Lymphoma Therapy and Adverse Events: Nursing Strategies for Thinking Critically and Acting Decisively
Background: Multiple treatment options, combined with disease heterogeneity, have created nursing challenges in the management of adverse events (AEs) during antilymphoma therapy. Testing has revealed that less than half of participating nurses correctly graded peripheral neuropathy and neutropenia related to antilymphoma regimens.
Objectives: This article identifies nursing challenges in the management of AEs associated with therapy for lymphomas and describes how strategies in critical thinking can help meet those challenges.
Methods: A comprehensive literature search in oncology nursing, nursing education, and critical thinking was conducted; participant responses to pre- and post-tests at nursing education programs were evaluated; and a roundtable meeting of authors was convened.
Findings: Oncology nurses can cultivate critical thinking skills, practice thinking critically in relation to team members and patients, leverage information from the Patient-Reported Outcomes Common Terminology Criteria for Adverse Events, and manage workflow to allow more opportunity for critical thinking.
Jump to a section
Disclosures: Wagner-Johnston has received a speakers’ fee from Gilead Sciences and has previously consulted for Gilead Sciences and Pharmacyclics. Delibovi received a consultation fee for her work on this article. Development of this supplement was supported by an educational grant from Celgene Corporation. (Disclosures in the printed article appear on page 12.)
Therapy for lymphomas has changed markedly in recent years (National Comprehensive Cancer Network [NCCN], 2016c, 2016d). Treatment of B-cell lymphomas has been enhanced by rituximab (Rituxan®) immunotherapy; novel targeted therapies, such as bortezomib (Velcade®), lenalidomide (Revlimid®), and idelalisib (Zydelig®); bendamustine (Treanda®) chemotherapy; and other innovations (Coiffier et al., 2002; Fowler et al., 2011, 2014; NCCN, 2016d; Rummel et al., 2013). Patients with T-cell lymphomas may now receive histone deacetylase inhibitors and other targeted agents (Duvic et al., 2009). For patients with Hodgkin lymphoma (HL), although chemotherapy remains the standard of care, targeted therapies and immunotherapies may also be indicated as second-line treatment (Ansell et al., 2013; Johnston et al., 2010; Moskowitz et al., 2015).
A growing number of treatment options, combined with the heterogeneity of HL and non-Hodgkin lymphoma (NHL), have created nursing challenges in the management of adverse events (AEs). Nurses are the most trusted healthcare team members (Newport, 2012). This puts the oncology nurse in a strong position to assess toxicities with the highest level of patient input, to gather information about overall distress related to the toxicities, and to learn how the toxicities are impacting the patient’s routine activities and daily quality of life. Nurses are called on to identify a host of AEs in a wide variety of regimens, and differentiate these AEs from complex lymphoma symptom patterns. Nurses must then grade each AE accurately, and determine which of the multiple anti-lymphoma drugs in the regimen may have caused the AE. Finally, nurses must collaborate with the interdisciplinary care team to recommend whether to adjust therapy based on the grade and description of the AE, distress level, and impact on quality of life. The recommendation from nurses is essential to help the patient and clinical team come to a shared decision on therapy adjustment.
These activities require nurses to think critically and act decisively by conducting a logical and systematic analysis to determine AE grade, deciding on a recommendation for intervention, and communicating persuasively to the interdisciplinary team, the patient, and the caregiver (Brenner, Hughes, & Sutphen, 2008). This skill set is not limited to lymphoma; it applies across all types of malignancies. Lymphoma treatment, because it encompasses so many regimens and virtually all types of AEs, provides an optimal model for AE management across cancer care. The problems and solutions presented in this article are applicable to lymphomas but certainly extend beyond lymphoma to other cancers (see Appendix A). 
Nurses can access a number of valuable resources to enhance their knowledge and competency regarding the assessment and management of AEs. Among these are guidelines from the NCCN (2016a, 2016b) on supportive care and the Oncology Nursing Society’s ([ONS’s], 2016) Putting Evidence Into Practice resources. An overview of all lymphoma treatment toxicities is beyond the scope of this article but can be found in McFadden, Poniatowski, and Temple’s (2006) Contemporary Issues in Lymphoma: A Nursing Perspective and, for more recently introduced regimens, in NCCN (2016c, 2016d) guidelines for NHL and HL.
