienna, Austria.
Zhang, Y., Kwekkeboom, K., & Petrini, M. (2015). Uncertainty, self-efficacy, and self-care behavior in patients with breast cancer undergoing chemotherapy in China. Cancer Nursing, 38, E19–E26. https://doi.org/10.1097/ncc.0000000000000165




