Symptom Clusters in Lymphoma Survivors Before, During, and After Chemotherapy: A Prospective Study
Objectives: To explore symptom clusters (SCs) in lymphoma survivors before, during, and after chemotherapy.
Sample & Setting: 61 lymphoma survivors from a medical center in central Taiwan were enrolled in the study.
Methods & Variables: A prospective observational study design was adopted. The MD Anderson Symptom Inventory was used to measure symptoms. The 13 symptoms assessed by the MD Anderson Symptom Inventory were evaluated after diagnosis and before chemotherapy (T1), after the fourth cycle of chemotherapy (T2), and after completion of chemotherapy (T3). Data were analyzed using mean, frequency, and latent profile analysis.
Results: Three SCs were identified at T1, four at T2, and three at T3. Fatigue was the predominant symptom in each SC for the participants over time. Fatigue, disturbed sleep, and numbness constituted an SC at T2 and T3. An SC consisting of multiple psychological symptoms was found only at T1.
Implications for Nursing: This study describes methods for grouping SCs. An SC of fatigue, disturbed sleep, and numbness was identified at T2 and T3. By familiarizing themselves with this SC, clinicians can be attentive to patients’ concurrent symptoms and implement early prevention measures and timely symptom management.
Jump to a section
Taiwan’s 2,967 new cases of and 1,275 deaths from lymphoma in 2019 indicate the increasing prevalence of the disease (Health Promotion Administration, 2022). Worldwide, 544,352 new cases of and 259,793 deaths from the disease were reported in 2020 (Sung et al., 2021). As the American Cancer Society (2022) observed, treatment for lymphoma differs from that for other types of solid cancer. Chemotherapy, the sole form of treatment, shows a greater than 70% overall five-year survival rate. Because lymphoma is not among the five most severe types of cancer in terms of incidence or mortality, relatively less attention has been paid to the clinical care of lymphoma survivors and research into the disease. However, the medical cost of lymphoma care is still considerable (National Health Insurance Administration, 2022). In addition, even when treatment eases the progression of the disease, patients with lymphoma often continue to experience short- and long-term physiologic and psychological symptoms (Troy et al., 2019).
The consensus in the literature is that cancer survivors experience an average of 10–15 symptoms during treatment (Lin et al., 2020). These overlapping symptoms may impede survivors’ physical functioning, interfere with the effects of treatment, or cut treatment short, in addition to reducing patients’ daily living function and quality of life (Ciavarella et al., 2017; Ji et al., 2017; Spichiger et al., 2011). However, most clinical cancer care emphasizes the management of individual symptoms, although the Oncology Nursing Society proposed that symptom clusters (SCs) be identified as a research priority in symptom science (Knobf et al., 2015; Von Ah et al., 2019).
Lenz et al. (1997) and Armstrong (2003) proposed the theory of unpleasant symptoms and the symptom experience model, respectively, as frameworks for assessing associations and synergy among symptoms. However, a lack of consistency in the conceptualization and definition of SCs has been noted (Barsevick, 2016).
Many oncology nursing scholars have focused on SCs as a priority of symptom science for more than a decade. Kim et al. (2005) and Dodd, Miaskowski, et al. (2001) used different statistical methods to arrive at definitions of an SC as involving two and three associated symptoms, respectively. The etiology of the symptoms associated with an SC may vary across patients (Dodd, Miaskowski, et al., 2001; Kim et al., 2005). Researchers have also sought to establish a scientific basis for interpreting the patterns of association, interactions, and synergy among the symptoms that comprise an SC to identify the best management for the multiple symptoms that cancer survivors experience (Dodd, Janson, et al., 2001).
The methodology differences in forming an SC and the lack of a precise mechanism to confirm an SC have limited SC research studies, resulting in a lack of effective interventions for SCs (Berger et al., 2013). There remains a need to explore new methodologies for identifying SCs and to figure out mechanisms of SCs for the development of SC-targeted interventions (Miaskowski et al., 2017).
The literature on SCs has tended to focus on cross-sectional studies of solid cancers, such as breast (Lee et al., 2020), gynecologic (Kim et al., 2018; Pozzar et al., 2021), and bladder cancers (Ren et al., 2017), or on lymphoma survivors (Bolukbas & Kutluturkan, 2014; Sezgin & Bektaş, 2020). To date, there has been only one longitudinal, prospective study of SCs among lymphoma survivors (Feng et al., 2017). Some studies have relied on the Memorial Symptom Assessment Scale (MSAS) as the research instrument and, for the method, factor correlation or cluster analysis to describe, enumerate, and provide a typology for SCs across the dimensions of occurrence and severity in lymphoma survivors who are receiving chemotherapy (Bolukbas & Kutluturkan, 2014; Feng et al., 2017; Sezgin & Bektaş, 2020). Because efforts to establish consistent analytical methods for the identification of SCs in lymphoma literature have been largely ineffective, no universal definition is currently in use, nor do standards exist regarding the appropriate number and level of correlation among the symptoms that comprise an SC (Barsevick, 2016). Indeed, SCs have seldom played a role in the care of cancer survivors in clinical settings (Bellury & Clark, 2018).