Challenges
Lymphoma treatment options are so vast that virtually every possible AE may occur. For example, hematologic toxicities that include anemia, neutropenia, and thrombocytopenia are common to cytotoxic drugs and molecularly targeted agents. IV doxorubicin (Adriamycin®), cyclophosphamide (Cytoxan®), oral lenalidomide, and numerous other drugs may produce grade 3–4 neutropenia (Baxter Healthcare, 2010; Celgene Corporation, 2015; Pfizer Laboratories, 2015) Peripheral neuropathy may occur with drugs as diverse as bortezomib (Velcade®), vincristine (Oncovin®), carboplatin (Paraplatin®), brentuximab vedotin (Adcetris®), alemtuzumab (Lemtrada®), and many more (Genzyme Corporation, 2014; Grisold, Cavaletti, & Windebank, 2012; Millennium Pharmaceuticals, Inc., 2015; Seattle Genetics, 2015). Serious mucocutaneous reactions are associated with a range of drugs—from the antibody rituximab to cytotoxic therapy like bendamustine (Genentech, 2014; Teva Pharmaceuticals, 2015). Fatigue and gastrointestinal (GI) effects are ubiquitous reactions to anticancer therapy (NCCN, 2016a, 2016b), and some injectable drugs, notably antibodies, carry high risk for hypersensitivity infusion reactions (Genentech, 2014; Genzyme Corporation, 2014). Older chemotherapy drugs, which are still heavily relied upon in current regimens, are characterized by potentially severe and dose-limiting GI, mucosal, and hematologic AEs. 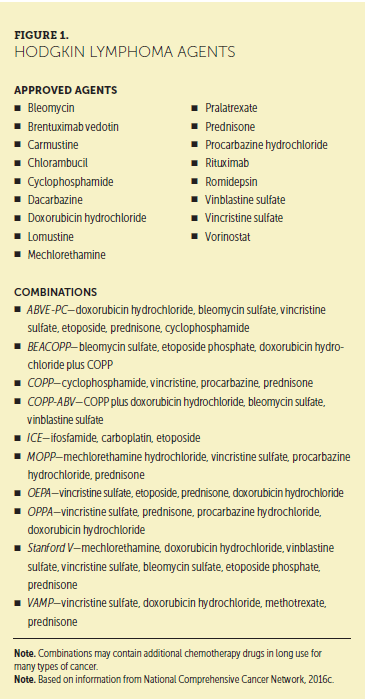
Because antilymphoma therapy often relies on drug combinations (see Figures 1 and 2), additive or synergistic toxicity of drugs in the regimen may occur. The introduction of novel agents into combinations may produce new interactions or toxicities that will not be fully understood without more clinical experience. The potential exacerbation of toxicities in combination therapy complicates the already intricate set of AEs in antilymphoma therapy. In addition, various lymphomas have disease-related symptom patterns that mimic treatment-related symptoms. For example, pruritus and fatigue may occur in HL and B-cell NHLs, and anemia in diffuse large B-cell lymphoma (NCCN, 2016c, 2016d).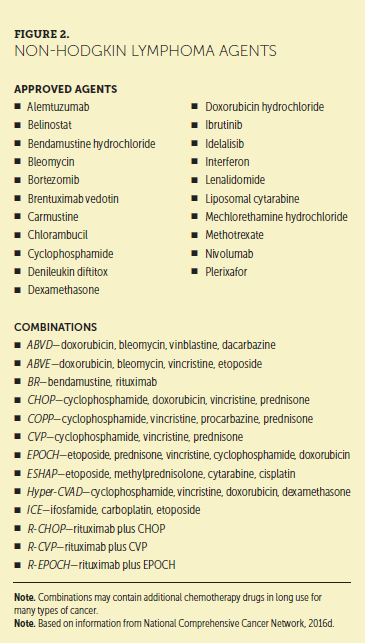
In this complex setting, inaccurate grading of AEs, uncertainty or errors in decisions to adjust therapy, and ineffective team communication represent significant hurdles to effective care (Cirillo et al., 2009; Schulmeister, 2006). Unfavorable outcomes can result, including risks to patient safety (e.g., toxicities graded inaccurately low) and less-optimal treatment efficacy (e.g., drugs are withheld because of inaccurate high grades on AEs or miscommunication of grade). These hurdles contribute to conditions that impede critical thinking in nursing practice.
Grading
During lymphoma treatment, nurses confront the documented challenge of AE grading during assessment and care (Cirillo et al., 2009; Schulmeister, 2006; Trotti, Colevas, Setser, & Basch, 2007). Grading inaccuracy can lead to clinical deficits, including underestimation of toxicity and the potential for avoidable risk, as well as overestimation of toxicity, leading to unnecessary dose reduction or therapy termination. Grading of many AEs include impact on quality of life, so that the nurse, by making this quality-of-life assessment, is among the most strongly positioned healthcare team member to evaluate and report this critical consideration.
Participant responses during 10 nursing education programs, conducted at ONS Chapters, revealed gaps in grading knowledge among nurses (Rogers, 2015). An author of the current article (AG) served as a faculty member for these programs. Program participants were presented with six cases and were informed that the cases pertained to peripheral neuropathy, neutropenia, or infusion reactions. They were asked to identify the symptom or AE, grade it, and, if appropriate, provide treatment options for that toxicity and grade. Participant responses were analyzed by an advisor to this article, Barbara Rogers, CRNP, MN, AOCN®, ANP-BC. Responses revealed that only a minority of participating nurses could correctly grade the severity of two common AEs in lymphoma therapy—peripheral neuropathy and neutropenia. In another activity, a higher proportion of participants could correctly grade infusion reactions, but their ability to grade accurately was far from universal for this potentially serious AE (see Table 1). 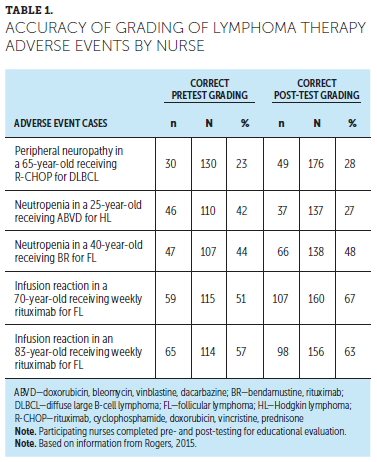
In a blinded survey (Cirillo et al., 2009), nurses graded AEs more accurately than physicians via the well-established criteria of the Common Terminology Criteria for Adverse Events (CTCAE) (see Table 2). In the study, accuracy was measured by the key parameter of patient–clinician agreement (Cirillo et al., 2009; National Cancer Institute [NCI], 2010; Pakhomov, Jacobsen, Chute, & Roger, 2008). However, better accuracy did not always mean higher nurse accuracy. Nurse–patient agreement in the grading of sensory neuropathy, mucositis, and asthenia was 65% or less by kappa coefficient (KC) (defined as the percentage by which agreement exceeds that expected by chance). Nurse–patient agreement was higher for GI AEs (range = 74%KC to 85%KC) (Cirillo et al., 2009). 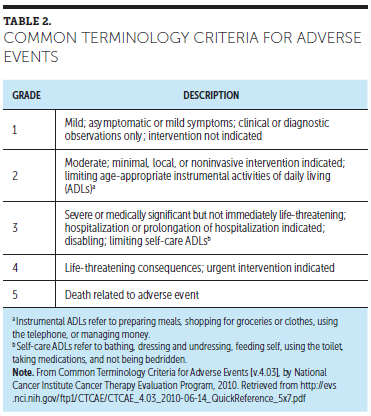
Although nurses may exhibit a skill gap in grading AEs, they display much less of a gap in the identification of AEs. In pre- and post-testing during the ONS Chapters education programs, more than 91% of nurses correctly identified peripheral neuropathy, neutropenia, and infusion reaction related to lymphoma treatment. In addition, survey data show patient–nurse agreement of 75%KC or greater for identification of six treatment-emergent AEs—asthenia, nausea, mucositis, sensory neuropathy, constipation, and diarrhea—all of which may occur during one or more forms of lymphoma therapy (Cirillo et al., 2009).