This study was conducted to help fill this gap in the literature. Latent profile analysis (LPA) served to identify SCs in lymphoma survivors and to compare and contrast their occurrence and severity over time. LPA and cluster analysis are person-centered analytical methods. The former is based on a finite mixture model, which uses group data. In terms of its accuracy in grouping patients based on the occurrence and severity of their symptoms, this approach is superior to cluster analysis using distance measures, and it is also superior to factor and correlation analysis, which are variable centered (Ciavarella et al., 2017; Lu et al., 2016). Knowledge of whether SCs vary across the dimensions of the symptom experience can inform efforts to improve symptom management as well as provide a scientific basis for the identification of SCs. Accordingly, this study explored SCs in lymphoma survivors before, during, and after chemotherapy.
Methods
Study Design and Sampling
This was a prospective, observational study. Convenience sampling was used to enroll participants who were aged 19 years or older, newly diagnosed with lymphoma, about to receive the R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) regimen, able to speak or read Mandarin, and willing to sign the written informed consent form required for participation in this study. Individuals who had been diagnosed with a secondary cancer or psychiatric disease were excluded.
Study Procedure
The institutional review board of China Medical University Hospital in Taichung, Taiwan, approved the study protocol (CMUH108-REC1-053). The research was conducted from June 2019 to November 2021 at a China Medical University Hospital. The study measurement occurred at the following three time points, ranging from before treatment to 10 weeks after treatment: after diagnosis and before chemotherapy (T1), after the fourth cycle of chemotherapy (T2), and after completion of chemotherapy (T3). The primary investigator and trained research nurses administered the initial questionnaire and performed the second and third assessments.
Measures
The questionnaire asked participants to supply their demographic information, including gender, age, body mass index, marital status, living status (i.e., living alone or with others), religious beliefs, education level, employment status, use of tobacco and alcohol, and exercise behavior. The clinical information gathered consisted of diagnosis, cancer stage, chemotherapy regimen, and cycles of chemotherapy received.
The MD Anderson Symptom Inventory (MDASI), which consists of 13 items of symptoms and 6 items of interference with daily living, was used to measure symptoms. Each item was assessed on an 11-point Likert-type scale ranging from 0 to 10. The scoring distinguished symptoms from interference with daily living, with total scores ranging from 0 to 130 for the former and 0 to 60 for the latter, and with higher scores indicating greater severity. The Cronbach’s alpha for the 13 symptoms measured in the study was 0.87, and for the symptom interference factors was about 0.9 (Wang et al., 2004).
Statistical Analyses
The data analysis was performed using IBM SPSS Statistics, version 21.0, and R software. Means, SDs, numbers, and percentages were used to describe the demographic and clinical information as well as the distribution of symptoms. LPA was used to explore the SCs at each time point. A combination of the lowest Bayesian information criterion (BIC) and the lowest parametric bootstrapped likelihood ratio test (Schmidt et al., 2021) served to determine which model best fit the data.
LPA was used to identify the SCs and predominant symptoms at each time point. After the LPA grouping of the SCs at each time point, the severity of each symptom was described based on the mean and SD, and the occurrence of symptoms based on the numbers and percentages of participants affected. Finally, the comparison of the five most severe and frequent symptoms in each SC was performed at each time point and over time to identify the predominant symptoms and SCs.
Results
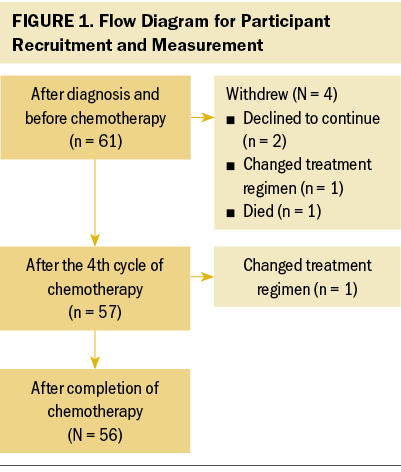
A total of 61 newly diagnosed lymphoma survivors participated in this study, of whom 56 completed all the measures (a retention rate of 91.8%). The attrition rates for the second and third measures were 6.56% (T1–T2) and 1.75% (T2–T3), respectively. Details of participant recruitment are presented in Figure 1.
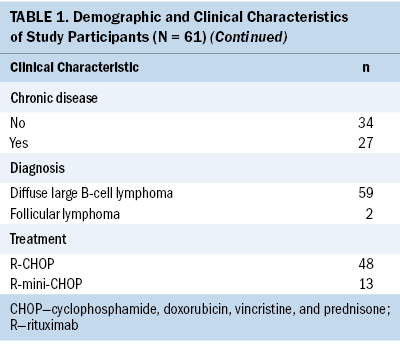
Demographic and Clinical Characteristics
Table 1 presents the characteristics of the participants in this study. All were lymphoma survivors. The average age of participants was 59.13 years, and the mean body mass index was 25.13 kg/m2. Most participants were male, married, lived with family members, held religious beliefs, had one or more college degrees, were not employed, did not use tobacco or alcohol, and exercised regularly. Regarding their clinical status, most had been diagnosed with stage III diffuse large B-cell lymphoma, had no chronic disease, had received R-CHOP and six cycles of chemotherapy, and had received the second rituximab infusion subcutaneously.