Barriers to Grading
Undeniably, grading of AEs is difficult. A nurse engaged in grading is engaged in an analytic process that demands high-level critical thinking. Several aspects, however, conspire to impair the nurse’s ability to think critically during grading. The first arises from the CTCAE itself, which has the potential for incomplete reporting (Trotti et al., 2007). CTCAE is clinician-centric rather than patient-centric; it does not capture patient self-report of AEs, severity, or impact. Instead, the CTCAE captures the clinician’s analysis of laboratory-based events (e.g., blood counts), events observable on examination (e.g., tremor), and symptomatic events (e.g., nausea) (Basch et al., 2014; Trotti et al., 2007). However, accruing evidence has shown that patient self-reports of toxicity are a better gauge of health status than clinician reports, and that clinicians underreport the severity of AEs in comparison to patients (Basch et al., 2006, 2009, 2014; Brunner et al., 2011; Dueck et al., 2015; Pakhomov et al., 2008; Quinten et al., 2011). In other words, the CTCAE may fail to provide the evidence needed for patient–clinician agreement on AEs, a key measure of grading validity (Pakhomov et al., 2008). In addition, CTCAE criteria, although in widespread use for decades, have never been formally validated (Trotti et al., 2007), further compromising CTCAE evidence on the severity of AEs. Because evidence is the input for the critical thinking process, attaining the output of accurate grading can be difficult with the current CTCAE.
Another aspect that impedes critical thinking in grading AEs resides in the language of the CTCAE. Grading verbiage has changed between CTCAE version 3.0 (NCI, 2006) and the current version 4.03 (NCI, 2010). Different clinicians may use different terminology depending on the version with which they were trained, leading to inconsistent critical analyses. This problem has been documented for hypersensitivity infusion reactions (DeMoor et al., 2011). A review of 222 cases of hypersensitivity infusion reactions found that different versions of the grading system led to inconsistencies in grading and clinician response to the reaction in about 50% of cases (DeMoor et al., 2011).
The significant weaknesses in the CTCAE has prompted action by the NCI. To overcome the incompleteness and lack of validation, the NCI has spearheaded the development of the Patient-Reported Outcomes CTCAE (PRO-CTCAE) for use in clinical trials (Basch et al., 2014; Brunner et al., 2011). Although the PRO-CTCAE has not been adapted for use in community clinical practice, its validation study did include patients with lymphoma (Basch et al., 2014), and its implementation in the research setting is instructive for all oncology practitioners.
An additional problem in AE grading is the disjunction between nursing workflow and the critical reasoning process needed for grading. The nursing day offers little time for this process. An observational study revealed that frequent task switching and unpredictable demands characterize the nursing workflow (Cornell et al., 2010; Cornell, Riordan, Townsend-Gervis, & Mobley, 2011). For example, in an observational study, 68% of nurse tasks on a pediatric oncology unit were one minute or less in duration (Cornell et al., 2011). As a result, nurses rarely encounter the conditions—such as workflow control and uninterrupted time—needed for critical thinking (Cornell et al., 2010, 2011).
Another issue of concern is nurse visibility and involvement in the care of patients with lymphoma. The less present and visible nurses are, the less they are able to gather reports, histories, and other evidence regarding patients’ AEs. Sustaining a nursing presence requires greater effort than ever because more oral cancer therapies taken by the patient at home have been introduced for the treatment of lymphomas (Yagasaki & Komatsu, 2013). Telephonic assessment is useful for triage of patients and identification of severe AEs but requires experienced clinicians to conduct interviews and supplementation with face-to-face assessments during scheduled office visits (Kondo et al., 2015; Towle, 2009).
Lastly, the specific nature of critical thinking itself within the discipline of nursing can pose problems. Experts in critical thinking in nursing suggest that, for the nurse, critical thinking occurs within social relationships that involve the patient, caregiver, and multidisciplinary care team. To think critically in any context, including the grading of an AE, presupposes insightful, collegial dialogue and shared observation; the nurse cannot make a critical analysis in isolation (Brenner et al., 2008; Raymond-Seniuk & Profetto-McGrath, 2011). This raises a question: In a day full of interruptions and task switching, how can nurses find the time to exchange ideas with others?
Decision Making
Shared decision making is currently a key model in oncology practice (Clark, Nelson, Valerio, Gong, Taylor-Fishwick, & Fletcher, 2009; Frerichs, Hahlweg, Müller, Adis, & Scholl, 2016). Across the interprofessional team and between clinicians and patients, shared decision making is an empowering partnership in care (Clark et al., 2009). When a clinician shares decision making with a patient, three supportive roles appear to be most important: help the patient understand the issue being decided, listen to the patient’s concerns, and include in discussions what matters most to the issue (Elwyn et al., 2012; Katz, Belkora, & Elwyn, 2014). Shared decision making may be impeded by limitations to critical thought. For example, some patients are reluctant to share decision making because they engage in “counterfactual thinking,” meaning that they may anticipate feeling regret or self-recrimination if they share in a decision, like stopping a particular drug related to AEs and then, months or years later, experience disease progression (Katz et al., 2014). When nurses have difficulty recognizing and analyzing such reasoning, they may be unable to fully empower the patient as a decision maker.
Within the context of shared decision making, the nurse may be called on to offer evidence, in the form of AE grading and narrative description, with critical consideration of quality of life and distress related to the AE. With this information, the nurse helps the patient participate in the decision to hold treatment or reduce the dose. With systemic therapies, the development of a toxicity and establishing its grade typically determines the decision to continue, reduce, or hold treatment. For example, the decision to reduce bortezomib dose from 1.3 mg/m2 to 1 mg/m2 in mantle-cell lymphoma requires precision in grading. If a patient has grade 1 neuropathy with no impact on quality of life and no pain, then no bortezomib dose adjustment is required. However, grade 2 neuropathy with pain (with limitations on instrumental activities of daily living) and grade 3 neuropathy (limitations on self-care activities of daily living) require holding bortezomib until the neurologic toxicity resolves (Millennium Pharmaceuticals, Inc., 2015; NCI, 2010). Misgrading could produce dosing or treatment error.
Medication errors are not uncommon, including giving the wrong dose and giving a dose after a discontinuation order (Ford, Killebrew, Fugitt, Jacobsen, & Prystas, 2006). Nurses attribute errors to factors that unfavorably influence critical thinking (Cornell et al., 2010, 2011), such as adding tasks to the workload, chaos in the work environment, being swamped or overwhelmed, emotional responses, and a loss of focus (Roth, Wieck, Fountain, & Haas, 2015; Valiee, Peyrovi, & Nasrabadi, 2014). Of note, nurses themselves have cited a lack of critical thinking as one of the top five most likely causes of nursing errors in the hospital (Roth et al., 2015).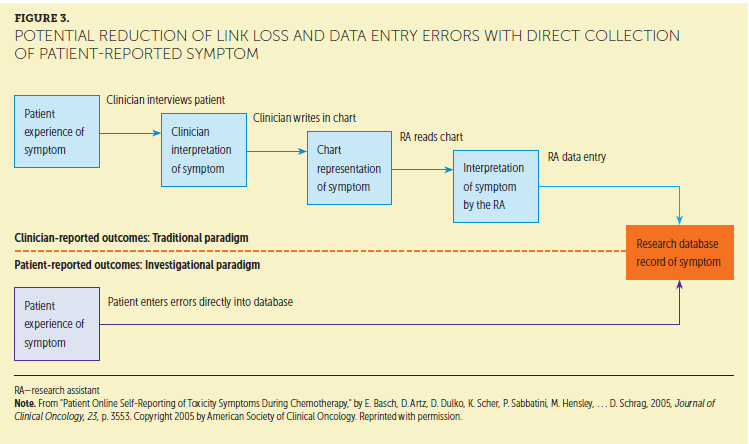
Communication
Even when AEs are graded accurately, communication of the grade and recommended approaches can pose challenges. Barriers to effective communication may result from deficits in critical thinking relevant to managing AEs in lymphoma. These deficits include the following:
• Lack of reasoning across relationships—an inability to understand, anticipate, and compensate for the types of errors that may occur between patient report, upload to the medical record, interpretation and CTCAE grading, and subsequent team discussion. These errors can include gaps in communication; loss of shared interpretation of symptoms among professionals, patients, and caregivers (link loss); and recording and documentation errors (Basch et al., 2005) (see Figure 3).
• Problems in evidence processing—keeping up with new toxicities when a novel therapy or regimen is introduced in the already complex care of lymphoma (e.g., diarrhea and cough with idelalisib in follicular lymphoma) is challenging (Coutré et al., 2015).
• Using a terse or incomplete narrative to describe an AE, which does not take into account the listener’s need for evidence—for example, communicating grade only by the number, rather than also describing its clinical features. An example of this would be writing or saying only “grade 3 fatigue” with lenalidomide without providing information on the patient’s inability to find relief with rest, specific limitations in daily activities, and how well the patient is coping (Celgene Corporation, 2015; NCI, 2010); incomplete narrative also means that a nurse may not have documented the details needed to trigger accurate grading.
• Lack of curiosity and self-reflection limits the ability to ask questions, solicit feedback, and identify misunderstandings.
Improving Practice: Tools for Thinking Critically and Acting Decisively
The nursing community has been an important contributor in education to improve critical thinking (Delibovi, 2015). Oncology nurses can use the guidance of nurse educators to enhance critical thinking to improve accuracy in the grading of AEs, to increase precision and confidence in recommendations for the adjustment of therapy, and to communicate recommended approaches clearly and convincingly to aid in the process of shared decision making.
Cultivating Critical Thinking Skills
The nursing literature has identified several important skills for critical thinking among nurses (Brenner et al., 2008; Papathanasiou, Kleisaris, Fradelos, Kakou, & Kourkouta, 2014: Papp et al., 2014; Phelps et al., 2009; Raymond-Seniuk & Profetto-McGrath, 2011):
• Flexibility—the ability to remain curious, to ask questions, to stay alert for unexpected circumstances, and to alter preconceived notions on the basis of observations
• Willingness to analyze situations and statements deeply
• A systematic and orderly approach to all decision making, including a consistent methodology and use of evidence to support decisions
• A focus on problem solving
• The ability to expect, understand, and embrace differences in perspective between oneself and other people
• Self-reflectiveness and the ability to admit error and uncertainty.
These skills are highly relevant to best practices in the process of AE grading, decision making, and communicating in antilymphoma therapy (see Figure 4). For example, even a simple best practice, such as comparing a blood count or a report of neuropathy to the actual text of the CTCAE, cannot be fully actualized without critical thinking. The reason is that patient data and best practices qualify as what nurse educators term "messy information" (Rowles, Morgan, Burns, & Merchant, 2013). Patient data can be scattered, fragmented, or incomplete. Best practices may be gathered from various potentially conflicting sources, such as cancer center memos, published guidelines, first-person articles by experts, facility medical directors, and even third-party payors. Often, no guidance may exist on how to apply a best practice to a particular patient scenario. 