Predominant Symptoms and SCs at Each Time Point
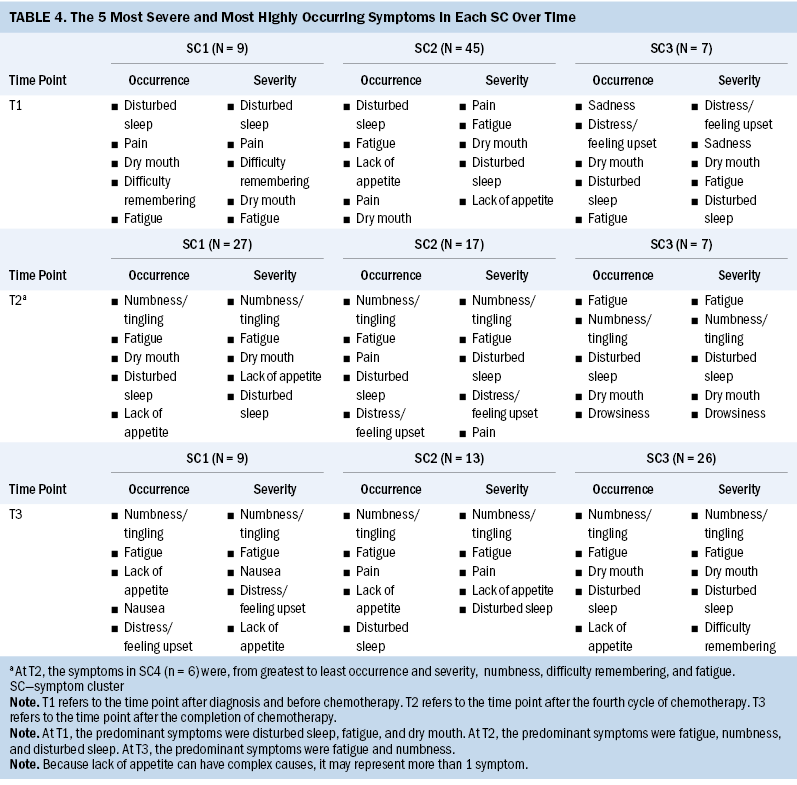
Based on the fit index for BIC and bootstrapped likelihood ratio test derived from the LPA results, three SCs (BIC = 3,109.82, p = 0.001) were identified at T1, four (BIC = 2,453.11, p = 0.008) at T2, and three (BIC = 2,177.86, p = 0.001) at T3. Tables 2 and 3 present the severity and occurrence of the symptoms in each SC over time.
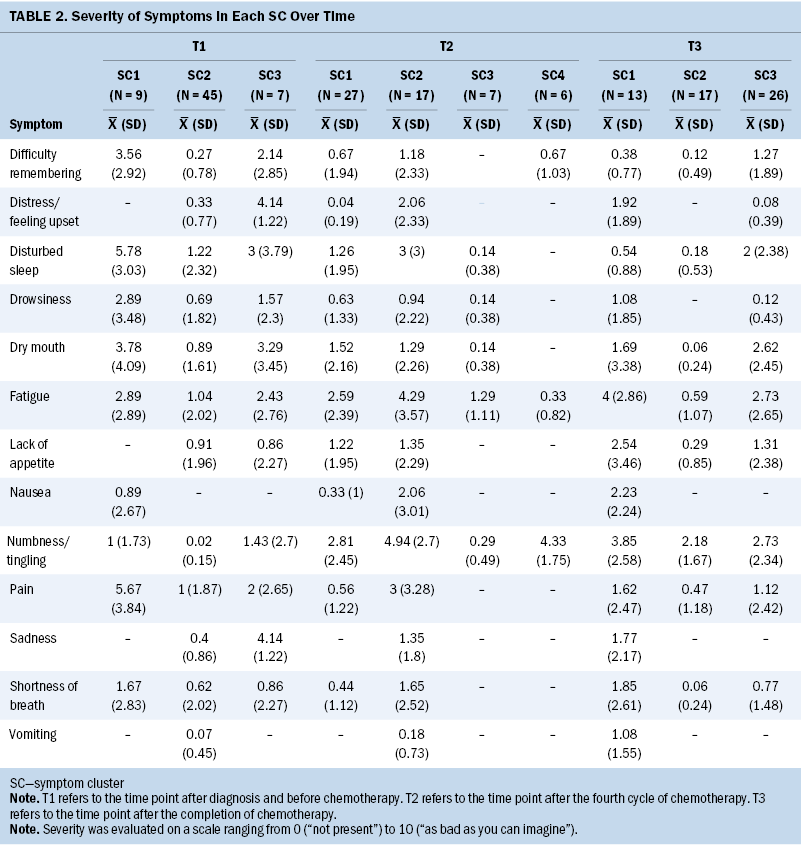
Table 4 presents a comparison of the predominant symptoms for each SC in terms of severity and occurrence over time. The predominant symptoms for SCs 1, 2, and 3 at T1 were disturbed sleep, fatigue, and dry mouth. SC3 at T1 was the only cluster consisting of more than one psychological symptom (i.e., sadness, distress, and feeling upset). The severity and occurrence of the five predominant symptoms and their priority in the three SCs were inconsistent at T1. In addition, fatigue and numbness/tingling were the predominant symptoms in each SC at T2 and T3, whereas disturbed sleep was also one of the predominant symptoms at T2.