What should be done, for instance, if a patient reports trouble buttoning his shirt, but is unable to answer most of the symptom-focused questions on the cancer center’s peripheral neuropathy assessment tool? To solve this problem, a nurse might analyze the patient’s report deeply, reach out once more to the patient for additional evidence of neuropathy, and try to understand the difference between the clinical perspective and the patient’s perspective on his symptoms—three of the critical thinking skills cited earlier in this article. Therefore, to follow best practices, critical thinking is required to assimilate and use the messy information pertinent to each case (Rowles et al., 2013).
Critical Thinking in Relationship
Considering the perspectives of others is a particularly meaningful part of the nursing skill set (Papp et al., 2014; Raymond-Seniuk & Profetto-McGrath, 2011). Part of the definition of a challenged thinker, a nurse who struggles to think critically, is resistance to consideration of others’ perspectives (Papp et al., 2014). The effort to “think in relationship” has profound and positive effects on communication regarding AEs and adjustment of therapy. When explaining an AE or recommending a dose adjustment, it matters who receives the explanation or recommendation. Depending on prior experience, healthcare professionals, patients, and caregivers may have different levels of familiarity with grading schema. Therefore, when communicating with different individuals, the nurse cannot assume that writing or saying “grade 3 sensory neuropathy” will be sufficient. To take into account others’ perspectives, a statement of the grade should be accompanied by a narrative description of the AE that include features, such as onset, location, and duration; conditions that worsen or alleviate the AE; the nature of the symptoms; severity; and interference with quality of life. Nurses may even want to create a template or reminder to include these features that functions as a verbal or written script for communication. Scripting is a technique borrowed from successful nursing education for critical thinking (Su & Juestel, 2010), and it may be a useful tool in clinical practice as well.
In antilymphoma therapy, a key aspect of the patient–nurse relationship, nursing presence, has become more challenging. Challenges to nurse visibility stem from the growing number of oral, self-administered therapies available (Yagasaki & Komatsu, 2013), including approved therapies such as lenalidomide (mantle- cell lymphoma), vorinostat (cutaneous T-cell lymphoma), idelalisib (follicular lymphoma, small lymphocytic lymphoma), and others (Celgene Corporation, 2015; Gilead Sciences, 2014; Merck Sharp & Dohme Co., 2015). When patients receive oral anti-lymphoma therapy, “thinking in relationship” is an important nursing skill that involves understanding the perspective of the patient at home. This patient may be hesitant to make a call to report an AE, may forget the importance of watching for AEs, or may generally feel more distant from the nurse. Understanding the patient’s perspective in such cases can prompt nurses to make more frequent and vigorous outreach by phone or email or on the occasion of a visit to the cancer center.
Leverage Learning From the PRO-CTCAE
The patient-centric PRO-CTCAE has been shown to be valid, reliable, and responsive, indicating that its patient-centeredness is an asset (Dueck et al., 2015). It also is designed to help minimize communication gaps, link loss, and problems in recording error (Basch et al., 2005, 2014). PRO-CTCAE use is gaining momentum in the clinical trial setting, but is not yet widely used in clinical care.
Questions posed to patients via the PRO-CTCAE were designed with three features: plain language terminology for the symptom; a focus on eliciting frequency, severity, and interference with activity from the patient; and a mechanism to gather the recall period for the symptom (Basch et al., 2014). Sample question formats include: “In the past, how OFTEN did you have [symptom]?”; “In the past, what was the SEVERITY of your [symptom] at its worst?”; “In the past, how much did [symptom] INTERFERE with your usual or daily activities?” (Basch et al., 2014, p. 1). Data have shown a strong consensus among key stakeholders in the implementation of the PRO-CTCAE that including patient reporting of adverse symptoms would be useful for improving understanding of the patient experience in a cancer clinical trial (Brunner et al., 2011). In addition, a high proportion of stakeholders endorsed administration of PRO-CTCAE clinical trials to improve completeness, accuracy, and efficiency of AE data collection (Brunner et al., 2011) (see Figure 5). These findings may suggest that nurses in community clinical practice may gather better input for critical analysis of CTCAE grade by asking patients to describe their own experience of frequency, severity, and interference with an AE, potentially leading to more accurate grading.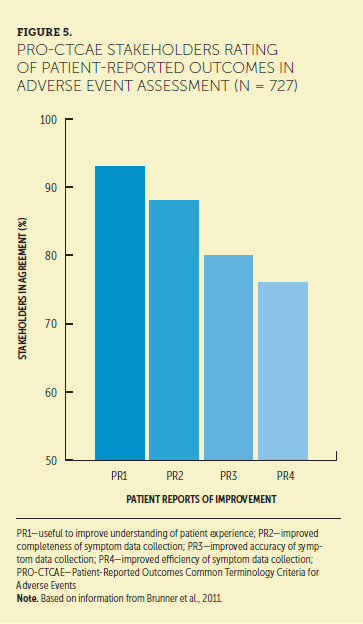
Manage Workflow
Chaos and task switching are part of the nursing day, but they are not conducive to the critical thinking needed for accurate grading, confident decision making on the adjustment of therapy, and well-organized communication (Cornell et al., 2010, 2011). Interventions to improve workflow may have a positive impact on critical thinking (Cornell et al., 2011). These include methods that nurses can deploy themselves, such as seeking out education on time and workflow management, delegation of routine or repetitive tasks to non-nursing staff, and more skillful and systematic use of technology (Cornell et al., 2011). Other methods must be championed at the institutional level, such as workflow evaluation and redesign, implementation of better software systems, and reallocation of duties (Cornell et al., 2011).