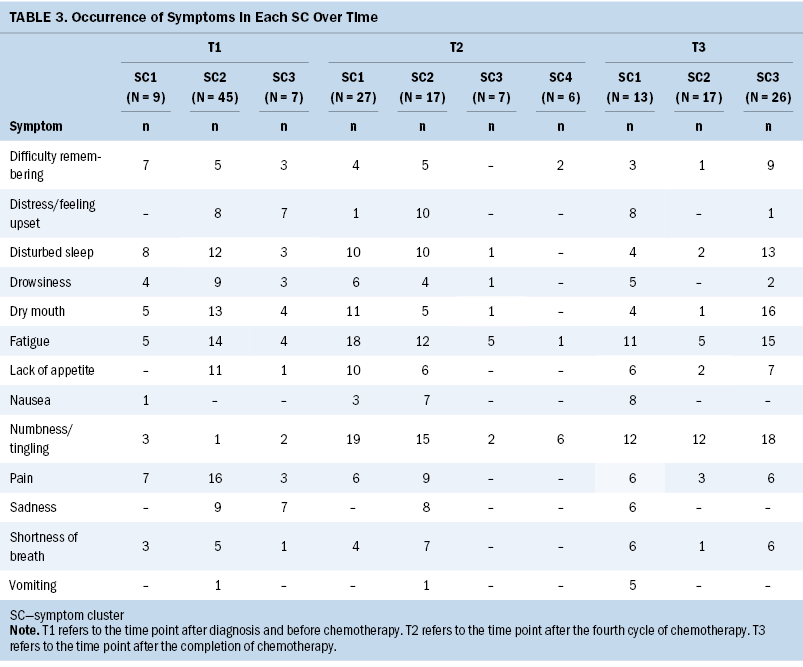
The predominant symptoms at T1 were disturbed sleep, fatigue, and dry mouth, with SCs including the following symptoms: (a) disturbed sleep, fatigue, dry mouth, pain, and lack of appetite (n = 45); (b) disturbed sleep, fatigue, dry mouth, pain, and difficulty remembering (n = 9); and (c) disturbed sleep, fatigue, dry mouth, distress/feeling upset, and sadness (n = 7). The predominant symptoms at T2 were numbness/tingling, fatigue, and disturbed sleep, with SCs including the following symptoms: (a) numbness/tingling, fatigue, disturbed sleep, dry mouth, and lack of appetite (n = 27); (b) numbness/tingling, fatigue, disturbed sleep, pain, and distress/feeling upset (n = 17); (c) numbness/tingling, fatigue, disturbed sleep, dry mouth, and drowsiness (n = 7); and (d) numbness/tingling, fatigue, and difficulty remembering (n = 6). The predominant SCs at T3 were numbness/tingling and fatigue, with SCs including the following symptoms: (a) numbness/tingling, fatigue, dry mouth, and disturbed sleep (n = 26); (b) numbness/tingling, fatigue, and pain (n = 17); and (c) numbness/tingling, fatigue, nausea, and distress/feeling upset (n = 13).
The symptoms experienced in SCs by most of the survivors at T1 consisted of disturbed sleep, fatigue, dry mouth, pain, and lack of appetite (n = 61); those experienced at T2 consisted of numbness/tingling, fatigue, disturbed sleep, dry mouth, and lack of appetite (n = 57); and those experienced at T3 consisted of numbness/tingling, fatigue, dry mouth, and disturbed sleep (n = 56). Fatigue was the predominant symptom for these participants before, during, and after receiving chemotherapy and, together with disturbed sleep and numbness/tingling, constituted an SC during and after treatment.
Discussion
Three SCs were identified among the lymphoma survivors who participated in this study after diagnosis and before chemotherapy (T1), four SCs after the fourth cycle of chemotherapy (T2), and three SCs after completion of chemotherapy (T3). The predominant symptoms in the three SCs were disturbed sleep, fatigue, and dry mouth at T1; numbness/tingling, fatigue, and disturbed sleep at T2; and numbness/tingling and fatigue at T3. Fatigue was the predominant symptom for the participants before, during, and after chemotherapy, and fatigue, disturbed sleep, and numbness/tingling formed an SC during and after chemotherapy.
Severity, Occurrence, and Predominant Symptoms at Each Time Point
This study found that the predominant symptoms across the three SCs at T1 were disturbed sleep, fatigue, and dry mouth. Most participants experienced disturbed sleep, fatigue, dry mouth, pain, and lack of appetite SC (n = 45). Only one other study, by Feng et al. (2017), has used a prospective design to identify SCs in lymphoma survivors; they reported (a) a psychology-related SC including sadness, worry, nervousness, difficulty sleeping, and feelings of irritability and (b) a disease-related SC including pain and swelling of the arms or legs, with both SCs occurring before chemotherapy. Pain and disturbed sleep were the only symptoms identified in the study by Feng et al. (2017) and the current study; however, Feng et al. (2017) provided no information regarding the occurrence and severity of these symptoms but simply assigned them to different SCs. The current study did identify a psychological SC including distress, feeling upset, and sadness at T1 that resembles the psychology-related SC identified by Feng et al. (2017), though with a somewhat different set of symptoms.
In the current study, the predominant symptoms in the SCs at T2 were numbness/tingling, fatigue, and disturbed sleep. Most participants (n = 27) experienced an SC consisting of these symptoms, along with dry mouth and lack of appetite, at this time point. This is in contrast to the results that Feng et al. (2017) reported, with the only similar symptoms being numbness/tingling and disturbed sleep. In addition, although Bolukbas and Kutluturkan (2014) identified seven SCs during treatment, the present study identified only four. The participants in the current study and in Bolukbas and Kutluturkan’s (2014) study alike reported numbness/tingling, fatigue, pain, dry mouth, and lack of appetite but were assigned to different SCs. Sezgin and Bektaş (2020) identified nine SCs during treatment, categorizing them into groups of three based on the occurrence, severity, and distress associated with the symptoms. They assigned gastrointestinal symptoms (i.e., nausea, vomiting, and dry mouth) to SCs based on the occurrence and severity of the symptoms. Dry mouth was the only symptom identified in both studies during treatment. The present study used LPA and likewise considered the severity and occurrence of the symptoms to identify SCs; this approach seems to be more consistent with the definition of an SC by Dodd, Miaskowski, et al. (2001) as a grouping of three concurrent and correlated symptoms.
The predominant symptoms in the SCs at T3 were numbness/tingling and fatigue. Most participants experienced an SC consisting of numbness/tingling, fatigue, dry mouth, and disturbed sleep (n = 26), which, again, bears resemblance to the SCs reported by Feng et al. (2017). However, Feng et al.’s (2017) designations of psychology-related and chemotherapy-related SCs identified during the sixth cycle of chemotherapy differed from those in the present study. Numbness is one of the chemotherapy-induced peripheral neuropathy symptoms that have been commonly reported in survivors during and after treatment over the past decade, and it is a bothersome late effect in the population with cancer (Seretny et al., 2014). Eikeland et al. (2021) found that long-term Hodgkin lymphoma survivors had a higher prevalence of peripheral neuropathy than the general Dutch population. Of note, de Rooij et al. (2021) conducted a secondary data analysis using a network comparison test to identify SCs in heterogeneous samples consisting of seven types of cancer, including lymphoma, and found fatigue to be a core symptom across types of cancer for short-term survivors (less than five years) and long-term (greater than five years) survivors.
In the present study, fatigue was the one symptom that was predominant among the survivors before, during, and after receiving R-CHOP. This result echoes those of previous studies conducted to identify SCs in other types of cancer (Beck et al., 2005; de Rooij et al., 2021; Dodd, Miaskowski, et al., 2001; Mathew et al., 2021). Indeed, fatigue remains the most common symptom from before diagnosis to end of life and may be a result of medical treatment (Fabi et al., 2020). In addition, in the present study, numbness/tingling was the predominant symptom during and after treatment. Numbness has been associated with certain chemotherapy agents, such as doxorubicin and vincristine, that are used to treat lymphoma (Saito et al., 2019). The typical symptoms of lymphoma that have been identified in past studies included lymphadenopathy, pain, fever, sweats, and fatigue (Storck et al., 2019), of which only pain and fatigue were identified in the present study. The SC identified in the current study consisting of fatigue, disturbed sleep, and numbness/tingling may be associated with lymphoma survivors during and after R-CHOP chemotherapy. The findings of fatigue and disturbed sleep in this study were echoed by the previous SC review articles (Berger et al., 2013; Miaskowski et al., 2017). The current study is superior in its use of LPA to identify two distinct symptom subgroups, in addition to comparing and contrasting the key dimensions of SC in terms of predominant symptoms, as well as the severity and prevalence of symptoms at different time points, as has been recommended in the SC literature (Armstrong et al., 2004).
To date, only three studies similar to the present study of SCs in individuals with lymphoma have been conducted. The number of SCs identified in earlier studies ranged from 2 to 7, and the number of symptoms in the SCs ranged from 2 to 15. The prospective study by Feng et al. (2017) used the MSAS to collect data before chemotherapy and after the first, third, and fourth cycles as well as factor analysis to group SCs in Chinese individuals with cancer. Bolukbas and Kutluturkan (2014) also used the MSAS—in this case, to collect data from 110 lymphoma survivors undergoing chemotherapy. They identified seven SCs through correlation analysis. In their cross-sectional study using the MSAS, Sezgin and Bektaş (2020) collected data from 109 lymphoma survivors undergoing chemotherapy and performed a hierarchical cluster analysis that accounted for the frequency, severity, and distress associated with symptoms to identify SCs. The MSAS, as originally conceived, included 32 symptoms, but it was not designed specifically for individuals with lymphoma (Chang et al., 2000; Portenoy et al., 1994). The present study used the MDASI, which assesses 13 symptoms, the recommended number for cancer populations, and is commonly used in a variety of cancer research and clinical settings (Lin et al., 2007). The MDASI has several advantages over other tools, particularly its ability to assess a concise but adequate set of symptoms that are applicable to most individuals with cancer and its ready intelligibility for patients irrespective of education (Wang et al., 2004).