Conclusion
In antilymphoma therapy, proper management of AEs is essential to good clinical outcomes. The response to AEs cannot be optimal without precise grading of toxicities, adjustment of therapy based on grade and AE description, and effective interdisciplinary and patient–clinician communication. Building nursing competency in these areas requires development of critical thinking, which enables nurses to implement best practices decisively and appropriately in each clinical case.
The authors gratefully acknowledge Deborah Watkins Bruner, RN, PhD, FAAN, Kevin Brigle, PhD, NP, Katherine L. Byar, MSN, APN, BC, BMTCN®, and Barbara Rogers CRNP, MN, AOCN®, ANP-BC, for their expert advice.
About the Author(s)
Amy Goodrich, MSN, CRNP, is a nurse practitioner and Nina Wagner-Johnston, MD, is an associate professor of oncology, both in the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins University in Baltimore, MD; and Dana Delibovi, MA, MS, is a bioscience writer and an adjunct professor in the Philosophy Department at Lindenwood University in St. Charles, MO. Goodrich can be reached at goodram@jhmi.edu, with copy to CJONEditor@ons.org. (Submitted September 2016. Accepted November 12, 2016.). The authors take full responsibility for this content. The article has been reviewed by independent peer reviewers to ensure that it is objective and free from bias. Mention of specific products and opinions related to those products do not indicate or imply endorsement by the Oncology Nursing Society.
References
Ansell, S.M., Lesokhin, A.M., Borrello, I. Halwani, A., Scott, E.C., Guitierrez, M., . . . Armand, P. (2013). PD-1 blockade with nivolumab in relapsed or refractory Hodgkin’s lymphoma. New England Journal of Medicine, 372, 311–319. doi:10.1056/NEJMoa1411087
Basch, E., Artz, D., Dulko, D., Scher, K., Sabbatini, P., Hensley, M., . . . Schrag, D. (2005). Patient online self-reporting of toxicity symptoms during chemotherapy. Journal of Clinical Oncology, 23, 3553–3561.
Basch, E., Iasonos, A., McDonough, T., Barz, A., Culkin, A., Kris, M.G., . . . Schrag, D. (2006). Patient versus clinician symptom reporting using the National Cancer Institute common terminology criteria for adverse events. Lancet Oncology, 7, 903–909.
Basch, E., Jia, X., Heller, G., Barz, A., Sit, L., Fruscione, M., . . . Schrag, D. (2009). Adverse symptom event reporting by patients vs clinicians: Relationships with clinical outcomes. Journal of the National Cancer Institute, 101, 1624–1632. doi:10.1093/jnci/djp386
Basch, E., Reeve, B.B., Mitchell, S.A., Clauser, S.B., Minasian, L.M., Dueck, A.C., . . . Schrag, D. (2014). Development of the National Cancer Institute’s patient-reported outcomes version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE). Journal of the National Cancer Institute, 106, dju244. doi:10.1093/jnci/dju244
Baxter Healthcare. (2010). Endoxan™ (cyclophosphamide) [Package insert]. Retrieved from http://www.baxterhealthcare.com.au/downloads/healthcare_professionals/c…
Brenner, P., Hughes, R.G., & Sutphen, M. (2008) Clinical reasoning, decision-making, and action: Thinking critically and clinically. In R.G. Hughes (Ed.), Patient safety and quality: An evidence-based handbook for nurses (pp. 87–109). Rockville, MD: Agency for Healthcare Research and Quality.
Brunner, D.W., Hanisch, L.J., Reeve, B.B., Trotti, A.M., Schrag, D., Sit, L., . . . Basch, E.M. (2011). Stakeholder perspectives on implementing the National Cancer Institute’s patient-reported outcomes version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE). Translational Behavioral Medicine, 1, 110–122. doi:10.1007/s13142-011-0025-3
Celgene Corporation. (2015). Revlimid® (lenalidomide) [Package insert]. Retrieved from http://www.revlimid.com/wp-content/uploads/full-prescribing-information…
Cirillo, M., Venturini, M., Ciccarelli, L., Coati, F., Bortolami, O., & Verlato, G. (2009). Clinician versus nurse symptom reporting using the National Cancer Institute-Common Terminology Criteria for Adverse Events during chemotherapy: Results of a comparison based on patient’s self-reported questionnaire. Annals of Oncology, 20, 1929–1935. doi:10.1093/annonc/mdp287
Clark, N.M., Nelson, B.W., Valerio, M.A., Gong, Z.M., Taylor-Fishwick, J.C., & Fletcher, M. (2009). Consideration of shared decision making in nursing: A review of clinicians’ perceptions and interventions. Open Nursing Journal, 3, 65–75. doi:10.2174/1874434600903010065
Coiffier, B., Lepage, E., Briere, J., Herbrecht, R., Tilly, H., Bouabdallah, R., . . . Gisselbrecht, C. (2002). CHOP chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large-B-cell lymphoma. New England Journal of Medicine, 346, 235–242.
Cornell, P., Herrin-Griffith, D., Keim, C., Petschonek, S., Sanders, A.M., D’Mello, S., . . . Shepherd, G. (2010). Transforming nursing workflow, part 1: The chaotic nature of nurse activities. Journal of Nursing Administration, 40, 366–373. doi:10.1097/NNA.0b013e3181ee4261
Cornell, P., Riordan, M., Townsend-Gervis, M., & Mobley, R. (2011). Barriers to critical thinking: Workflow interruptions and task switching among nurses. Journal of Nursing Administration, 41, 407–414. doi:10.1097/NNA.0b013e31822edd42
Coutré, S.E., Barrientos, J.C., Brown, J.R., de Vos, S., Furman, R.R., Keating, M.J., . . . Zelenetz, A.D. (2015). Management of adverse events associated with idelalisib treatment: Expert panel opinion. Leukemia and Lymphoma, 56, 2779–2786. doi:10.3109/10428194.2015.1022770
Delibovi, D. (2015). Critical thinking and character. In S. Wisdom, & L. Leavitt (Eds.), Handbook of research on advancing critical thinking in higher education (pp. 21–47). Hershey, PA: IGI Global.