The statistical extraction of SCs is another key area in need of further investigation to determine whether statistically derived SCs constitute real clusters (Mathew et al., 2021). Previous statistical analyses of SCs have commonly used variables-centered methods such as factor or correlation analysis, leading to the grouping of many more symptoms in an SC. However, the somewhat limited scholarly discussion of the occurrence and severity of symptoms has made it difficult to identify the predominant symptoms. In a systematic review of the use of latent models, Mathew et al. (2021) helped to identify the optimal analytical method for identifying clinically relevant SCs. The present study involved the use of person-centered LPA and accounted for the occurrence and severity of symptoms in specific SCs over time. LPA is a precise approach that involves identifying symptoms that individuals experience as an SC (Lu et al., 2016). This study is innovative in proposing criteria and methods for grouping symptoms into SCs. However, more studies of this sort are needed to confirm the current results in lymphoma survivors and validate the methodology for other types of cancer.
Limitations
The generalizability of this study is limited because of the homogeneity of the sample and the small sample size. This limitation is a result of the restriction of participation to individuals who had received R-CHOP treatment, the most common regimen for lymphoma survivors. In addition, the participants were recruited from only one medical center, located in central Taiwan. The relatively smaller sample size is appropriate for the study’s prospective, repeated measures design; still, the authors recommend further studies to increase the number of participants to enhance the power of the study.
Implications for Nursing
The findings presented in this article identified a pattern of symptoms and changes in SCs dependent on whether the individuals were about to receive, were receiving, or had completed chemotherapy for lymphoma. In light of the SCs described in this article, clinicians should be on the alert for concurrent symptoms and also make efforts to provide early prevention and in-time management of the SC consisting of fatigue, disturbed sleep, and numbness/tingling in lymphoma survivors receiving R-CHOP chemotherapy. In addition, the authors recommend using the MDASI in SC research and for periodic assessments in clinical settings because it is rapid, easy to follow, and reliable.

Conclusions
In this study, fatigue, disturbed sleep, and numbness/tingling were found to form the predominant SC in individuals with lymphoma who were receiving R-CHOP chemotherapy. This research was also innovative in using LPA to group SCs at various time points and in comparing the occurrence and severity of symptoms over time to identify the SCs experienced by lymphoma survivors before, during, and after chemotherapy. To verify the results and validate the rules and methods proposed in this study, further studies are needed involving larger numbers of lymphoma survivors and survivors of other types of cancer.
The authors gratefully acknowledge all participants for their contributions to this study.
About the Authors
Chih-Jung Wu, RN, PhD, is an assistant professor at China Medical University in Taichung City; Li-Yuan Bai, MD, PhD, is a professor in the Department of Hematology and Oncology at China Medical University Hospital in Taichung; Yu-Chi Chen, RN, PhD, is a professor in the Department of Nursing at National Yang Ming Chiao Tung University in Taipei; Ching-Feng Wu, MD, is a physician in the Department of Cardiovascular Surgery at China Medical University Hospital, Kuan-Chia Lin, PhD, is a professor in the Institute of Hospital and Health Care Administration at National Yang Ming Chiao Tung University; and Ya-Jung Wang, RN, PhD, is an associate professor in the Department of Nursing at DaYeh University in Dacun, all in Taiwan. No financial relationships to disclose. C.-J. Wu, Bai, and Wang contributed to the conceptualization and design. C.-J. Wu, C.-F. Wu, and Wang completed the data collection. C.-J. Wu, C.-F. Wu, Lin, and Wang provided statistical support. C.-J. Wu, Bai, Chen, and Wang provided the analysis. C.-J. Wu, Chen, C.-F. Wu, and Wang contributed to the manuscript preparation. Wang can be reached at wangyj@mail.dyu.edu.tw, with copy to ONFEditor@ons.org. (Submitted June 2022. Accepted November 6, 2022.)
References
American Cancer Society. (2022). Survival rates and factors that affect prognosis (outlook) for non-Hodgkin lymphoma. https://www.cancer.org/cancer/non-hodgkin-lymphoma/detection-diagnosis-…
Armstrong, T.S. (2003). Symptoms experience: A concept analysis. Oncology Nursing Forum, 30(4), 601–606. https://doi.org/10.1188/03.ONF.601-606
Armstrong, T.S., Cohen, M.Z., Eriksen, L.R., & Hickey, J.V. (2004). Symptom clusters in oncology patients and implications for symptom research in people with primary brain tumors. Journal of Nursing Scholarship, 36(3), 197–206. https://doi.org/10.1111/j.1547-5069.2004.04038.x
Barsevick, A. (2016). Defining the symptom cluster: How far have we come? Seminars in Oncology Nursing, 32(4), 334–350. https://doi.org/10.1016/j.soncn.2016.08.001
Beck, S.L., Dudley, W.N., & Barsevick, A. (2005). Pain, sleep disturbance, and fatigue in patients with cancer: Using a mediation model to test a symptom cluster. Oncology Nursing Forum, 32(3), E48–E55. https://doi.org/10.1188/04.ONF.E48-E55
Bellury, L.M., & Clark, J.C. (2018). Searching for simplicity in the complexity of symptom clusters. Oncology Nursing Forum, 45(3), 284–286. https://doi.org/10.1188/18.ONF.284-286
Berger, A.M., Yennu, S., & Million, R. (2013). Update on interventions focused on symptom clusters: What has been tried and what have we learned? Current Opinion in Supportive and Palliative Care, 7(1), 60–66.
Bolukbas, F., & Kutluturkan, S. (2014). Symptoms and symptom clusters in non Hodgkin’s lymphoma patients in Turkey. Asian Pacific Journal of Cancer Prevention, 15(17), 7153–7158. https://doi.org/10.7314/apjcp.2014.15.17.7153
Chang, V.T., Hwang, S.S., Feuerman, M., Kasimis, B.S., & Thaler, H.T. (2000). The Memorial Symptom Assessment Scale Short Form (MSAS-SF). Cancer, 89(5), 1162–1171.
Ciavarella, S., Minoia, C., Quinto, A.M., Oliva, S., Carbonara, S., Cormio, C., . . . Guarini, A. (2017). Improving provision of care for long-term survivors of lymphoma. Clinical Lymphoma, Myeloma and Leukemia, 17(12), e1–e9.
de Rooij, B.H., Oerlemans, S., van Deun, K., Mols, F., de Ligt, K.M., Husson, O., . . . Schoormans, D. (2021). Symptom clusters in 1,330 survivors of 7 cancer types from the PROFILES registry: A network analysis. Cancer, 127(24), 4665–4674. https://doi.org/10.1002/cncr.33852
Dodd, M., Janson, S., Facione, N., Faucett, J., Froelicher, E.S., Humphreys, J., . . . Taylor, D. (2001). Advancing the science of symptom management. Journal of Advanced Nursing, 33(5), 668–676. https://doi.org/10.1046/j.1365-2648.2001.01697.x
Dodd, M.J., Miaskowski, C., & Paul, S.M. (2001). Symptom clusters and their effect on the functional status of patients with cancer. Oncology Nursing Forum, 28(3), 465–470.
Eikeland, S.A., Smeland, K.B., Mols, F., Fagerli, U.-M., Bersvendsen, H.S., Kiserud, C.E., & Fosså, A. (2021). Chemotherapy-induced peripheral neuropathy after modern treatment of Hodgkin’s lymphoma: Symptom burden and quality of life. Acta Oncologica, 60(7), 911–920. //https://doi.org/10.1080/0284186X.2021.1917776
Fabi, A., Bhargava, R., Fatigoni, S., Guglielmo, M., Horneber, M., Roila, F., . . . Ripamonti, C.I. (2020). Cancer-related fatigue: ESMO clinical practice guidelines for diagnosis and treatment. Annals of Oncology, 31(6), 713–723. https://doi.org/10.1016/j.annonc.2020.02.016
Feng, L., Feng, L., & Meng, L. (2017). Study on symptom clusters of young and middle-aged patients with lymphoma undergoing chemotherapy. Chinese Journal of Nursing, 52(12), 1459–1463. https://doi.org/10.3761/j.issn.0254-1769.2017.12.010
Health Promotion Administration. (2022, January). Cancer registry annual report, 2019, Taiwan. Ministry of Health and Welfare. https://www.hpa.gov.tw/File/Attach/14913/File_18302.pdf
Ji, Y.-B., Bo, C.-L., Xue, X.-J., Weng, E.-M., Gao, G.-C., Dai, B.-B., . . . Xu, C.-P. (2017). Association of inflammatory cytokines with the symptom cluster of pain, fatigue, depression, and sleep disturbance in Chinese patients with cancer. Journal of Pain and Symptom Management, 54(6), 843–852.
Kim, H.-J., McGuire, D.B., Tulman, L., & Barsevick, A.M. (2005). Symptom clusters: Concept analysis and clinical implications for cancer nursing. Cancer Nursing, 28(4), 270–282. https://doi.org/10.1097/00002820-200507000-00005
Kim, M., Kim, K., Lim, C., & Kim, J.-S. (2018). Symptom clusters and quality of life according to the survivorship stage in ovarian cancer survivors. Western Journal of Nursing Research, 40(9), 1278–1300. https://doi.org/10.1177/0193945917701688
Knobf, M.T., Cooley, M.E., Duffy, S., Doorenbos, A., Eaton, L., Given, B., . . . Mallory, G. (2015). The 2014–2018 Oncology Nursing Society Research Agenda. Oncology Nursing Forum, 42(5), 450–465. https://doi.org/10.1188/15.ONF.450-465
Lee, L., Ross, A., Griffith, K., Jensen, R.E., & Wallen, G.R. (2020). Symptom clusters in breast cancer survivors: A latent class profile analysis. Oncology Nursing Forum, 47(1), 89–100. https://doi.org/10.1188/20.ONF.89-100
Lenz, E.R., Pugh, L.C., Milligan, R.A., Gift, A., & Suppe, F. (1997). The middle-range theory of unpleasant symptoms: An update. Advances in Nursing Science, 19(3), 14–27. https://doi.org/10.1097/00012272-199703000-00003
Lin, C.-C., Chang, A.-P., Cleeland, C.S., Mendoza, T.R., & Wang, X.S. (2007). Taiwanese version of the M.D. Anderson Symptom Inventory: Symptom assessment in cancer patients. Journal of Pain and Symptom Management, 33(2), 180–188. https://doi.org/10.1016/j.jpainsymman.2006.07.018
Lin, Y., Docherty, S.L., Porter, L.S., & Bailey, D.E., Jr. (2020). Symptom experience and self-management for multiple co-occurring symptoms in patients with gastric cancer: A qualitative study. European Journal of Oncology Nursing, 49, 101860. https://doi.org/10.1016/j.ejon.2020.101860