DeMoor, P.A., Matusov, Y., Kelly, C., Kolan, S., Barnachea, L., & Bazhenova, L.A. (2011). A retrospective review of the frequency and nature of acute hypersensitivity reactions at a medium-sized infusion center: Comparison to reported values and inconsistencies found in literature. Journal of Cancer, 2, 153–164.
Dueck, A.C., Mendoza, T.R., Mitchell, S.A., Reeve, B.B., Castro, K.M., Rogak, L.J., . . . Basch, E. (2015). Validity and reliability of the US National Cancer Institute’s Patient-Reported Outcomes Version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE). JAMA Oncology, 1, 1051–1059. doi:10.1001/jamaoncol.2015.2639
Duvic, M., Olsen, E.A., Breneman, D., Pacheco, T.R., Parker, S., Vonderheid, E.C., . . . Geskin, L.J. (2009). Evaluation of the long-term tolerability and clinical benefit of vorinostat in patients with advanced cutaneous T-cell lymphoma. Clinical Lymphoma and Myeloma, 9, 412–416. doi:10.3816/CLM.2009.n.082
Elwyn, G., Frosch, D., Thomson, R., Joseph-Williams, N., Lloyd, A., Kinnersley, P., . . . Barry, M. (2012). Shared decision making: A model for clinical practice. Journal of General Internal Medicine, 27, 1361–1367.
Ford, C.D., Killebrew, J., Fugitt, P., Jacobsen, J., & Prystas, E.M. (2006). Study of medication errors on a community hospital oncology ward. Journal of Oncology Practice, 2(4), 149–154.
Fowler, N., Kahl, B.S., Lee, P., Matous, J.V., Cashen, A.F., Jacobs, S.A., . . . Cheson, B.D. (2011). Bortezomib, bendamustine, and rituximab in patients with relapsed or refractory follicular lymphoma: The phase II VERTICAL study. Journal of Clinical Oncology, 29, 3389–3395. doi:10.1200/JCO.2010.32.1844
Fowler, N.H., Davis, R.E., Rawal, S., Nastoupil, L., Hagemeister, F.B., McLaughlin, P., . . . Neelapu, S.S. (2014). Safety and activity of lenalidomide and rituximab in untreated indolent lymphoma: An open-label, phase 2 trial. Lancet, 15, 1311–1318. doi:10.1016/S1470-2045(14)70455-3
Frerichs, W., Hahlweg, P., Müller, E., Adis, C., & Scholl, I. (2016). Shared decision-making in oncology—A qualitative analysis of healthcare providers’ views in current practice. PLOS ONE, 11, e0149789. doi:10.1371/journal.pone.0149789
Genentech. (2014). Rituxan® (rituximab) [Package insert]. Retrieved from https://www.gene.com/download/pdf/rituxan_prescribing.pdf
Genzyme Corporation. (2014). Campath® (alemtuzumab) [Package insert]. Retrieved from http://www.campath.com/pdfs/2014-09-Campath_US_PI.pdf
Gilead Sciences. (2014). Zydelig® (idelalisib) [Package insert]. Retrieved from http://www.gilead.com/~/media/files/pdfs/medicines/oncology/zydelig/zyd…
Grisold, W., Cavaletti, G., & Windebank, A.J. (2012). Peripheral neuropathies from chemotherapeutics and targeted agents: Diagnosis, treatment, and prevention. Neuro-Oncology, 14(Suppl. 4), iv45–iv54. doi:10.1093/neuonc/nos203
Johnston, P.B., Inwards, D.J., Colgan, J.P., Laplant, B.R., Kabat, B.F., Habermann, T.M., . . . Witzig, T.E. (2010). A phase II trial of the oral mTOR inhibitor everolimus in relapsed Hodgkin lymphoma. American Journal of Hematology, 85, 320–324. doi:10.1002/ajh.21664
Katz, S.J., Belkora, J., & Elwyn, G. (2014). Shared decision making for treatment of cancer: Challenges and opportunities. Journal of Oncology Practice, 10, 206–208. doi:10.1200/JOP.2014.001434
Kondo, S., Shiba, S., Udagawa, R., Ryushima, Y., Yano, M., Uehara, T., . . . Hashimoto, J. (2015). Assessment of adverse events via a telephone consultation service for cancer patients receiving ambulatory chemotherapy. BMC Research Notes, 8, 315.
McFadden, E.V., Poniatowski, B., & Temple, S.V. (Eds). (2006). Contemporary issues in lymphoma: A nursing perspective. Sudbury, MA: Jones and Bartlett.