Lu, M.-S., Chen, Q.-Z., He, J.-R., Wei, X.-L., Lu, J.-H., Li, S.-H., . . . Qiu, X. (2016). Maternal dietary patterns and fetal growth: A large prospective cohort study in China. Nutrients, 8(5), 257. https://doi.org/10.3390/nu8050257
Mathew, A., Tirkey, A.J., Li, H., Steffen, A., Lockwood, M.B., Patil, C.L., & Doorenbos, A.Z. (2021). Symptom clusters in head and neck cancer: A systematic review and conceptual model. Seminars in Oncology Nursing, 37(5), 151215. https://doi.org/10.1016/j.soncn.2021.151215
Miaskowski, C., Barsevick, A., Berger, A., Casagrande, R., Grady, P.A., Jacobsen, P., . . . Marden, S. (2017). Advancing symptom science through symptom cluster research: Expert panel proceedings and recommendations. Journal of the National Cancer Institute, 109(4), djw253. https://doi.org/10.1093/jnci/djw253
National Health Insurance Administration. (2022). Cancer cost ranking. Ministry of Health and Welfare. https://www.nhi.gov.tw/Content_List.aspx?n=AE8F3C1B6EC35217&topn=CDA985…
Portenoy, R.K., Thaler, H.T., Kornblith, A.B., Lepore, J.M., Friedlander-Klar, H., Kiyasu, E., . . . Scher, H. (1994). The Memorial Symptom Assessment Scale: An instrument for the evaluation of symptom prevalence, characteristics and distress. European Journal of Cancer, 30A(9), 1326–1336. https://doi.org/10.1016/0959-8049(94)90182-1
Pozzar, R.A., Hammer, M.J., Cooper, B.A., Kober, K.M., Chen, L.-M., Paul, S.M., . . . Miaskowski, C. (2021). Symptom clusters in patients with gynecologic cancer receiving chemotherapy. Oncology Nursing Forum, 48(4), 441–452. https://doi.org/10.1188/21.ONF.441-452
Ren, H., Tang, P., Zhao, Q., & Ren, G. (2017). Symptom clusters and related factors in bladder cancer patients three months after radical cystectomy. BMC Urology, 17(1), 65. https://doi.org/10.1186/s12894-017-0255-x
Saito, T., Okamura, A., Inoue, J., Makiura, D., Doi, H., Yakushijin, K., . . . Ono, R. (2019). Anemia is a novel predictive factor for the onset of severe chemotherapy-induced peripheral neuropathy in lymphoma patients receiving rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisolone therapy. Oncology Research, 27(4), 469–474. https://doi.org/10.3727/096504018X15267574931782
Schmidt, M.N., Seddig, D., Davidov, E., Mørup, M., Jon Albers, K., Bauer, J.M., & Kano Glückstad, F. (2021). Latent profile analysis of human values: What is the optimal number of clusters? Methodology, 17(2), 127–148.
Seretny, M., Currie, G.L., Sena, E.S., Ramnarine, S., Grant, R., MacLeod, M.R., . . . Fallon, M. (2014). Incidence, prevalence, and predictors of chemotherapy-induced peripheral neuropathy: A systematic review and meta-analysis. Pain, 155(12), 2461–2470. https://doi.org/10.1016/j.pain.2014.09.020
Sezgin, M.G., & Bektaş, H. (2020). Symptom clustering and its effect on functional status in lymphoma patients. Florence Nightingale Journal of Nursing, 28(2), 143–154. https://doi.org/10.5152/FNJN.2020.19107
Spichiger, E., Müller-Fröhlich, C., Denhaerynck, K., Stoll, H., Hantikainen, V., & Dodd, M. (2011). Prevalence of symptoms, with a focus on fatigue, and changes of symptoms over three months in outpatients receiving cancer chemotherapy. Swiss Medical Weekly, 141, w13303.
Storck, K., Brandstetter, M., Keller, U., & Knopf, A. (2019). Clinical presentation and characteristics of lymphoma in the head and neck region. Head and Face Medicine, 15(1), 1. https://doi.org/10.1186/s13005-018-0186-0
Sung, H., Ferlay, J., Siegel, R.L., Laversanne, M., Soerjomataram, I., Jemal, A., & Bray, F. (2021). Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Clinicians, 71(3), 209–249. https://doi.org/10.3322/caac.21660
Troy, J.D., Locke, S.C., Samsa, G.P., Feliciano, J., Richhariya, A., & LeBlanc, T.W. (2019). Patient-reported distress in Hodgkin lymphoma across the survivorship continuum. Supportive Care in Cancer, 27(7), 2453–2462.
Von Ah, D., Brown, C.G., Brown, S.J., Bryant, A.L., Davies, M., Dodd, M., . . . Cooley, M.E. (2019). Research Agenda of the Oncology Nursing Society: 2019–2022. Oncology Nursing Forum, 46(6), 654–669. https://doi.org/10.1188/19.ONF.654-669
Wang, X.S., Wang, Y., Guo, H., Mendoza, T.R., Hao, X.-S., & Cleeland, C.S. (2004). Chinese version of the M.D. Anderson Symptom Inventory: Validation and application of symptom measurement in cancer patients. Cancer, 101(8), 1890–1901. https://doi.org/10.1002/cncr.20448