Merck Sharp & Dohme Co. (2015). Zolinza® (vorinostat) [Package insert]. Retrieved from https://www.merck.com/product/usa/pi_circulars/z/zolinza/zolinza_pi.pdf
Millennium Pharmaceuticals, Inc. (2015). Velcade® (bortezomib) [Package insert]. Retrieved from http://www.velcade.com/files/PDFs/VELCADE_PRESCRIBING_INFORMATION.pdf
Moskowitz, C.H., Nademanee, A., Masszi, T., Agura, E., Holowiecki, J., Abidi, M.H., . . . Walewski, J. (2015). Brentuximab vedotin as consolidation therapy after autologous stem-cell transplantation in patients with Hodgkin’s lymphoma at risk of relapse or progression (AETHERA): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet, 385, 1853–1862. doi:10.1016/S0140-6736(15)60165-9
National Cancer Institute. (2006). Common Terminology Criteria for Adverse Events (CTCAE) [v.3.0]. Retrieved from https://ctep.cancer.gov/protocoldevelopment/electronic_applications/doc…
National Cancer Institute. (2010). Common Terminology Criteria for Adverse Events (CTCAE) [v.4.03]. Retrieved from http://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03_2010-06-14_QuickReference_…
National Comprehensive Cancer Network. (2016a). NCCN Clinical Practice Guidelines in Oncology: Antiemesis [v.2.2016] Retrieved from https://www.nccn.org/professionals/physician_gls/pdf/antiemesis.pdf
National Comprehensive Cancer Network. (2016b). NCCN Clinical Practice Guidelines in Oncology: Cancer-related fatigue [v.1.2016]. Retrieved from https://www.nccn.org/professionals/physician_gls/pdf/fatigue.pdf
National Comprehensive Cancer Network. (2016c). NCCN Clinical Practice Guidelines in Oncology: Hodgkin lymphoma [v.3.2016]. Retrieved from https://www.nccn.org/professionals/physician_gls/pdf/hodgkins.pdf
National Comprehensive Cancer Network. (2016d). NCCN Clinical Practice Guidelines in Oncology: Non-Hodgkin’s lymphomas [v.3.2016]. Retrieved from https://www.nccn.org/professionals/physician_gls/pdf/nhl.pdf
Newport, F. (2012). Congress retains low honesty rating. Retrieved from http://www.gallup.com/poll/159035/congress-retains-low-honesty-rating.a…
Oncology Nursing Society. (2016). PEP rating system overview. Retrieved from https://www.ons.org/practice-resources/pep
Pakhomov, S.V., Jacobsen, S.J., Chute, C.G., & Roger, V.L. (2008). Agreement between patient-reported symptoms and their documentation in the medical record. American Journal of Managed Care, 14, 530–539.
Papathanasiou, I.V., Kleisiaris, C.F., Fradelos, E.C., Kakou, K., & Kourkouta, L. (2014). Critical thinking: The development of an essential skill for nursing students. Acta Informatica Medica, 22, 283–286. doi:10.5455/aim.2014.22.283-286
Papp, K.K., Huang, G.C., Lauzon Clabo, L.M., Delva, D., Fischer, M., Konopasek, L., . . . Gusic, M. (2014). Milestones of critical thinking: A developmental model for medicine and nursing. Academic Medicine, 89, 715–720. doi:10.1097/ACM.0000000000000220
Pfizer Laboratories. (2015). Adriamycin® (doxorubicin hydrochloride) [Package insert]. Retrieved from http://www.pfizer.com/system/files/products/material_safety_data/Doxoru…
Phelps, S.E., Wallen, G., Cusack, G., Castro, K., Muehlbauer, P., Shelburne, N., & Woolery, M. (2009). Staff development story: Concept mapping: A staff development strategy for enhancing oncology critical thinking. Journal for Nurses in Staff Development, 25, 42–47. doi:10.1097/NND.0b013e3181963849
Quinten, C., Maringwa, J., Gotay, C.C., Martinelli, F., Coens, C., Reeve, B.B., . . . Bottomley, A. (2011). Patient self-reports of symptoms and clinician ratings as predictors of overall cancer survival. Journal of the National Cancer Institute, 103, 1851–1858. doi:10.1093/jnci/djr485
Raymond-Seniuk, C., & Profetto-McGrath, J. (2011). Can one learn to think critically?—A philosophical exploration. Open Nursing Journal, 5, 45–51. doi:10.2174/1874434601105010045
Rogers, B. (2015). [Supplement report to outcomes measurement report for: Identifying, preventing, and treating serious side effects leading to termination of therapy in lymphoma.] Unpublished raw data.
Roth, C., Wieck, K.L., Fountain, R., & Haas, B.K. (2015). Hospital nurses’ perceptions of human factors contributing to nursing errors. Journal of Nursing Administration, 45, 263–269. doi:10.1097/NNA.0000000000000196
Rowles, J., Morgan, C., Burns, S., & Merchant, C. (2013). Faculty perceptions of critical thinking at a health sciences university. Journal of the Scholarship of Teaching and Learning, 13(4), 21–35.
Rummel, M.J., Niederle, N., Maschmeyer, G., Banat, G.A., von Grünhagen, U., Losem, C., . . . Brugger, W. (2013). Bendamustine plus rituximab versus CHOP plus rituximab as first-line treatment for patients with indolent and mantle-cell lymphomas: An open-label, multicentre, randomised, phase 3 non-inferiority trial. Lancet, 381, 1203–1210. doi:10.1016/S0140-6736(12)61763-2
Schulmeister, L. (2006). Preventing chemotherapy errors. Oncologist, 11, 463–468.
Seattle Genetics. (2015). Adcetris® (brentuximab vedotin) [Package insert]. Retrieved from http://www.seattlegenetics.com/pdf/adcetris_USPI.pdf
Su, W.M., & Juestel, M.J. (2010). Direct teaching of thinking skills using clinical simulation. Nurse Educator, 35(5), 197–204. doi:10.1097/NNE.0b013e3181e33969
Teva Pharmaceuticals. (2015). Trenada® (bendamustine hydrochloride) [Package insert]. Retrieved from http://www.treandahcp.com/pdf/TREANDA_final_PI.pdf
Towle, E. (2009). Telephone triage in today’s oncology practice. Journal of Oncology Practice, 5, 61. doi:10.1200/JOP.0921502
Trotti, A., Colevas, A.D., Setser, A., & Basch, E. (2007). Patient-reported outcomes and the evolution of adverse event reporting in oncology. Journal of Clinical Oncology, 25, 5121–5127.
Valiee, S., Peyrovi, H., & Nasrabadi, A.N. (2014). Critical care nurses’ perception of nursing error and its causes: A qualitative study. Contemporary Nurse, 46, 206–213. doi:10.5172/conu.2014.46.2.206
Yagasaki, K., & Komatsu, H. (2013). The need for a nursing presence in oral chemotherapy. Clinical Journal of Oncology Nursing, 17, 512–516. doi:10.1188/13.CJON.512-516




